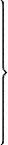Project Gutenberg's Conversations on Chemistry, V. 1-2, by Jane Marcet
This eBook is for the use of anyone anywhere at no cost and with
almost no restrictions whatsoever. You may copy it, give it away or
re-use it under the terms of the Project Gutenberg License included
with this eBook or online at www.gutenberg.org
Title: Conversations on Chemistry, V. 1-2
In Which the Elements of that Science Are Familiarly
Explained and Illustrated by Experiments
Author: Jane Marcet
Release Date: October 13, 2008 [EBook #26908]
Language: English
Character set encoding: UTF-8
*** START OF THIS PROJECT GUTENBERG EBOOK CONVERSATIONS ON CHEMISTRY, V. 1-2 ***
Produced by Louise Hope
Caution: DO NOT TRY THIS AT HOME.
This text uses utf-8 (unicode)
file encoding. If the apostrophes and quotation marks in this paragraph
appear as garbage, you may have an incompatible browser or unavailable
fonts. First, make sure that the browser’s “character set” or “file
encoding” is set to Unicode (UTF-8). You may also need to change your
browser’s default font.
The original book was published in two volumes. The format is
reproduced for this e-text, except that the author’s preface (originally
in Volume I) and the combined index (Volume II) are in this
introductory file.
See the end of this file for notes on
scientific terminology, spelling, Plates and chapter numbering.
CONVERSATIONS
ON
CHEMISTRY;
IN WHICH
THE ELEMENTS OF THAT SCIENCE
ARE
FAMILIARLY EXPLAINED
AND
ILLUSTRATED BY EXPERIMENTS.
IN TWO VOLUMES.
The Fifth Edition, revised, corrected, and
considerably enlarged.

LONDON:
PRINTED FOR LONGMAN, HURST, REES, ORME, AND BROWN,
PATERNOSTER-ROW.
1817.
Printed by A. Strahan,
Printers-Street, London.
ADVERTISEMENT.
The Author, in this fifth edition, has
endeavoured to give an account of the principal discoveries which have
been made within the last four years in Chemical Science, and of the
various important applications, such as the gas-lights, and the
miner’s-lamp, to which they have given rise. But in regard to doctrines
or principles, the work has undergone no material alteration.
London, July, 1817.
v
In venturing to offer to the public, and
more particularly to the female sex, an Introduction to Chemistry, the
author, herself a woman, conceives that some explanation may be
required; and she feels it the more necessary to apologise for the
present undertaking, as her knowledge of the subject is but recent, and
as she can have no real claims to the title of chemist.
On attending for the first time experimental lectures, the author
found it almost impossible to derive any clear or satisfactory
information from the rapid demonstrations which are usually, and perhaps
necessarily, crowded into popular courses of this kind. But frequent
opportunities having
vi
afterwards occurred of conversing with a friend on the subject of
chemistry, and of repeating a variety of experiments, she became better
acquainted with the principles of that science, and began to feel highly
interested in its pursuit. It was then that she perceived, in attending
the excellent lectures delivered at the Royal Institution, by the
present Professor of Chemistry, the great advantage which her previous
knowledge of the subject, slight as it was, gave her over others who had
not enjoyed the same means of private instruction. Every fact or
experiment attracted her attention, and served to explain some theory to
which she was not a total stranger; and she had the gratification to
find that the numerous and elegant illustrations, for which that school
is so much distinguished, seldom failed to produce on her mind the
effect for which they were intended.
Hence it was natural to infer, that familiar conversation was, in
studies of this kind, a most useful auxiliary source of
information;
vii
and more especially to the female sex, whose education is seldom
calculated to prepare their minds for abstract ideas, or scientific
language.
As, however, there are but few women who have access to this mode of
instruction; and as the author was not acquainted with any book that
could prove a substitute for it, she thought that it might be useful for
beginners, as well as satisfactory to herself, to trace the steps by
which she had acquired her little stock of chemical knowledge, and to
record, in the form of dialogue, those ideas which she had first derived
from conversation.
But to do this with sufficient method, and to fix upon a mode of
arrangement, was an object of some difficulty. After much hesitation,
and a degree of embarrassment, which, probably, the most competent
chemical writers have often felt in common with the most superficial,
a mode of division was adopted, which, though the most natural,
does not always admit of being
viii
strictly pursued—it is that of treating first of the simplest
bodies, and then gradually rising to the most intricate compounds.
It is not the author’s intention to enter into a minute vindication
of this plan. But whatever may be its advantages or inconveniences, the
method adopted in this work is such, that a young pupil, who should
occasionally recur to it, with a view to procure information on
particular subjects, might often find it obscure or unintelligible; for
its various parts are so connected with each other as to form an
uninterrupted chain of facts and reasonings, which will appear
sufficiently clear and consistent to those only who may have patience to
go through the whole work, or have previously devoted some attention to
the subject.
It will, no doubt, be observed, that in the course of these
Conversations, remarks are often introduced, which appear much too acute
for the young pupils, by whom
ix
they are supposed to be made. Of this fault the author is fully aware.
But, in order to avoid it, it would have been necessary either to omit a
variety of useful illustrations, or to submit to such minute
explanations and frequent repetitions, as would have rendered the work
tedious, and therefore less suited to its intended purpose.
In writing these pages, the author was more than once checked in her
progress by the apprehension that such an attempt might be considered by
some, either as unsuited to the ordinary pursuits of her sex, or
ill-justified by her own recent and imperfect knowledge of the subject.
But, on the one hand, she felt encouraged by the establishment of those
public institutions, open to both sexes, for the dissemination of
philosophical knowledge, which clearly prove that the general opinion no
longer excludes women from an acquaintance with the elements of science;
and, on the other, she flattered herself that whilst the impressions
made upon her mind, by the wonders
x
of Nature, studied in this new point of view, were still fresh and
strong, she might perhaps succeed the better in communicating to others
the sentiments she herself experienced.
The reader will soon perceive, in perusing this work, that he is
often supposed to have previously acquired some slight knowledge of
natural philosophy, a circumstance, indeed, which appears very
desirable. The author’s original intention was to commence this work by
a small tract, explaining, on a plan analogous to this, the most
essential rudiments of that science. This idea she has since abandoned;
but the manuscript was ready, and might, perhaps, have been printed at
some future period, had not an elementary work of a similar description,
under the tide of “Scientific Dialogues,” been pointed out to her,
which, on a rapid perusal, she thought very ingenious, and well
calculated to answer its intended object.
Page numbers have been retained to give an idea of the relative length
of each Conversation.
I.xvi
ERRATA.
| Vol. I. page 56. |
last line but one, for “caloric,” read “calorific.” |
| 179. |
Note, for “Plate XII.” r. “Plate XIII.” |
CONVERSATIONS
ON
CHEMISTRY;
IN WHICH
THE ELEMENTS OF THAT SCIENCE
ARE
FAMILIARLY EXPLAINED
AND
ILLUSTRATED BY EXPERIMENTS.
IN TWO VOLUMES.
The Fifth Edition, revised, corrected, and
considerably enlarged.
VOL. I.
ON SIMPLE BODIES.

LONDON:
PRINTED FOR LONGMAN, HURST, REES, ORME, AND BROWN,
PATERNOSTER-ROW.
1817.
xi
OF
THE FIRST VOLUME.
ON SIMPLE BODIES.
|
CONVERSATION I. |
|
| ON THE GENERAL PRINCIPLES OF CHEMISTRY. |
Page 1 |
Connexion between Chemistry and Natural
Philosophy.—Improved State of modern Chemistry.—Its use in
the Arts.—The general Objects of Chemistry.—Definition of
Elementary Bodies.—Definition of Decomposition.—Integrant
and Constituent Particles.—Distinction between Simple and Compound
Bodies.—Classification of Simple Bodies.—Of Chemical
Affinity, or Attraction of Composition.—Examples of Composition
and Decomposition. |
|
|
CONVERSATION II. |
|
| ON LIGHT AND HEAT. |
26 |
Light and Heat capable of being separated.—Dr. Herschel’s
Experiments.—Phosphorescence.—Of Caloric.—Its two
Modifications.—Free Caloric.—Of the three
xii
different States of Bodies, solid, fluid, and aeriform.—Dilatation
of solid Bodies.—Pyrometer.—Dilatation of
Fluids.—Thermometer.—Dilatation of Elastic Fluids.—Air
Thermometer.—Equal Diffusion of Caloric.—Cold a Negative
Quality.—Professor Prevost’s Theory of the Radiation of
Heat.—Professor Pictet’s Experiments on the Reflexion of
Heat.—Mr. Leslie’s Experiments on the Radiation of
Heat. |
|
|
CONVERSATION III. |
|
| CONTINUATION OF THE SUBJECT. |
70 |
Of the different Power of Bodies to conduct Heat.—Attempt
to account for this Power.—Count Rumford’s Theory of the
non-conducting Power of Fluids.—Phenomena of Boiling.—Of
Solution in general.—Solvent Power of Water.—Difference
between Solution and Mixture.—Solvent Power of Caloric.—Of
Clouds, Rain, Dr. Wells’ theory of Dew, Evaporation,
&c.—Influence of Atmospherical Pressure on
Evaporation.—Ignition. |
|
|
CONVERSATION IV. |
|
| ON COMBINED CALORIC, COMPREHENDING SPECIFIC HEAT
AND LATENT HEAT. |
122 |
Of Specific Heat.—Of the different Capacities of Bodies for
Heat.—Specific Heat not perceptible by the Senses.—How to be
ascertained.—Of Latent Heat.—Distinction between Latent and
Specific Heat.—Phenomena attending the Melting of Ice and the
Formation of Vapour.—Phenomena attending the Formation of Ice, and
the Condensation of Elastic Fluids.—Instances of Condensation, and
consequent Disengagement of Heat, produced by Mixtures, by the Slaking
of Lime.—General
xiii
Remarks on Latent Heat.—Explanation of the Phenomena of Ether
boiling, and Water freezing, at the same Temperature.—Of the
Production of Cold by
Evaporation.—Calorimeter.—Meteorological Remarks. |
|
|
CONVERSATION V. |
|
| ON THE CHEMICAL AGENCIES OF ELECTRICITY. |
160 |
Of Positive and Negative Electricity.—Galvani’s
Discoveries.—Voltaic Battery.—Electrical
Machine.—Theory of Voltaic Excitement. |
|
|
CONVERSATION VI. |
|
| ON OXYGEN AND NITROGEN. |
181 |
The Atmosphere composed of Oxygen and Nitrogen in the State of
Gas.—Definition of Gas.—Distinction between Gas and
Vapour.—Oxygen essential to Combustion and
Respiration.—Decomposition of the Atmosphere by
Combustion.—Nitrogen Gas obtained by this Process.—Of
Oxygenation in general.—Of the Oxydation of Metals.—Oxygen
Gas obtained from Oxyd of Manganese.—Description of a Water-Bath
for collecting and preserving Gases.—Combustion of Iron Wire in
Oxygen Gas.—Fixed and volatile Products of
Combustion.—Patent Lamps.—Decomposition of the Atmosphere by
Respiration.—Recomposition of the Atmosphere. |
|
|
CONVERSATION VII. |
|
| ON HYDROGEN. |
214 |
Of Hydrogen.—Of the Formation of Water by the Combustion of
Hydrogen.—Of the Decomposition of Water.
xiv
—Detonation of Hydrogen Gas.—Description of Lavoisier’s
Apparatus for the formation of Water.—Hydrogen Gas essential to
the Production of Flame.—Musical Tones produced by the Combustion
of Hydrogen Gas within a Glass Tube.—Combustion of Candles
explained.—Gas lights.—Detonation of Hydrogen Gas in Soap
Bubbles.—Air Balloons.—Meteorological Phenomena ascribed to
Hydrogen Gas.—Miner’s Lamp.
The final two pages of the Table of Contents for Volume I were
missing from the available text; everything after “Decomposition of
Water” was supplied from earlier and later editions, compared against
the body text. The section marked “Diamond” (Conv. IX) was called
“Diamond is
Carbon(e) in a state of perfect purity” in the 4th edn., “Diamond”
alone in later editions.
|
|
|
CONVERSATION VIII. |
|
| ON SULPHUR AND PHOSPHORUS. |
256 |
Natural History of
Sulphur.—Sublimation.—Alembic.—Combustion of Sulphur
in Atmospheric Air.—Of Acidification in
general.—Nomenclature of the Acids.—Combustion of Sulphur in
Oxygen Gas.—Sulphuric Acid.—Sulphurous
Acid.—Decomposition of Sulphur.—Sulphurated Hydrogen
Gas.—Harrogate, or Hydro-sulphurated
Waters.—Phosphorus.—History of its Discovery.—Its
Combustion in Oxygen Gas.—Phosphoric Acid.—Phosphorus
Acid.—Eudiometer.—Combination of Phosphorus with
Sulphur.—Phosphorated Hydrogen Gas.—Nomenclature of Binary
Compounds.—Phosphoret of Lime burning under Water. |
|
|
CONVERSATION IX. |
|
| ON CARBON. |
282 |
Method of obtaining pure Charcoal.—Method of making common
Charcoal.—Pure Carbon not to be obtained by
Art.—Diamond.—Properties of Carbon.—Combustion of
Carbon.—Production of Carbonic Acid Gas.—Carbon susceptible
of only one Degree of Acidification.—Gaseous Oxyd of
Carbon.—Of Seltzer Water and other Mineral
Waters.—Effervescence.—Decomposition of Water by
Carbon.—Of Fixed and Essential Oils.—Of the Combustion of
Lamps and Candles.—Vegetable Acids.—Of the Power of Carbon
to revive Metals. |
|
|
CONVERSATION X. |
|
| ON METALS. |
314 |
Natural History of Metals.—Of Roasting, Smelting,
&c.—Oxydation of metals by the Atmosphere.—Change of
Colours produced by different degrees of Oxydation.—Combustion of
Metals.—Perfect Metals burnt by Electricity only.—Some
Metals revived by Carbon and other Combustibles.—Perfect Metals
revived by Heat alone.—Of the Oxydation of certain Metals by the
Decomposition of Water. Power of Acids to promote this
Effect.—Oxydation of Metals by Acids.—Metallic Neutral
Salts.—Previous oxydation of the Metal
requisite.—Crystallisation.—Solution distinguished from
Dissolution.—Five metals susceptible of
acidification.—Meteoric Stones.—Alloys, Soldering, Plating,
&c.—Of Arsenic, and of the caustic Effects of Oxygen.—Of
Verdigris, Sympathetic Ink, &c.—Of the new Metals discovered
by Sir H. Davy. |
|
CONVERSATIONS
ON
CHEMISTRY.
CONVERSATION I.
ON THE GENERAL PRINCIPLES OF
CHEMISTRY.

MRS. B.
AS you have now acquired some
elementary notions of Natural
Philosophy, I am going to propose to you another branch of
science, to which I am particularly anxious that you should devote a
share of your attention. This is Chemistry, which is so closely connected with Natural
Philosophy, that the study of the one must be incomplete without some
knowledge of the other; for, it is obvious that we can derive but a very
imperfect idea of bodies from the study of the general laws by which
they are governed, if we remain totally ignorant of their intimate
nature.
2
CAROLINE.
To confess the truth, Mrs. B., I am not disposed to form a very
favourable idea of chemistry, nor do I expect to derive much
entertainment from it. I prefer the sciences which exhibit nature
on a grand scale, to those that are confined to the minutiæ of petty
details. Can the studies which we have lately pursued, the general
properties of matter, or the revolutions of the heavenly bodies, be
compared to the mixing up of a few insignificant drugs? I grant,
however, there may be entertaining experiments in chemistry, and should
not dislike to try some of them: the distilling, for instance, of
lavender, or rose water . . . . . .
MRS. B.
I rather imagine, my dear Caroline, that your want of taste for
chemistry proceeds from the very limited idea you entertain of its
object. You confine the chemist’s laboratory to the narrow precincts of
the apothecary’s and perfumer’s shops, whilst it is subservient to an
immense variety of other useful purposes. Besides, my dear, chemistry is
by no means confined to works of art. Nature also has her laboratory,
which is the universe, and there she is incessantly employed in chemical
operations. You are surprised, Caroline, but I assure you that the most
wonderful and the most interesting phenomena of nature are
3
almost all of them produced by chemical powers. What Bergman, in the
introduction to his history of chemistry, has said of this science, will
give you a more just and enlarged idea of it. The knowledge of nature
may be divided, he observes, into three periods. The first was that in
which the attention of men was occupied in learning the external forms
and characters of objects, and this is called Natural History. In
the second, they considered the effects of bodies acting on each other
by their mechanical power, as their weight and motion, and this
constitutes the science of Natural Philosophy. The third period
is that in which the properties and mutual action of the elementary
parts of bodies was investigated. This last is the science of Chemistry, and I have no doubt you will soon
agree with me in thinking it the most interesting.
You may easily conceive, therefore, that without entering into the
minute details of practical chemistry, a woman may obtain such a
knowledge of the science as will not only throw an interest on the
common occurrences of life, but will enlarge the sphere of her ideas,
and render the contemplation of nature a source of delightful
instruction.
CAROLINE.
If this is the case, I have certainly been much
4
mistaken in the notion I had formed of chemistry. I own that I
thought it was chiefly confined to the knowledge and preparation of
medicines.
MRS. B.
That is only a branch of chemistry which is called Pharmacy; and,
though the study of it is certainly of great importance to the world at
large, it belongs exclusively to professional men, and is therefore the
last that I should advise you to pursue.
EMILY.
But, did not the chemists formerly employ themselves in search of the
philosopher’s stone, or the secret of making gold?
MRS. B.
These were a particular set of misguided philosophers, who dignified
themselves with the name of Alchemists, to distinguish their pursuits
from those of the common chemists, whose studies were confined to the
knowledge of medicines.
But, since that period, chemistry has undergone so complete a
revolution, that, from an obscure and mysterious art, it is now become a
regular and beautiful science, to which art is entirely subservient. It
is true, however, that we are indebted to the alchemists for many very
useful discoveries, which sprung from their fruitless attempts
5
to make gold, and which, undoubtedly, have proved of infinitely greater
advantage to mankind than all their chimerical pursuits.
The modern chemists, instead of directing their ambition to the vain
attempt of producing any of the original substances in nature, rather
aim at analysing and imitating her operations, and have sometimes
succeeded in forming combinations, or effecting decompositions, no
instances of which occur in the chemistry of Nature. They have little
reason to regret their inability to make gold, whilst, by their
innumerable inventions and discoveries, they have so greatly stimulated
industry and facilitated labour, as prodigiously to increase the
luxuries as well as the necessaries of life.
EMILY.
But, I do not understand by what means chemistry can facilitate
labour; is not that rather the province of the mechanic?
MRS. B.
There are many ways by which labour may be rendered more easy,
independently of mechanics; but even the machine, the most wonderful in
its effects, the Steam-engine, cannot be understood without the
assistance of chemistry. In agriculture, a chemical knowledge of
the nature of soils, and of vegetation, is highly useful; and, in those
6
arts which relate to the comforts and conveniences of life, it would be
endless to enumerate the advantages which result from the study of this
science.
CAROLINE.
But, pray, tell us more precisely in what manner the discoveries of
chemists have proved so beneficial to society?
MRS. B.
That would be an injudicious anticipation; for you would not
comprehend the nature of such discoveries and useful applications, as
well as you will do hereafter. Without a due regard to method, we cannot
expect to make any progress in chemistry. I wish to direct your
observations chiefly to the chemical operations of Nature; but those of
Art are certainly of too high importance to pass unnoticed. We shall
therefore allow them also some share of our attention.
EMILY.
Well, then, let us now set to work regularly. I am very anxious
to begin.
MRS. B.
The object of chemistry is to obtain a knowledge of the intimate
nature of bodies, and of their mutual action on each other. You find
therefore,
7
Caroline, that this is no narrow or confined science, which comprehends
every thing material within our sphere.
CAROLINE.
On the contrary, it must be inexhaustible; and I am a loss to
conceive how any proficiency can be made in a science whose objects are
so numerous.
MRS. B.
If every individual substance were formed of different materials, the
study of chemistry would, indeed, be endless; but you must observe that
the various bodies in nature are composed of certain elementary
principles, which are not very numerous.
CAROLINE.
Yes; I know that all bodies are composed of fire, air, earth, and
water; I learnt that many years ago.
MRS. B.
But you must now endeavour to forget it. I have already informed
you what a great change chemistry has undergone since it has become a
regular science. Within these thirty years especially, it has
experienced an entire revolution, and it is now proved, that neither
fire, air, earth, nor water, can be called elementary bodies. For an
8
elementary body is one that has never been decomposed, that is to say,
separated into other substances; and fire, air, earth, and water, are
all of them susceptible of decomposition.
EMILY.
I thought that decomposing a body was dividing it into its minutest
parts. And if so, I do not understand why an elementary substance
is not capable of being decomposed, as well as any other.
MRS. B.
You have misconceived the idea of decomposition; it is very
different from mere division. The latter simply reduces a body
into parts, but the former separates it into the various ingredients, or
materials, of which it is composed. If we were to take a loaf of bread,
and separate the several ingredients of which it is made, the flour, the
yeast, the salt, and the water, it would be very different from cutting
or crumbling the loaf into pieces.
EMILY.
I understand you now very well. To decompose a body is to separate
from each other the various elementary substances of which it
consists.
CAROLINE.
But flour, water, and other materials of bread,
9
according to our definition, are not elementary substances?
MRS. B.
No, my dear; I mentioned bread rather as a familiar comparison, to
illustrate the idea, than as an example.
The elementary substances of which a body is composed are called the
constituent parts of that body; in decomposing it, therefore, we
separate its constituent parts. If, on the contrary, we divide a body by
chopping it to pieces, or even by grinding or pounding it to the finest
powder, each of these small particles will still consist of a portion of
the several constituent parts of the whole body: these are called the
integrant parts; do you understand the difference?
EMILY.
Yes, I think, perfectly. We decompose a body into its
constituent parts; and divide it into its integrant
parts.
MRS. B.
Exactly so. If therefore a body consists of only one kind of
substance, though it may be divided into its integrant parts, it is not
possible to decompose it. Such bodies are therefore called simple
or elementary, as they are the elements of which all other bodies
are composed. Compound
10
bodies are such as consist of more than one of these elementary
principles.
CAROLINE.
But do not fire, air, earth, and water, consist, each of them, but of
one kind of substance?
MRS. B.
No, my dear; they are every one of them susceptible of being
separated into various simple bodies. Instead of four, chemists now
reckon upwards of forty elementary substances. The existence of most of
these is established by the clearest experiments; but, in regard to a
few of them, particularly the most subtle agents of nature, heat,
light, and electricity, there is yet much uncertainty, and
I can only give you the opinion which seems most probably deduced from
the latest discoveries. After I have given you a list of the elementary
bodies, classed according to their properties, we shall proceed to
examine each of them separately, and then consider them in their
combinations with each other.
Excepting the more general agents of nature, heat, light, and
electricity, it would seem that the simple form of bodies is that of a
metal.
CAROLINE.
You astonish me! I thought the metals were only
11
one class of minerals, and that there were besides, earths, stones,
rocks, acids, alkalies, vapours, fluids, and the whole of the animal and
vegetable kingdoms.
MRS. B.
You have made a tolerably good enumeration, though I fear not
arranged in the most scientific order. All these bodies, however, it is
now strongly believed, may be ultimately resolved into metallic
substances. Your surprise at this circumstance is not singular, as the
decomposition of some of them, which has been but lately accomplished,
has excited the wonder of the whole philosophical world.
But to return to the list of simple bodies—these being usually
found in combination with oxygen, I shall class them according to
their properties when so combined. This will, I think, facilitate
their future investigation.
EMILY.
Pray what is oxygen?
MRS. B.
A simple body; at least one that is supposed to be so, as it has
never been decomposed. It is always found united with the negative
electricity. It will be one of the first of the elementary bodies whose
properties I shall explain to you, and, as
12
you will soon perceive, it is one of the most important in nature; but
it would be irrelevant to enter upon this subject at present. We must
now confine our attention to the enumeration and classification of the
simple bodies in general. They may be arranged as follows:
CLASS I.
Comprehending the imponderable agents, viz.
HEAT or CALORIC,
LIGHT,
ELECTRICITY.
CLASS II.
Comprehending agents capable of uniting with inflammable bodies, and in
most instances of effecting their combustion.
OXYGEN,
CHLORINE,
IODINE.*
CLASS III.
Comprehending bodies capable of uniting with oxygen, and, forming with
it various compounds. This class may be divided as follows:
DIVISION 1.
13
DIVISION 2.
Bodies forming acids.
| NITROGEN, |
forming nitric acid. |
| SULPHUR, |
forming sulphuric acid. |
| PHOSPHORUS, |
forming phosphoric acid. |
| CARBON, |
forming carbonic acid. |
| BORACIUM, |
forming boracic acid. |
| FLUORIUM, |
forming fluoric acid. |
| MURIATIUM, |
forming muriatic acid. |
DIVISION 3.
Metallic bodies forming alkalies.
| POTASSIUM, |
forming potash. |
| SODIUM, |
forming soda. |
| AMMONIUM, |
forming ammonia. |
DIVISION 4.
Metallic bodies forming earths.
| CALCIUM, |
or metal forming lime. |
| MAGNIUM, |
forming magnesia. |
| BARIUM, |
forming barytes. |
| STRONTIUM, |
forming strontites. |
| SILICIUM, |
forming silex. |
| ALUMIUM, |
forming alumine. |
| YTTRIUM, |
forming yttria. |
|
14
GLUCIUM, |
forming glucina. |
| ZIRCONIUM, |
forming zirconi.* |
DIVISION 5.
Metals, either naturally metallic, or yielding their oxygen to carbon or
to heat alone.
Subdivision 1.
Malleable Metals.
GOLD,
PLATINA,
PALLADIUM,
SILVER*
MERCURY†
TIN,
COPPER,
IRON,
LEAD,
NICKEL,
ZINC.
Subdiv. 2.
Brittle Metals.
ARSENIC,
BISMUTH,
ANTIMONY,
MANGANESE,
15
TELLURIUM,
COBALT,
TUNGSTEN,
MOLYBDENUM,
TITANIUM,
CHROME,
URANIUM,
COLUMBIUM or TANTALIUM,
IRIDIUM,
OSMIUM,
RHODIUM.*
CAROLINE.
Oh, what a formidable list! You will have much to do to explain it,
Mrs. B.; for I assure you it is perfectly unintelligible to me, and
I think rather perplexes than assists me.
MRS. B.
Do not let that alarm you, my dear; I hope that hereafter this
classification will appear quite clear, and, so far from perplexing you,
will assist you in arranging your ideas. It would be in vain to attempt
forming a division that would appear perfectly clear to a beginner: for
you may easily conceive that a chemical division being necessarily
founded on properties with which you are almost wholly unacquainted, it
is impossible that you should at once be able to understand its meaning
or appreciate its utility.
16
But, before we proceed further, it will be necessary to give you some
idea of chemical attraction, a power on which the whole science
depends.
Chemical Attraction, or the Attraction of Composition,
consists in the peculiar tendency which bodies of a different nature
have to unite with each other. It is by this force that all the
compositions, and decompositions, are effected.
EMILY.
What is the difference between chemical attraction, and the
attraction of cohesion, or of aggregation, which you often mentioned to
us, in former conversations?
MRS. B.
The attraction of cohesion exists only between particles of the
same nature, whether simple or compound; thus it unites the
particles of a piece of metal which is a simple substance, and likewise
the particles of a loaf of bread which is a compound. The attraction of
composition, on the contrary, unites and maintains, in a state of
combination, particles of a dissimilar nature; it is this power
that forms each of the compound particles of which bread consists; and
it is by the attraction of cohesion that all these particles are
connected into a single mass.
17
EMILY.
The attraction of cohesion, then, is the power which unites the
integrant particles of a body: the attraction of composition that which
combines the constituent particles. Is it not so?
MRS. B.
Precisely: and observe that the attraction of cohesion unites
particles of a similar nature, without changing their original
properties; the result of such an union, therefore, is a body of the
same kind as the particles of which it is formed; whilst the attraction
of composition, by combining particles of a dissimilar nature, produces
compound bodies, quite different from any of their constituents. If, for
instance, I pour on the piece of copper, contained in this glass,
some of this liquid (which is called nitric acid), for which it has a
strong attraction, every particle of the copper will combine with a
particle of acid, and together they will form a new body, totally
different from either the copper or the acid.
Do you observe the internal commotion that already begins to take
place? It is produced by the combination of these two substances; and
yet the acid has in this case to overcome not only the resistance which
the strong cohesion of the particles of copper opposes to their
combination with it, but also to overcome the weight of the copper,
which
18
makes it sink to the bottom of the glass, and prevents the acid from
having such free access to it as it would if the metal were suspended in
the liquid.
EMILY.
The acid seems, however, to overcome both these obstacles without
difficulty, and appears to be very rapidly dissolving the copper.
MRS. B.
By this means it reduces the copper into more minute parts than could
possibly be done by any mechanical power. But as the acid can act only
on the surface of the metal, it will be some time before the union of
these two bodies will be completed.
You may, however, already see how totally different this compound is
from either of its ingredients. It is neither colourless, like the acid,
nor hard, heavy, and yellow like the copper. If you tasted it, you would
no longer perceive the sourness of the acid. It has at present the
appearance of a blue liquid; but when the union is completed, and the
water with which the acid is diluted is evaporated, the compound will
assume the form of regular crystals, of a fine blue colour, and
perfectly transparent*. Of these I can shew you a
19
specimen, as I have prepared some for that purpose.
CAROLINE.
How very beautiful they are, in colour, form, and transparency!
EMILY.
Nothing can be more striking than this example of chemical
attraction.
MRS. B.
The term attraction has been lately introduced into chemistry
as a substitute for the word affinity, to which some chemists
have objected, because it originated in the vague notion that chemical
combinations depended upon a certain resemblance, or relationship,
between particles that are disposed to unite; and this idea is not only
imperfect, but erroneous, as it is generally particles of the most
dissimilar nature, that have the greatest tendency to combine.
CAROLINE.
Besides, there seems to be no advantage in using a variety of terms
to express the same meaning; on the contrary it creates confusion; and
as we are well acquainted with the term Attraction in natural
philosophy, we had better adopt it in chemistry likewise.
20
MRS. B.
If you have a clear idea of the meaning, I shall leave you at
liberty to express it in the terms you prefer. For myself,
I confess that I think the word Attraction best suited to the
general law that unites the integrant particles of bodies; and Affinity
better adapted to that which combines the constituent particles, as it
may convey an idea of the preference which some bodies have for others,
which the term attraction of composition does not so well
express.
EMILY.
So I think; for though that preference may not result from any
relationship, or similitude, between the particles (as you say was once
supposed), yet, as it really exists, it ought to be expressed.
MRS. B.
Well, let it be agreed that you may use the terms affinity,
chemical attraction and attraction of composition,
indifferently, provided you recollect that they have all the same
meaning.
EMILY.
I do not conceive how bodies can be decomposed by chemical
attraction. That this power should be the means of composing them, is
very obvious; but that it should, at the same time, produce exactly the
contrary effect, appears to me very singular.
21
MRS. B.
To decompose a body is, you know, to separate its constituent parts,
which, as we have just observed, cannot be done by mechanical means.
EMILY.
No: because mechanical means separate only the integrant particles;
they act merely against the attraction of cohesion, and only divide a
compound into smaller parts.
MRS. B.
The decomposition of a body is performed by chemical powers. If you
present to a body composed of two principles, a third, which has a
greater affinity for one of them than the two first have for each other,
it will be decomposed, that is, its two principles will be separated by
means of the third body. Let us call two ingredients, of which the body
is composed, A and B. If we present to it another
ingredient C, which has a greater affinity for B than that which
unites A and B, it necessarily follows that B will quit A to
combine with C. The new ingredient, therefore, has effected a
decomposition of the original body A B; A has been left alone,
and a new compound, B C, has been formed.
EMILY.
We might, I think, use the comparison of two
22
friends, who were very happy in each other’s society, till a third
disunited them by the preference which one of them gave to the
new-comer.
MRS. B.
Very well. I shall now show you how this takes place in
chemistry.
Let us suppose that we wish to decompose the compound we have just
formed by the combination of the two ingredients, copper and nitric
acid; we may do this by presenting to it a piece of iron, for which the
acid has a stronger attraction than for copper; the acid will,
consequently, quit the copper to combine with the iron, and the copper
will be what the chemists call precipitated, that is to say, it
will be thrown down in its separate state, and reappear in its simple
form.
In order to produce this effect, I shall dip the blade of this knife
into the fluid, and, when I take it out, you will observe, that, instead
of being wetted with a bluish liquid, like that contained in the glass,
it will be covered with a thin coat of copper.
CAROLINE.
So it is really! but then is it not the copper, instead of the acid,
that has combined with the iron blade?
MRS. B.
No; you are deceived by appearances: it is
23
the acid which combines with the iron, and, in so doing, deposits or
precipitates the copper on the surface of the blade.
EMILY.
But, cannot three or more substances combine together, without any of
them being precipitated?
MRS. B.
That is sometimes the case; but, in general, the stronger affinity
destroys the weaker; and it seldom happens that the attraction of
several substances for each other is so equally balanced as to produce
such complicated compounds.
CAROLINE.
But, pray, Mrs. B., what is the cause of the chemical attraction of
bodies for each other? It appears to me more extraordinary or unnatural,
if I may use the expression, than the attraction of cohesion, which
unites particles of a similar nature.
MRS. B.
Chemical attraction may, like that of cohesion or gravitation, be one
of the powers inherent in matter which, in our present state of
knowledge, admits of no other satisfactory explanation than an immediate
reference to a divine cause. Sir H. Davy, however, whose important
discoveries have
24
opened such improved views in chemistry, has suggested an hypothesis
which may throw great light upon that science. He supposes that there
are two kinds of electricity, with one or other of which all bodies are
united. These we distinguish by the names of positive and
negative electricity; those bodies are disposed to combine, which
possess opposite electricities, as they are brought together by the
attraction which these electricities have for each other. But, whether
this hypothesis be altogether founded on truth or not, it is impossible
to question the great influence of electricity in chemical
combinations.
EMILY.
So, that we must suppose that the two electricities always attract
each other, and thus compel the bodies in which they exist to
combine?
CAROLINE.
And may not this be also the cause of the attraction of cohesion?
MRS. B.
No, for in particles of the same nature the same electricities must
prevail, and it is only the different or opposite electric fluids that
attract each other.
CAROLINE.
These electricities seem to me to be a kind of
25
chemical spirit, which animates the particles of bodies, and draws them
together.
EMILY.
If it is known, then, with which of the electricities bodies are
united, it can be inferred which will, and which will not, combine
together?
MRS. B.
Certainly.—I should not omit to mention, that some doubts have
been entertained whether electricity be really a material agent, or
whether it might not be a power inherent in bodies, similar to, or,
perhaps identical with, attraction.
EMILY.
But what then would be the electric spark which is visible, and must therefore be
really material?
MRS. B.
What we call the electric spark, may, Sir H. Davy says, be merely the
heat and light, or fire produced by the chemical combinations with which
these phenomena are always connected. We will not, however, enter more
fully on this important subject at present, but reserve the principal
facts which relate to it to a future conversation.
Before we part, however, I must recommend you to fix in your memory
the names of the simple bodies, against our next interview.
26

CAROLINE.
We have learned by heart the names of all
the simple bodies which you have enumerated, and we are now ready to
enter on the examination of each of them successively. You will begin,
I suppose, with LIGHT?
MRS. B.
Respecting the nature of light we have little more than conjectures.
It is considered by most philosophers as a real substance, immediately
emanating from the sun, and from all luminous bodies, from which it is
projected in right lines with prodigious velocity. Light, however, being
imponderable, it cannot be confined and examined by itself; and
therefore it is to the effects it produces on other bodies, rather than
to its immediate nature, that we must direct our attention.
The connection between light and heat is very obvious; indeed, it is
such, that it is extremely
27
difficult to examine the one independently of the other.
EMILY.
But, is it possible to separate light from heat; I thought they
were only different degrees of the same thing, fire?
MRS. B.
I told you that fire was not now considered as a simple element.
Whether light and heat be altogether different agents, or not,
I cannot pretend to decide; but, in many cases, light may be
separated from heat. The first discovery of this was made by a
celebrated Swedish chemist, Scheele. Another very striking illustration
of the separation of heat and light was long after pointed out by Dr.
Herschell. This philosopher discovered that these two agents were
emitted in the rays of the sun, and that heat was less refrangible than
light; for, in separating the different coloured rays of light by a
prism (as we did some time ago), he found that the greatest heat was
beyond the spectrum, at a little distance from the red rays, which, you
may recollect, are the least refrangible.
EMILY.
I should like to try that experiment.
28
MRS. B.
It is by no means an easy one: the heat of a ray of light, refracted
by a prism, is so small, that it requires a very delicate thermometer to
distinguish the difference of the degree of heat within and without the
spectrum. For in this experiment the heat is not totally separated from
the light, each coloured ray retaining a certain portion of it, though
the greatest part is not sufficiently refracted to fall within the
spectrum.
EMILY.
I suppose, then, that those coloured rays which are the least
refrangible, retain the greatest quantity of heat?
MRS. B.
They do so.
EMILY.
Though I no longer doubt that light and heat can be separated, Dr.
Herschell’s experiment does not appear to me to afford sufficient proof
that they are essentially different; for light, which you call a simple
body, may likewise be divided into the various coloured rays.
MRS. B.
No doubt there must be some difference in the various coloured rays.
Even their chemical powers
29
are different. The blue rays, for instance, have the greatest effect in
separating oxygen from bodies, as was found by Scheele; and there exist
also, as Dr. Wollaston has shown, rays more refrangible than the blue,
which produce the same chemical effect, and, what is very remarkable,
are invisible.
EMILY.
Do you think it possible that heat may be merely a modification of
light?
MRS. B.
That is a supposition which, in the present state of natural
philosophy, can neither be positively affirmed nor denied. Let us,
therefore, instead of discussing theoretical points, be contented with
examining what is known respecting the chemical effects of light.
Light is capable of entering into a kind of transitory union with
certain substances, and this is what has been called phosphorescence.
Bodies that are possessed of this property, after being exposed to the
sun’s rays, appear luminous in the dark. The shells of fish, the bones
of land animals, marble, limestone, and a variety of combinations of
earths, are more or less powerfully phosphorescent.
30
CAROLINE.
I remember being much surprised last summer with the phosphorescent
appearance of some pieces of rotten wood, which had just been dug out of
the ground; they shone so bright that I at first supposed them to be
glow-worms.
EMILY.
And is not the light of a glow-worm of a phosphorescent nature?
MRS. B.
It is a very remarkable instance of phosphorescence in living
animals; this property, however, is not exclusively possessed by the
glow-worm. The insect called the lanthorn-fly, which is peculiar to warm
climates, emits light as it flies, producing in the dark a remarkably
sparkling appearance. But it is more common to see animal matter in a
dead state possessed of a phosphorescent quality; sea fish is often
eminently so.
EMILY.
I have heard that the sea has sometimes had the appearance of being
illuminated, and that the light is supposed to proceed from the spawn of
fishes floating on its surface.
31
MRS. B.
This light is probably owing to that or some other animal matter. Sea
water has been observed to become luminous from the substance of a fresh
herring having been immersed in it; and certain insects, of the Medusa
kind, are known to produce similar effects.
But the strongest phosphorescence is produced by chemical
compositions prepared for the purpose, the most common of which consists
of oyster shells and sulphur, and is known by the name of Canton’s
Phosphorus.
EMILY.
I am rather surprised, Mrs. B., that you should have said so much of
the light emitted by phosphorescent bodies without taking any notice of
that which is produced by burning bodies.
MRS. B.
The light emitted by the latter is so intimately connected with the
chemical history of combustion, that I must defer all explanation of it
till we come to the examination of that process, which is one of the
most interesting in chemical science.
Light is an agent capable of producing various chemical changes. It
is essential to the welfare both of the animal and vegetable kingdoms;
for men and plants grow pale and sickly if deprived of
32
its salutary influence. It is likewise remarkable for its property of
destroying colour, which renders it of great consequence in the process
of bleaching.
EMILY.
Is it not singular that light, which in studying optics we were
taught to consider as the source and origin of colours, should have also
the power of destroying them?
CAROLINE.
It is a fact, however, that we every day experience; you know how it
fades the colours of linens and silks.
EMILY.
Certainly. And I recollect that endive is made to grow white instead
of green, by being covered up so as to exclude the light. But by what
means does light produce these effects?
MRS. B.
This I cannot attempt to explain to you until you have obtained a
further knowledge of chemistry. As the chemical properties of light can
be accounted for only in their reference to compound bodies, it would be
useless to detain you any longer on this subject; we may therefore pass
on to the examination of heat, or caloric, with which we are somewhat
better acquainted.
33
Heat and Light may be always distinguished by the different
sensations they produce, Light affects the sense of sight;
Caloric that of feeling; the one produces Vision, the
other the sensation of Heat.
Caloric is found to exist in a variety of forms or modifications, and
I think it will be best to consider it under the two following heads,
viz.
1. FREE OR RADIANT CALORIC.
2. COMBINED CALORIC.
The first, FREE or RADIANT CALORIC, is also called HEAT OF TEMPERATURE; it comprehends all heat which
is perceptible to the senses, and affects the thermometer.
EMILY.
You mean such as the heat of the sun, of fire, of candles, of stoves;
in short, of every thing that burns?
MRS. B.
And likewise of things that do not burn, as, for instance, the warmth
of the body; in a word, all heat that is sensible, whatever may
be its degree, or the source from which it is derived.
CAROLINE.
What then are the other modifications of caloric?
34
It must be a strange kind of heat that cannot be perceived by our
senses.
MRS. B.
None of the modifications of caloric should properly be called
heat; for heat, strictly speaking, is the sensation produced by
caloric, on animated bodies; this word, therefore, in the accurate
language of science, should be confined to express the sensation. But
custom has adapted it likewise to inanimate matter, and we say the
heat of an oven, the heat of the sun, without any reference
to the sensation which they are capable of exciting.
It was in order to avoid the confusion which arose from thus
confounding the cause and effect, that modern chemists adopted the new
word caloric, to denote the principle which produces heat; yet
they do not always, in compliance with their own language, limit the
word heat to the expression of the sensation, since they still
frequently employ it in reference to the other modifications of caloric
which are quite independent of sensation.
CAROLINE.
But you have not yet explained to us what these other modifications
of caloric are.
MRS. B.
Because you are not acquainted with the properties
35
of free caloric, and you know that we have agreed to proceed with
regularity.
One of the most remarkable properties of free caloric is its power of
dilating bodies. This fluid is so extremely subtle, that it
enters and pervades all bodies whatever, forces itself between their
particles, and not only separates them, but frequently drives them
asunder to a considerable distance from each other. It is thus that
caloric dilates or expands a body so as to make it occupy a greater
space than it did before.
EMILY.
The effect it has on bodies, therefore, is directly contrary to that
of the attraction of cohesion; the one draws the particles together, the
other drives them asunder.
MRS. B.
Precisely. There is a continual struggle between the attraction of
aggregation, and the expansive power of caloric; and from the action of
these two opposite forces, result all the various forms of matter, or
degrees of consistence, from the solid, to the liquid and aëriform
state. And accordingly we find that most bodies are capable of passing
from one of these forms to the other, merely in consequence of their
receiving different quantities of caloric.
36
CAROLINE.
That is very curious; but I think I understand the reason of it. If a
great quantity of caloric is added to a solid body, it introduces itself
between the particles in such a manner as to overcome, in a considerable
degree, the attraction of cohesion; and the body, from a solid, is then
converted into a fluid.
MRS. B.
This is the case whenever a body is fused or melted; but if you add
caloric to a liquid, can you tell me what is the consequence?
CAROLINE.
The caloric forces itself in greater abundance between the particles
of the fluid, and drives them to such a distance from each other, that
their attraction of aggregation is wholly destroyed: the liquid is then
transformed into vapour.
MRS. B.
Very well; and this is precisely the case with boiling water, when it
is converted into steam or vapour, and with all bodies that assume an
aëriform state.
EMILY.
I do not well understand the word aëriform?
37
MRS. B.
Any elastic fluid whatever, whether it be merely vapour or permanent
air, is called aëriform.
But each of these various states, solid, liquid, and aëriform, admit
of many different degrees of density, or consistence, still arising
(chiefly at least) from the different quantities of caloric the bodies
contain. Solids are of various degrees of density, from that of gold, to
that of a thin jelly. Liquids, from the consistence of melted glue, or
melted metals, to that of ether, which is the lightest of all liquids.
The different elastic fluids (with which you are not yet acquainted) are
susceptible of no less variety in their degrees of density.
EMILY.
But does not every individual body also admit of different degrees of
consistence, without changing its state?
MRS. B.
Undoubtedly; and this I can immediately show you by a very simple
experiment. This piece of iron now exactly fits the frame, or ring, made
to receive it; but if heated red hot, it will no longer do so, for its
dimensions will be so much increased by the caloric that has penetrated
into it, that it will be much too large for the frame.
The iron is now red hot; by applying it to the frame, we shall see
how much it is dilated.
38
EMILY.
Considerably so indeed! I knew that heat had this effect on bodies,
but I did not imagine that it could be made so conspicuous.
MRS. B.
By means of this instrument (called a Pyrometer) we may estimate, in
the most exact manner, the various dilatations of any solid body by
heat. The body we are now going to submit to trial is this small iron
bar; I fix it to this apparatus, (Plate I. Fig. 1.) and then heat it by lighting the three lamps
beneath it: when the bar expands, it increases in length as well as
thickness; and, as one end communicates with this wheel-work, whilst the
other end is fixed and immoveable, no sooner does it begin to dilate
than it presses against the wheel-work, and sets in motion the index,
which points out the degrees of dilatation on the dial-plate.
Plate I.
Vol. I. p. 38.
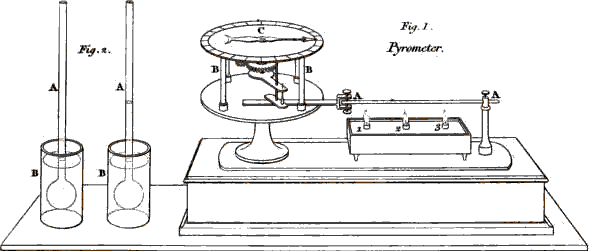
Fig. 1
A.A Bar of Metal.
1.2.3 Lamps burning.
B.B Wheel work.
C Index.
Fig. 2
A.A Glass tubes with bulbs.
B.B Glasses of water in which they are immersed.
Larger
view
EMILY.
This is, indeed, a very curious instrument; but I do not understand
the use of the wheels: would it not be more simple, and answer the
purpose equally well, if the bar, in dilating, pressed against the
index, and put it in motion without the intervention of the wheels?
39
MRS. B.
The use of the wheels is merely to multiply the motion, and therefore
render the effect of the caloric more obvious; for if the index moved no
more than the bar increased in length, its motion would scarcely be
perceptible; but by means of the wheels it moves in a much greater
proportion, which therefore renders the variations far more
conspicuous.
By submitting different bodies to the test of the pyrometer, it is
found that they are far from dilating in the same proportion. Different
metals expand in different degrees, and other kinds of solid bodies vary
still more in this respect. But this different susceptibility of
dilatation is still more remarkable in fluids than in solid bodies, as I
shall show you. I have here two glass tubes, terminated at one end
by large bulbs. We shall fill the bulbs, the one with spirit of wine,
the other with water. I have coloured both liquids, in order that
the effect may be more conspicuous. The spirit of wine, you see, dilates
by the warmth of my hand as I hold the bulb.
EMILY.
It certainly does, for I see it is rising into the tube. But water,
it seems, is not so easily affected by heat; for scarcely any change is
produced on it by the warmth of the hand.
40
MRS. B.
True; we shall now plunge the bulbs into hot water, (Plate I. Fig. 2.) and you will see
both liquids rise in the tubes; but the spirit of wine will ascend
highest.
CAROLINE.
How rapidly it expands! Now it has nearly reached the top of the
tube, though the water has hardly begun to rise.
EMILY.
The water now begins to dilate. Are not these glass tubes, with
liquids rising within them, very like thermometers?
MRS. B.
A thermometer is constructed exactly on the same principle, and these
tubes require only a scale to answer the purpose of thermometers: but
they would be rather awkward in their dimensions. The tubes and bulbs of
thermometers, though of various sizes, are in general much smaller than
these; the tube too is hermetically closed, and the air excluded from
it. The fluid most generally used in thermometers is mercury, commonly
called quicksilver, the dilatations and contractions of which correspond
more exactly to the additions, and subtractions, of caloric, than those
of any other fluid.
41
CAROLINE.
Yet I have often seen coloured spirit of wine used in
thermometers.
MRS. B.
The expansions and contractions of that liquid are not quite so
uniform as those of mercury; but in cases in which it is not requisite
to ascertain the temperature with great precision, spirit of wine will
answer the purpose equally well, and indeed in some respects better, as
the expansion of the latter is greater, and therefore more conspicuous.
This fluid is used likewise in situations and experiments in which
mercury would be frozen; for mercury becomes a solid body, like a piece
of lead or any other metal, at a certain degree of cold: but no degree
of cold has ever been known to freeze spirit of wine.
A thermometer, therefore, consists of a tube with a bulb, such as you
see here, containing a fluid whose degrees of dilatation and contraction
are indicated by a scale to which the tube is fixed. The degree which
indicates the boiling point, simply means that, when the fluid is
sufficiently dilated to rise to this point, the heat is such that water
exposed to the same temperature will boil. When, on the other hand, the
fluid is so much condensed as to sink to the freezing point, we know
that water will freeze at that temperature.
42
The extreme points of the scales are not the same in all thermometers,
nor are the degrees always divided in the same manner. In different
countries philosophers have chosen to adopt different scales and
divisions. The two thermometers most used are those of Fahrenheit, and
of Reaumur; the first is generally preferred by the English, the latter
by the French.
EMILY.
The variety of scale must be very inconvenient, and I should think
liable to occasion confusion, when French and English experiments are
compared.
MRS. B.
The inconvenience is but very trifling, because the different
gradations of the scales do not affect the principle upon which
thermometers are constructed. When we know, for instance, that
Fahrenheit’s scale is divided into 212 degrees, in which 32° corresponds
with the freezing point, and 212° with the point of boiling water: and
that Reaumur’s is divided only into 80 degrees, in which 0° denotes the
freezing point, and 80° that of boiling water, it is easy to compare the
two scales together, and reduce the one into the other. But, for greater
convenience, thermometers are sometimes constructed with both these
scales, one
43
on either side of the tube; so that the correspondence of the different
degrees of the two scales is thus instantly seen. Here is one of these
scales, (Plate II.
Fig. 1.) by which you can at once perceive that each degree of
Reaumur’s corresponds to 2¼ of Fahrenheit’s division. But I believe the
French have, of late, given the preference to what they call the
centigrade scale, in which the space between the freezing and the
boiling point is divided into 100 degrees.
Plate II.
Vol. I. p. 42.
Larger
view
CAROLINE.
That seems to me the most reasonable division, and I cannot guess why
the freezing point is called 32°, or what advantage is derived
from it.
MRS. B.
There really is no advantage in it; and it originated in a mistaken
opinion of the instrument-maker, Fahrenheit, who first constructed these
thermometers. He mixed snow and salt together, and produced by that
means a degree of cold which he concluded was the greatest possible, and
therefore made his scale begin from that point. Between that and boiling
water he made 212 degrees, and the freezing point was found to be at
32°.
44
EMILY.
Are spirit of wine, and mercury, the only liquids used in the
construction of thermometers?
MRS. B.
I believe they are the only liquids now in use, though some others,
such as linseed oil, would make tolerable thermometers: but for
experiments in which a very quick and delicate test of the changes of
temperature is required, air is the fluid sometimes employed. The bulb
of air thermometers is filled with common air only, and its expansion
and contraction are indicated by a small drop of any coloured liquor,
which is suspended within the tube, and moves up and down, according as
the air within the bulb and tube expands or contracts. But in general,
air thermometers, however sensible to changes of temperature, are by no
means accurate in their indications.
I can, however, show you an air thermometer of a very peculiar
construction, which is remarkably well adapted for some chemical
experiments, as it is equally delicate and accurate in its
indications.
CAROLINE.
It looks like a double thermometer reversed, the tube being bent, and
having a large bulb at each of its extremities. (Plate II. Fig. 2.)
45
EMILY.
Why do you call it an air thermometer; the tube contains a coloured
liquid?
MRS. B.
But observe that the bulbs are filled with air, the liquid being
confined to a portion of the tube, and answering only the purpose of
showing, by its motion in the tube, the comparative dilatation or
contraction of the air within the bulbs, which afford an indication of
their relative temperature. Thus if you heat the bulb A, by the
warmth of your hand, the fluid will rise towards the bulb B, and
the contrary will happen if you reverse the experiment.
But if, on the contrary, both tubes are of the same temperature, as
is the case now, the coloured liquid, suffering an equal pressure on
each side, no change of level takes place.
CAROLINE.
This instrument appears, indeed, uncommonly delicate. The fluid is
set in motion by the mere approach of my hand.
MRS. B.
You must observe, however, that this thermometer cannot indicate the
temperature of any particular body, or of the medium in which it is
46
immersed; it serves only to point out the difference of
temperature between the two bulbs, when placed under different
circumstances. For this reason it has been called differential thermometer.
You will see by-and-bye to what particular purposes this instrument
applies.
EMILY.
But do common thermometers indicate the exact quantity of caloric
contained either in the atmosphere, or in any body with which they are
in contact?
MRS. B.
No: first, because there are other modifications of caloric which do
not affect the thermometer; and, secondly, because the temperature of a
body, as indicated by the thermometer, is only relative. When, for
instance, the thermometer remains stationary at the freezing point, we
know that the atmosphere (or medium in which it is placed, whatever it
may be) is as cold as freezing water; and when it stands at the boiling
point, we know that this medium is as hot as boiling water; but we do
not know the positive quantity of heat contained either in freezing or
boiling water, any more than we know the real extremes of heat and cold;
and consequently we cannot determine that of the body in which the
thermometer is placed.
47
CAROLINE.
I do not quite understand this explanation.
MRS. B.
Let us compare a thermometer to a well, in which the water rises to
different heights, according as it is more or less supplied by the
spring which feeds it: if the depth of the well is unfathomable, it must
be impossible to know the absolute quantity of water it contains; yet we
can with the greatest accuracy measure the number of feet the water has
risen or fallen in the well at any time, and consequently know the
precise quantity of its increase or diminution, without having the least
knowledge of the whole quantity of water it contains.
CAROLINE.
Now I comprehend it very well; nothing appears to me to explain a
thing so clearly as a comparison.
EMILY.
But will thermometers bear any degree of heat?
MRS. B.
No; for if the temperature were much above the highest degree marked
on the scale of the thermometer, the mercury would burst the tube in an
attempt to ascend. And at any rate, no thermometer can be applied to
temperatures higher than the boiling
48
point of the liquid used in its construction, for the steam, on the
liquid beginning to boil, would burst the tube. In furnaces, or whenever
any very high temperature is to be measured, a pyrometer, invented
by Wedgwood, is used for that purpose. It is made of a certain
composition of baked clay, which has the peculiar property of
contracting by heat, so that the degree of contraction of this substance
indicates the temperature to which it has been exposed.
EMILY.
But is it possible for a body to contract by heat? I thought
that heat dilated all bodies whatever.
MRS. B.
This is not an exception to the rule. You must recollect that the
bulk of the clay is not compared, whilst hot, with that which it has
when cold; but it is from the change which the clay has undergone by
having been heated that the indications of this instrument are
derived. This change consists in a beginning fusion which tends to unite
the particles of clay more closely, thus rendering it less pervious or
spongy.
Clay is to be considered as a spongy body, having many interstices or
pores, from its having contained water when soft. These interstices are
49
by heat lessened, and would by extreme heat be entirely obliterated.
CAROLINE.
And how do you ascertain the degrees of contraction of Wedgwood’s
pyrometer?
MRS. B.
The dimensions of a piece of clay are measured by a scale graduated
on the side of a tapered groove, formed in a brass ruler; the more the
clay is contracted by the heat, the further it will descend into the
narrow part of the tube.
Before we quit the subject of expansion, I must observe to you
that, as liquids expand more readily than solids, so elastic fluids,
whether air or vapour, are the most expansible of all bodies.
It may appear extraordinary that all elastic fluids whatever, undergo
the same degree of expansion from equal augmentations of
temperature.
EMILY.
I suppose, then, that all elastic fluids are of the same density?
MRS. B.
Very far from it; they vary in density, more than either liquids or
solids. The uniformity of their expansibility, which at first may appear
singular, is, however, readily accounted for. For if the different
susceptibilities of expansion of bodies
50
arise from their various degrees of attraction of cohesion, no such
difference can be expected in elastic fluids, since in these the
attraction of cohesion does not exist, their particles being on the
contrary possessed of an elastic or repulsive power; they will therefore
all be equally expanded by equal degrees of caloric.
EMILY.
True; as there is no power opposed to the expansive force of caloric
in elastic bodies, its effect must be the same in all of them.
MRS. B.
Let us now proceed to examine the other properties of free
caloric.
Free caloric always tends to diffuse itself equally, that is to say,
when two bodies are of different temperatures, the warmer gradually
parts with its heat to the colder, till they are both brought to the
same temperature. Thus, when a thermometer is applied to a hot body, it
receives caloric; when to a cold one, it communicates part of its own
caloric, and this communication continues until the thermometer and the
body arrive at the same temperature.
EMILY.
Cold, then, is nothing but a negative quality, simply implying the
absence of heat.
51
MRS. B.
Not the total absence, but a diminution of heat; for we know of no
body in which some caloric may not be discovered.
CAROLINE.
But when I lay my hand on this marble table I feel it
positively cold, and cannot conceive that there is any caloric
in it.
MRS. B.
The cold you experience consists in the loss of caloric that your
hand sustains in an attempt to bring its temperature to an equilibrium
with the marble. If you lay a piece of ice upon it, you will find that
the contrary effect will take place; the ice will be melted by the heat
which it abstracts from the marble.
CAROLINE.
Is it not in this case the air of the room, which being warmer than
the marble, melts the ice?
MRS. B.
The air certainly acts on the surface which is exposed to it, but the
table melts that part with which it is in contact.
CAROLINE.
But why does caloric tend to an equilibrium?
52
It cannot be on the same principle as other fluids, since it has no
weight?
MRS. B.
Very true, Caroline, that is an excellent objection. You might also,
with some propriety, object to the term equilibrium being applied
to a body that is without weight; but I know of no expression that would
explain my meaning so well. You must consider it, however, in a
figurative rather than a literal sense; its strict meaning is an
equal diffusion. We cannot, indeed, well say by what power it
diffuses itself equally, though it is not surprising that it should go
from the parts which have the most to those which have the least. This
subject is best explained by a theory suggested by Professor Prevost of
Geneva, which is now, I believe, generally adopted.
According to this theory, caloric is composed of particles perfectly
separate from each other, every one of which moves with a rapid velocity
in a certain direction. These directions vary as much as imagination can
conceive, the result of which is, that there are rays or lines of these
particles moving with immense velocity in every possible direction.
Caloric is thus universally diffused, so that when any portion of space
happens to be in the neighbourhood of another, which contains more
caloric, the colder portion receives a
53
quantity of calorific rays from the latter, sufficient to restore an
equilibrium of temperature. This radiation does not only take place in
free space, but extends also to bodies of every kind. Thus you may
suppose all bodies whatever constantly radiating caloric: those that are
of the same temperature give out and absorb equal quantities, so that no
variation of temperature is produced in them; but when one body contains
more free caloric than another, the exchange is always in favour of the
colder body, until an equilibrium is effected; this you found to be the
case when the marble table cooled your hand, and again when it melted
the ice.
CAROLINE.
This reciprocal radiation surprises me extremely; I thought,
from what you first said, that the hotter bodies alone emitted rays of
caloric which were absorbed by the colder; for it seems unnatural that a
hot body should receive any caloric from a cold one, even though it
should return a greater quantity.
MRS. B.
It may at first appear so, but it is no more extraordinary than that
a candle should send forth rays of light to the sun, which, you know,
must necessarily happen.
54
CAROLINE.
Well, Mrs. B—, I believe that I must give up the point. But I
wish I could see these rays of caloric; I should then have
greater faith in them.
MRS. B.
Will you give no credit to any sense but that of sight? You may feel
the rays of caloric which you receive from any body of a temperature
higher than your own; the loss of the caloric you part with in return,
it is true, is not perceptible; for as you gain more than you lose,
instead of suffering a diminution, you are really making an acquisition
of caloric. It is, therefore, only when you are parting with it to a
body of a lower temperature, that you are sensible of the sensation of
cold, because you then sustain an absolute loss of caloric.
EMILY.
And in this case we cannot be sensible of the small quantity of heat
we receive in exchange from the colder body, because it serves only to
diminish the loss.
MRS. B.
Very well, indeed, Emily. Professor Pictet, of Geneva, has made some
very interesting experiments, which prove not only that caloric radiates
from all bodies whatever, but that these rays may be reflected,
according to the laws of optics, in
55
the same manner as light. I shall repeat these experiments before
you, having procured mirrors fit for the purpose; and it will afford us
an opportunity of using the differential thermometer, which is
particularly well adapted for these experiments.—I place an
iron bullet, (Plate III.
Fig. 1.) about
two inches in diameter, and heated to a degree not sufficient to render
it luminous, in the focus of this large metallic concave mirror. The
rays of heat which fall on this mirror are reflected, agreeably to the
property of concave mirrors, in a parallel direction, so as to fall on a
similar mirror, which, you see, is placed opposite to the first, at the
distance of about ten feet; thence the rays converge to the focus of the
second mirror, in which I place one of the bulbs of this thermometer.
Now, observe in what manner it is affected by the caloric which is
reflected on it from the heated bullet.—The air is dilated in the
bulb which we placed in the focus of the mirror, and the liquor rises
considerably in the opposite leg.
Plate III.
Vol. I. p. 54

A.A. & B.B Concave mirrors fixed on stands.
C Heated Bullet placed in the focus of the mirror A.
D Thermometer, with its bulb placed in the focus of the
mirror B.
1.2.3.4 Rays of Caloric radiating from the bullet & falling on the
mirror A.
5.6.7.8 The same rays reflected from the mirror A to the mirror B.
9.10.11.12 The same rays reflected by the mirror B to the
Thermometer.
Larger
view
EMILY.
But would not the same effect take place, if the rays of caloric from
the heated bullet fell directly on the thermometer, without the
assistance of the mirrors?
MRS. B.
The effect would in that case be so trifling, at
56
the distance at which the bullet and the thermometer are from each
other, that it would be almost imperceptible. The mirrors, you know,
greatly increase the effect, by collecting a large quantity of rays into
a focus; place your hand in the focus of the mirror, and you will find
it much hotter there than when you remove it nearer to the bullet.
EMILY.
That is very true; it appears extremely singular to feel the heat
diminish in approaching the body from which it proceeds.
CAROLINE.
And the mirror which produces so much heat, by converging the rays,
is itself quite cold.
MRS. B.
The same number of rays that are dispersed over the surface of the
mirror are collected by it into the focus; but, if you consider how
large a surface the mirror presents to the rays, and, consequently, how
much they are diffused in comparison to what they are at the focus,
which is little more than a point, I think you can no longer wonder
that the focus should be so much hotter than the mirror.
The principal use of the mirrors in this experiment is, to prove that
the calorific
emanation is reflected in the same manner as light.
57
CAROLINE.
And the result, I think, is very conclusive.
MRS. B.
The experiment may be repeated with a wax taper instead of the
bullet, with a view of separating the light from the caloric. For this
purpose a transparent plate of glass must be interposed between the
mirrors; for light, you know, passes with great facility through glass,
whilst the transmission of caloric is almost wholly impeded by it. We
shall find, however, in this experiment, that some few of the calorific
rays pass through the glass together with the light, as the thermometer
rises a little; but, as soon as the glass is removed, and a free passage
left to the caloric, it will rise considerably higher.
EMILY.
This experiment, as well as that of Dr. Herschell’s, proves that
light and heat may be separated; for in the latter experiment the
separation was not perfect, any more than in that of Mr. Pictet.
CAROLINE.
I should like to repeat this experiment, with the difference of
substituting a cold body instead of the hot one, to see whether cold
would not be reflected as well as heat.
58
MRS. B.
That experiment was proposed to Mr. Pictet by an incredulous
philosopher like yourself, and he immediately tried it by substituting a
piece of ice in the place of the heated bullet.
CAROLINE.
Well, Mrs. B., and what was the result?
MRS. B.
That we shall see; I have procured some ice for the purpose.
EMILY.
The thermometer falls considerably!
CAROLINE.
And does not that prove that cold is not merely a negative
quality, implying simply an inferior degree of heat? The cold must be
positive, since it is capable of reflection.
MRS. B.
So it at first appeared to Mr. Pictet; but upon a little
consideration he found that it afforded only an additional proof of the
reflection of heat: this I shall endeavour to explain to you.
According to Mr. Prevost’s theory, we suppose that all bodies
whatever radiate caloric; the thermometer used in these experiments
therefore emits calorific rays in the same manner as any other
59
substance. When its temperature is in equilibrium with that of the
surrounding bodies, it receives as much caloric as it parts with, and no
change of temperature is produced. But when we introduce a body of a
lower temperature, such as a piece of ice, which parts with less caloric
than it receives, the consequence is, that its temperature is raised,
whilst that of the surrounding bodies is proportionally lowered.
EMILY.
If, for instance, I was to bring a large piece of ice into this room,
the ice would in time be melted, by absorbing caloric from the general
radiation which is going on throughout the room; and as it would
contribute very little caloric in return for what is absorbed, the room
would necessarily be cooled by it.
MRS. B.
Just so; and as in consequence of the mirrors, a more
considerable exchange of rays takes place between the ice and the
thermometer, than between these and any of the surrounding bodies, the
temperature of the thermometer must be more lowered than that of any
other adjacent object.
CAROLINE.
I confess I do not perfectly understand your explanation.
60
MRS. B.
This experiment is exactly similar to that made with the heated
bullet: for, if we consider the thermometer as the hot body (which it
certainly is in comparison to the ice), you may then easily understand
that it is by the loss of the calorific rays which the thermometer sends
to the ice, and not by any cold rays received from it, that the fall of
the mercury is occasioned: for the ice, far from emitting rays of cold,
sends forth rays of caloric, which diminish the loss sustained by the
thermometer.
Let us say, for instance, that the radiation of the thermometer
towards the ice is equal to 20, and that of the ice towards the
thermometer to 10: the exchange in favour of the ice is as 20 is to 10,
or the thermometer absolutely loses 10, whilst the ice
gains 10.
CAROLINE.
But if the ice actually sends rays of caloric to the thermometer,
must not the latter fall still lower when the ice is removed?
MRS. B.
No; for the space that the ice occupied, admits rays from all the
surrounding bodies to pass through it; and those being of the same
temperature as the thermometer, will not affect it, because as much heat
now returns to the thermometer as radiates from it.
61
CAROLINE.
I must confess that you have explained this in so satisfactory a
manner, that I cannot help being convinced now that cold has no real
claim to the rank of a positive being.
MRS. B.
Before I conclude the subject of radiation I must observe to you that
different bodies, (or rather surfaces,) possess the power of radiating
caloric in very different degrees.
Some very curious experiments have been made by Mr. Leslie on this
subject, and it was for this purpose that he invented the differential
thermometer; with its assistance he ascertained that black surfaces
radiate most, glass next, and polished surfaces the least of all.
EMILY.
Supposing these surfaces, of course, to be all of the same
temperature.
MRS. B.
Undoubtedly. I will now show you the very simple and ingenious
apparatus, by means of which he made these experiments. This cubical tin
vessel or canister, has each of its sides externally covered with
different materials; the one is simply blackened; the next is covered
with white
62
paper; the third with a pane of glass, and in the fourth the polished
tin surface remains uncovered. We shall fill this vessel with hot water,
so that there can be no doubt but that all its sides will be of the same
temperature. Now let us place it in the focus of one of the mirrors,
making each of its sides front it in succession. We shall begin with the
black surface.
CAROLINE.
It makes the thermometer which is in the focus of the other mirror
rise considerably. Let us turn the paper surface towards the mirror. The
thermometer falls a little, therefore of course this side cannot emit or
radiate so much caloric as the blackened side.
EMILY.
This is very surprising; for the sides are exactly of the same size,
and must be of the same temperature. But let us try the glass
surface.
MRS. B.
The thermometer continues falling, and with the plain surface it
falls still lower; these two surfaces therefore radiate less and
less.
CAROLINE.
I think I have found out the reason of this.
63
MRS. B.
I should be very happy to hear it, for it has not yet (to my
knowledge) been accounted for.
CAROLINE.
The water within the vessel gradually cools, and the thermometer in
consequence gradually falls.
MRS. B.
It is true that the water cools, but certainly in much less
proportion than the thermometer descends, as you will perceive if you
now change the tin surface for the black one.
CAROLINE.
I was mistaken certainly, for the thermometer rises again now that
the black surface fronts the mirror.
MRS. B.
And yet the water in the vessel is still cooling, Caroline.
EMILY.
I am surprised that the tin surface should radiate the least caloric, for a
metallic vessel filled with hot water, a silver teapot, for
instance, feels much hotter to the hand than one of black earthen
ware.
64
MRS. B.
That is owing to the different power which various bodies possess for
conducting caloric, a property which we shall presently
examine. Thus, although a metallic vessel feels warmer to the hand,
a vessel of this kind is known to preserve the heat of the liquid
within, better than one of any other materials; it is for this reason
that silver teapots make better tea than those of earthen ware.
EMILY.
According to these experiments, light-coloured dresses, in cold
weather, should keep us warmer than black clothes, since the latter
radiate so much more than the former.
MRS. B.
And that is actually the case.
EMILY.
This property, of different surfaces to radiate in different degrees,
appears to me to be at variance with the equilibrium of caloric; since
it would imply that those bodies which radiate most, must ultimately
become coldest.
Suppose that we were to vary this experiment, by using two metallic
vessels full of boiling water, the one blackened, the other not; would
not the black one cool the first?
65
CAROLINE.
True; but when they were both brought down to the temperature of the
room, the interchange of caloric between the canisters and the other
bodies of the room being then equal, their temperatures would remain the
same.
EMILY.
I do not see why that should be the case; for if different surfaces
of the same temperature radiate in different degrees when heated, why
should they not continue to do so when cooled down to the temperature of
the room?
MRS. B.
You have started a difficulty, Emily, which certainly requires
explanation. It is found by experiment that the power of absorption
corresponds with and is proportional to that of radiation; so that under
equal temperatures, bodies compensate for the greater loss they sustain
in consequence of their greater radiation by their greater absorption;
so that if you were to make your experiment in an atmosphere heated like
the canisters, to the temperature of boiling water, though it is true
that the canisters would radiate in different degrees, no change of
temperature would be produced in them, because they would each absorb
caloric in proportion to their respective radiation.
66
EMILY.
But would not the canisters of boiling water also absorb caloric in
different degrees in a room of the common temperature?
MRS. B.
Undoubtedly they would. But the various bodies in the room would not,
at a lower temperature, furnish either of the canisters with a
sufficiency of caloric to compensate for the loss they undergo; for,
suppose the black canister to absorb 400 rays of caloric, whilst the
metallic one absorbed only 200; yet if the former radiate 800, whilst
the latter radiates only 400, the black canister will be the first
cooled down to the temperature of the room. But from the moment the
equilibrium of temperature has taken place, the black canister, both
receiving and giving out 400 rays, and the metallic one 200, no change
of temperature will take place.
EMILY.
I now understand it extremely well. But what becomes of the surplus
of calorific rays, which good radiators emit and bad radiators refuse to
receive; they must wander about in search of a resting-place?
MRS. B.
They really do so; for they are rejected and sent
67
back, or, in other words, reflected by the bodies which are bad
radiators of caloric; and they are thus transmitted to other bodies
which happen to lie in their way, by which they are either absorbed or
again reflected, according as the property of reflection, or that of
absorption, predominates in these bodies.
CAROLINE.
I do not well understand the difference between radiating and
reflecting caloric, for the caloric that is reflected from a body
proceeds from it in straight lines, and may surely be said to radiate
from it?
MRS. B.
It is true that there at first appears to be a great analogy between
radiation and reflection, as they equally convey the idea
of the transmission of caloric.
But if you consider a little, you will perceive that when a body
radiates caloric, the heat which it emits not only proceeds from,
but has its origin in the body itself. Whilst when a body
reflects caloric, it parts with none of its own caloric, but only
reflects that which it receives from other bodies.
EMILY.
Of this difference we have very striking examples before us, in the
tin vessel of water, and the concave mirrors; the first radiates its own
heat,
68
the latter reflect the heat which they receive from other bodies.
CAROLINE.
Now, that I understand the difference, it no longer surprises me that
bodies which radiate, or part with their own caloric freely, should not
have the power of transmitting with equal facility that which they
receive from other bodies.
EMILY.
Yet no body can be said to possess caloric of its own, if all caloric
is originally derived from the sun.
MRS. B.
When I speak of a body radiating its own caloric, I mean that
which it has absorbed and incorporated either immediately from the sun’s
rays, or through the medium of any other substance.
CAROLINE.
It seems natural enough that the power of absorption should be in
opposition to that of reflection, for the more caloric a body receives,
the less it will reject.
EMILY.
And equally so that the power of radiation should correspond with
that of absorption. It is, in fact, cause and effect; for a body cannot
radiate
69
heat without having previously absorbed it; just as a spring that is
well fed flows abundantly.
MRS. B.
Fluids are in general very bad radiators of caloric; and air neither
radiates nor absorbs caloric in any sensible degree.
We have not yet concluded our observations on free caloric. But I
shall defer, till our next meeting, what I have further to say on this
subject. I believe it will afford us ample conversation for another
interview.
70

MRS. B.
In our last conversation, we began to
examine the tendency of caloric to restore an equilibrium of
temperature. This property, when once well understood, affords the
explanation of a great variety of facts which appeared formerly
unaccountable. You must observe, in the first place, that the effect of
this tendency is gradually to bring all bodies that are in contact to
the same temperature. Thus, the fire which burns in the grate,
communicates its heat from one object to another, till every part of the
room has an equal proportion of it.
EMILY.
And yet this book is not so cold as the table on which it lies,
though both are at an equal distance from the fire, and actually in
contact with each other, so that, according to your theory, they should
be exactly of the same temperature.
71
CAROLINE.
And the hearth, which is much nearer the fire than the carpet, is
certainly the colder of the two.
MRS. B.
If you ascertain the temperature of these several bodies by a
thermometer (which is a much more accurate test than your feeling), you
will find that it is exactly the same.
CAROLINE.
But if they are of the same temperature, why should the one feel
colder than the other?
MRS. B.
The hearth and the table feel colder than the carpet or the book,
because the latter are not such good conductors of heat as the
former. Caloric finds a more easy passage through marble and wood, than
through leather and worsted; the two former will therefore absorb heat
more rapidly from your hand, and consequently give it a stronger
sensation of cold than the two latter, although they are all of them
really of the same temperature.
CAROLINE.
So, then, the sensation I feel on touching a cold body, is in
proportion to the rapidity with which my hand yields its heat to that
body?
72
MRS. B.
Precisely; and, if you lay your hand successively on every object in
the room, you will discover which are good, and which are bad conductors
of heat, by the different degrees of cold you feel. But, in order to
ascertain this point, it is necessary that the several substances should
be of the same temperature, which will not be the case with those that
are very near the fire, or those that are exposed to a current of cold
air from a window or door.
EMILY.
But what is the reason that some bodies are better conductors of heat
than others?
MRS. B.
This is a point not well ascertained. It has been conjectured that a
certain union or adherence takes place between the caloric and the
particles of the body through which it passes. If this adherence be
strong, the body detains the heat, and parts with it slowly and
reluctantly; if slight, it propagates it freely and rapidly. The
conducting power of a body is therefore, inversely, as its tendency to
unite with caloric.
EMILY.
That is to say, that the best conductors are those that have the
least affinity for caloric.
73
MRS. B.
Yes; but the term affinity is objectionable in this case, because, as
that word is used to express a chemical attraction (which can be
destroyed only by decomposition), it cannot be applicable to the slight
and transient union that takes place between free caloric and the bodies
through which it passes; an union which is so weak, that it constantly
yields to the tendency which caloric has to an equilibrium. Now you
clearly understand, that the passage of caloric, through bodies that are
good conductors, is much more rapid than through those that are bad
conductors, and that the former both give and receive it more quickly,
and therefore, in a given time, more abundantly, than bad conductors,
which makes them feel either hotter or colder, though they may be, in
fact, both of the same temperature.
CAROLINE.
Yes, I understand it now; the table, and the book lying upon it,
being really of the same temperature, would each receive, in the same
space of time, the same quantity of heat from my hand, were their
conducting powers equal; but as the table is the best conductor of the
two, it will absorb the heat from my hand more rapidly, and consequently
produce a stronger sensation of cold than the book.
74
MRS. B.
Very well, my dear; and observe, likewise, that if you were to heat
the table and the book an equal number of degrees above the temperature
of your body, the table, which before felt the colder, would now feel
the hotter of the two; for, as in the first case it took the heat most
rapidly from your hand, so it will now impart heat most rapidly to it.
Thus the marble table, which seems to us colder than the mahogany one,
will prove the hotter of the two to the ice; for, if it takes heat more
rapidly from our hands, which are warmer, it will give out heat more
rapidly to the ice, which is colder. Do you understand the reason of
these apparently opposite effects?
EMILY.
Perfectly. A body which is a good conductor of caloric, affords it a
free passage; so that it penetrates through that body more rapidly than
through one which is a bad conductor; and consequently, if it is colder
than your hand, you lose more caloric, and if it is hotter, you gain
more than with a bad conductor of the same temperature.
MRS. B.
But you must observe that this is the case only when the conductors
are either hotter or colder than your hand; for, if you heat different
conductors
75
to the temperature of your body, they will all feel equally warm, since
the exchange of caloric between bodies of the same temperature is equal.
Now, can you tell me why flannel clothing, which is a very bad conductor
of heat, prevents our feeling cold?
CAROLINE.
It prevents the cold from penetrating .
. . . . . . .
MRS. B.
But you forget that cold is only a negative quality.
CAROLINE.
True; it only prevents the heat of our bodies from escaping so
rapidly as it would otherwise do.
MRS. B.
Now you have explained it right; the flannel rather keeps in the
heat, than keeps out the cold. Were the atmosphere of a higher
temperature than our bodies, it would be equally efficacious in keeping
their temperature at the same degree, as it would prevent the free
access of the external heat, by the difficulty with which it
conducts it.
EMILY.
This, I think, is very clear. Heat, whether external or internal,
cannot easily penetrate flannel;
76
therefore in cold weather it keeps us warm; and if the weather was
hotter than our bodies, it would keep us cool.
MRS. B.
The most dense bodies are, generally speaking, the best conductors of
heat; probably because the denser the body the greater are the number of
points or particles that come in contact with caloric. At the common
temperature of the atmosphere a piece of metal will feel much colder
than a piece of wood, and the latter than a piece of woollen cloth; this
again will feel colder than flannel; and down, which is one of the
lightest, is at the same time one of the warmest bodies.
CAROLINE.
This is, I suppose, the reason that the plumage of birds preserves
them so effectually from the influence of cold in winter?
MRS. B.
Yes; but though feathers in general are an excellent preservative
against cold, down is a kind of plumage peculiar to aquatic birds, and
covers their chest, which is the part most exposed to the water; for
though the surface of the water is not of a lower temperature than the
atmosphere, yet, as it is a better conductor of heat, it feels much
77
colder, consequently the chest of the bird requires a warmer covering
than any other part of its body. Besides, the breasts of aquatic birds
are exposed to cold not only from the temperature of the water, but also
from the velocity with which the breast of the bird strikes against it;
and likewise from the rapid evaporation occasioned in that part by the
air against which it strikes, after it has been moistened by dipping
from time to time into the water.
If you hold a finger of one hand motionless in a glass of water, and
at the same time move a finger of the other hand swiftly through water
of the same temperature, a different sensation will be soon
perceived in the different fingers.
Most animal substances, especially those which Providence has
assigned as a covering for animals, such as fur, wool, hair, skin,
&c. are bad conductors of heat, and are, on that account, such
excellent preservatives against the inclemency of winter, that our
warmest apparel is made of these materials.
EMILY.
Wood is, I dare say, not so good a conductor as metal, and it is for
that reason, no doubt, that silver teapots have always wooden
handles.
MRS. B.
Yes; and it is the facility with which metals
78
conduct caloric that made you suppose that a silver pot radiated more
caloric than an earthen one. The silver pot is in fact hotter to the
hand when in contact with it; but it is because its conducting power
more than counterbalances its deficiency in regard to radiation.
We have observed that the most dense bodies are in general the best
conductors; and metals, you know, are of that class. Porous bodies, such
as the earths and wood, are worse conductors, chiefly, I believe,
on account of their pores being filled with air; for air is a remarkably
bad conductor.
CAROLINE.
It is a very fortunate circumstance that air should be a bad
conductor, as it tends to preserve the heat of the body when exposed to
cold weather.
MRS. B.
It is one of the many benevolent dispensations of Providence, in
order to soften the inclemency of the seasons, and to render almost all
climates habitable to man.
In fluids of different densities, the power of conducting heat varies
no less remarkably; if you dip your hand into this vessel full of
mercury, you will scarcely conceive that its temperature is not lower
than that of the atmosphere.
79
CAROLINE.
Indeed I know not how to believe it, it feels so extremely
cold.—But we may easily ascertain its true temperature by the
thermometer.—It is really not colder than the air;—the
apparent difference then is produced merely by the difference of the
conducting power in mercury and in air.
MRS. B.
Yes; hence you may judge how little the sense of feeling is to be
relied on as a test of the temperature of bodies, and how necessary a
thermometer is for that purpose.
It has indeed been doubted whether fluids have the power of
conducting caloric in the same manner as solid bodies. Count Rumford,
a very few years since, attempted to prove, by a variety of
experiments, that fluids, when at rest, were not at all endowed with
this property.
CAROLINE.
How is that possible, since they are capable of imparting cold or
heat to us; for if they did not conduct heat, they would neither take it
from, nor give it to us?
MRS. B.
Count Rumford did not mean to say that fluids would not communicate
their heat to solid bodies;
80
but only that heat does not pervade fluids, that is to say, is not
transmitted from one particle of a fluid to another, in the same manner
as in solid bodies.
EMILY.
But when you heat a vessel of water over the fire, if the particles
of water do not communicate heat to each other, how does the water
become hot throughout?
MRS. B.
By constant agitation. Water, as you have seen, expands by heat in
the same manner as solid bodies; the heated particles of water,
therefore, at the bottom of the vessel, become specifically lighter than
the rest of the liquid, and consequently ascend to the surface, where,
parting with some of their heat to the colder atmosphere, they are
condensed, and give way to a fresh succession of heated particles
ascending from the bottom, which having thrown off their heat at the
surface, are in their turn displaced. Thus every particle is
successively heated at the bottom, and cooled at the surface of the
liquid; but as the fire communicates heat more rapidly than the
atmosphere cools the succession of surfaces, the whole of the liquid in
time becomes heated.
CAROLINE.
This accounts most ingeniously for the propagation
81
of heat upwards. But suppose you were to heat the upper surface of a
liquid, the particles being specifically lighter than those below, could
not descend: how therefore would the heat be communicated downwards?
MRS. B.
If there were no agitation to force the heated surface downwards,
Count Rumford assures us that the heat would not descend. In proof of
this he succeeded in making the upper surface of a vessel of water boil
and evaporate, while a cake of ice remained frozen at the bottom.
CAROLINE.
That is very extraordinary indeed!
MRS. B.
It appears so, because we are not accustomed to heat liquids by their
upper surface; but you will understand this theory better if I show you
the internal motion that takes place in liquids when they experience a
change of temperature. The motion of the liquid itself is indeed
invisible from the extreme minuteness of its particles; but if you mix
with it any coloured dust, or powder, of nearly the same specific
gravity as the liquid, you may judge of the internal motion of the
latter by that of the coloured dust it contains.—Do you see the
82
small pieces of amber moving about in the liquid contained in this
phial?
CAROLINE.
Yes, perfectly.
MRS. B.
We shall now immerse the phial in a glass of hot water, and the
motion of the liquid will be shown, by that which it communicates to the
amber.
EMILY.
I see two currents, the one rising along the sides of the phial, the
other descending in the centre: but I do not understand the reason of
this.
MRS. B.
The hot water communicates its caloric, through the medium of the
phial, to the particles of the fluid nearest to the glass; these dilate
and ascend laterally to the surface, where, in parting with their heat,
they are condensed, and in descending, form the central current.
CAROLINE.
This is indeed a very clear and satisfactory experiment; but how much
slower the currents now move than they did at first?
MRS. B.
It is because the circulation of particles has
83
nearly produced an equilibrium of temperature between the liquid in the
glass and that in the phial.
CAROLINE.
But these communicate laterally, and I thought that heat in liquids
could be propagated only upwards.
MRS. B.
You do not take notice that the heat is imparted from one liquid to
the other, through the medium of the phial itself, the external surface
of which receives the heat from the water in the glass, whilst its
internal surface transmits it to the liquid it contains. Now take the
phial out of the hot water, and observe the effect of its cooling.
EMILY.
The currents are reversed; the external current now descends, and the
internal one rises.—I guess the reason of this
change:—the phial being in contact with cold air instead of hot
water, the external particles are cooled instead of being heated; they
therefore descend and force up the central particles, which, being
warmer, are consequently lighter.
MRS. B.
It is just so. Count Rumford hence infers that no alteration of
temperature can take place in a fluid, without an internal motion of its
particles,
84
and as this motion is produced only by the comparative levity of the
heated particles, heat cannot be propagated downwards.
But though I believe that Count Rumford’s theory as to heat being
incapable of pervading fluids is not strictly correct, yet there is, no
doubt, much truth in his observation, that the communication is
materially promoted by a motion of the parts; and this accounts for the
cold that is found to prevail at the bottom of the lakes in Switzerland,
which are fed by rivers issuing from the snowy Alps. The water of these
rivers being colder, and therefore more dense than that of the lakes,
subsides to the bottom, where it cannot be affected by the warmer
temperature of the surface; the motion of the waves may communicate this
temperature to some little depth, but it can descend no further than the
agitation extends.
EMILY.
But when the atmosphere is colder than the lake, the colder surface
of the water will descend, for the very reason that the warmer will
not.
MRS. B.
Certainly: and it is on this account that neither a lake, nor any
body of water whatever, can be frozen until every particle of the water
has risen to the surface to give off its caloric to the colder
85
atmosphere; therefore the deeper a body of water is, the longer will be
the time it requires to be frozen.
EMILY.
But if the temperature of the whole body of water be brought down to
the freezing point, why is only the surface frozen?
MRS. B.
The temperature of the whole body is lowered, but not to the freezing
point. The diminution of heat, as you know, produces a contraction in
the bulk of fluids, as well as of solids. This effect, however, does not
take place in water below the temperature of 40 degrees, which is 8
degrees above the freezing point. At that temperature, therefore, the
internal motion, occasioned by the increased specific gravity of the
condensed particles, ceases; for when the water at the surface no longer
condenses, it will no longer descend, and leave a fresh surface exposed
to the atmosphere: this surface alone, therefore, will be further
exposed to its severity, and will soon be brought down to the freezing
point, when it becomes ice, which being a bad conductor of heat,
preserves the water beneath a long time from being affected by the
external cold.
CAROLINE.
And the sea does not freeze, I suppose, because
86
its depth is so great, that a frost never lasts long enough to bring
down the temperature of such a great body of water to 40 degrees?
MRS. B.
That is one reason why the sea, as a large mass of water, does not
freeze. But, independently of this, salt water does not freeze till it
is cooled much below 32 degrees, and with respect to the law of
condensation, salt water is an exception, as it condenses even many
degrees below the freezing point. When the caloric of fresh water,
therefore, is imprisoned by the ice on its surface, the ocean still
continues throwing off heat into the atmosphere, which is a most signal
dispensation of Providence to moderate the intensity of the cold in
winter.
CAROLINE.
This theory of the non-conducting power of liquids, does not,
I suppose, hold good with respect to air, otherwise the atmosphere
would not be heated by the rays of the sun passing through it?
MRS. B.
Nor is it heated in that way. The pure atmosphere is a perfectly
transparent medium, which neither radiates, absorbs, nor conducts
caloric, but transmits the rays of the sun to us without in any way
87
diminishing their intensity. The air is therefore not more heated, by
the sun’s rays passing through it, than diamond, glass, water, or any
other transparent medium.
CAROLINE.
That is very extraordinary! Are glass windows not heated then by the
sun shining on them?
MRS. B.
No; not if the glass be perfectly transparent. A most convincing
proof that glass transmits the rays of the sun without being heated by
them is afforded by the burning lens, which by converging the rays to a
focus will set combustible bodies on fire, without its own temperature
being raised.
EMILY.
Yet, Mrs. B., if I hold a piece of glass near the fire it is almost
immediately warmed by it; the glass therefore must retain some of the
caloric radiated by the fire? Is it that the solar rays alone pass
freely through glass without paying tribute? It seems unaccountable that
the radiation of a common fire should have power to do what the sun’s
rays cannot accomplish.
MRS. B.
It is not because the rays from the fire have more power, but rather
because they have less, that
88
they heat glass and other transparent bodies. It is true, however, that
as you approach the source of heat the rays being nearer each other, the
heat is more condensed, and can produce effects of which the solar rays,
from the great distance of their source, are incapable. Thus we should
find it impossible to roast a joint of meat by the sun’s rays, though it
is so easily done by culinary heat. Yet caloric emanated from burning
bodies, which is commonly called culinary heat, has neither the
intensity nor the velocity of solar rays. All caloric, we have said, is
supposed to proceed originally from the sun; but after having been
incorporated with terrestrial bodies, and again given out by them,
though its nature is not essentially altered, it retains neither the
intensity nor the velocity with which it first emanated from that
luminary; it has therefore not the power of passing through transparent
mediums, such as glass and water, without being partially retained by
those bodies.
EMILY.
I recollect that in the experiment on the reflection of heat, the
glass skreen which
you interposed between the burning taper and the mirror, arrested the
rays of caloric, and suffered only those of light to pass
through it.
CAROLINE.
Glass windows, then, though they cannot be
89
heated by the sun shining on them, may be heated internally by a fire in
the room? But, Mrs. B., since the atmosphere is not warmed by the
solar rays passing through it, how does it obtain heat; for all the
fires that are burning on the surface of the earth would contribute very
little towards warming it?
EMILY.
The radiation of heat is not confined to burning bodies: for all
bodies, you know, have that property; therefore, not only every thing
upon the surface of the earth, but the earth itself, must radiate heat;
and this terrestrial caloric, not having, I suppose, sufficient
power to traverse the atmosphere, communicates heat to it.
MRS. B.
Your inference is extremely well drawn, Emily; but the foundation on
which it rests is not sound; for the fact is, that terrestrial or
culinary heat, though it cannot pass through the denser transparent
mediums, such as glass or water, without loss, traverses the atmosphere
completely: so that all the heat which the earth radiates, unless it
meet with clouds or any foreign body to intercept its passage, passes
into the distant regions of the universe.
CAROLINE.
What a pity that so much heat should be wasted!
90
MRS. B.
Before you are tempted to object to any law of nature, reflect
whether it may not prove to be one of the numberless dispensations of
Providence for our good. If all the heat which the earth has received
from the sun, since the creation had been accumulated in it, its
temperature by this time would, no doubt, have been more elevated than
any human being could have borne.
CAROLINE.
I spoke indeed very inconsiderately. But, Mrs. B., though the
earth, at such a high temperature, might have scorched our feet, we
should always have had a cool refreshing air to breathe, since the
radiation of the earth does not heat the atmosphere.
EMILY.
The cool air would have afforded but very insufficient refreshment,
whilst our bodies were exposed to the burning radiation of the
earth.
MRS. B.
Nor should we have breathed a cool air; for though it is true that
heat is not communicated to the atmosphere by radiation, yet the air is
warmed by contact with heated bodies, in the same manner as solids or
liquids. The stratum of air which is immediately in contact with the
earth is heated by
91
it; it becomes specifically lighter and rises, making way for another
stratum of air which is in its turn heated and carried upwards; and thus
each successive stratum of air is warmed by coming in contact with the
earth. You may perceive this effect in a sultry day, if you attentively
observe the strata of air near the surface of the earth; they appear in
constant agitation, for though it is true the air is itself invisible,
yet the sun shining on the vapours floating in it, render them visible,
like the amber dust in the water. The temperature of the surface of the
earth is therefore the source from whence the atmosphere derives its
heat, though it is communicated neither by radiation, nor transmitted
from one particle of it to another by the conducting power; but every
particle of air must come in contact with the earth in order to receive
heat from it.
EMILY.
Wind then by agitating the air should contribute to cool the earth
and warm the atmosphere, by bringing a more rapid succession of fresh
strata of air in contact with the earth, and yet in general wind feels
cooler than still air?
MRS. B.
Because the agitation of the air carries off heat from the surface of
our bodies more rapidly than
92
still air, by occasioning a greater number of points of contact in a
given time.
EMILY.
Since it is from the earth and not the sun that the atmosphere
receives its heat, I no longer wonder that elevated regions should
be colder than plains and valleys; it was always a subject of
astonishment to me, that in ascending a mountain and approaching the
sun, the air became colder instead of being more heated.
MRS. B.
At the distance of about a hundred million of miles, which we are
from the sun, the approach of a few thousand feet makes no sensible
difference, whilst it produces a very considerable effect with regard to
the warming the atmosphere at the surface of the earth.
CAROLINE.
Yet as the warm air rises from the earth and the cold air descends to
it, I should have supposed that heat would have accumulated in the
upper regions of the atmosphere, and that we should have felt the air
warmer as we ascended?
MRS. B.
The atmosphere, you know, diminishes in density, and consequently in
weight, as it is more distant
93
from the earth; the warm air, therefore, rises only till it meets with a
stratum of air of its own density; and it will not ascend into the upper
regions of the atmosphere until all the parts beneath have been
previously heated. The length of summer even in warm climates does not
heat the air sufficiently to melt the snow which has accumulated during
the winter on very high mountains, although they are almost constantly
exposed to the heat of the sun’s rays, being too much elevated to be
often enveloped in clouds.
EMILY.
These explanations are very satisfactory; but allow me to ask you one
more question respecting the increased levity of heated liquids. You
said that when water was heated over the fire, the particles at the
bottom of the vessel ascended as soon as heated, in consequence of their
specific levity: why does not the same effect continue when the water
boils, and is converted into steam? and why does the steam rise from the
surface, instead of the bottom of the liquid?
MRS. B.
The steam or vapour does ascend from the bottom, though it seems to
arise from the surface of the liquid. We shall boil some water in this
Florence flask, (Plate IV.
Fig. 1.) in order that
94
you may be well acquainted with the process of ebullition;—you
will then see, through the glass, that the vapour rises in bubbles from
the bottom. We shall make it boil by means of a lamp, which is more
convenient for this purpose than the chimney fire.
Plate IV.
Vol. I. p. 84.
Fig. 2. Boiling water in a flask over a Patent lamp.
Larger view
(complete Plate)
EMILY.
I see some small bubbles ascend, and a great many appear all over the
inside of the flask; does the water begin to boil already?
MRS. B.
No; what you now see are bubbles of air, which were either dissolved
in the water, or attached to the inner surface of the flask, and which,
being rarefied by the heat, ascend in the water.
EMILY.
But the heat which rarefies the air inclosed in the water must rarefy
the water at the same time; therefore, if it could remain stationary in
the water when both were cold, I do not understand why it should
not when both are equally heated?
MRS. B.
Air being much less dense than water, is more easily rarefied; the
former, therefore, expands to a great extent, whilst the latter
continues to occupy
95
nearly the same space; for water dilates comparatively but very little
without changing its state and becoming vapour. Now that the water in
the flask begins to boil, observe what large bubbles rise from the
bottom of it.
EMILY.
I see them perfectly; but I wonder that they have sufficient power to
force themselves through the water.
CAROLINE.
They must rise, you know, from their specific levity.
MRS. B.
You are right, Caroline; but vapour has not in all liquids (when
brought to the degree of vaporization) the power of overcoming the
pressure of the less heated surface. Metals, for instance, mercury
excepted, evaporate only from the surface; therefore no vapour will
ascend from them till the degree of heat which is necessary to form it
has reached the surface; that is to say, till the whole of the liquid is
brought to a state of ebullition.
EMILY.
I have observed that steam, immediately issuing from the spout of a
teakettle, is less visible than at a further distance from it; yet it
must be more
96
dense when it first evaporates, than when it begins to diffuse itself in
the air.
MRS. B.
When the steam is first formed, it is so perfectly dissolved by
caloric, as to be invisible. In order however to understand this, it
will be necessary for me to enter into some explanation respecting the
nature of SOLUTION. Solution takes
place whenever a body is melted in a fluid. In this operation the body
is reduced to such a minute state of division by the fluid, as to become
invisible in it, and to partake of its fluidity; but in common solutions
this happens without any decomposition, the body being only divided into
its integrant particles by the fluid in which it is melted.
CAROLINE.
It is then a mode of destroying the attraction of aggregation.
MRS. B.
Undoubtedly.—The two principal solvent fluids are water,
and caloric. You may have observed that if you melt salt in
water, it totally disappears, and the water remains clear, and
transparent as before; yet though the union of these two bodies appears
so perfect, it is not produced by any chemical combination; both the
salt and the water remain unchanged; and if you were to separate
97
them by evaporating the latter, you would find the salt in the same
state as before.
EMILY.
I suppose that water is a solvent for solid bodies, and caloric for
liquids?
MRS. B.
Liquids of course can only be converted into vapour by caloric. But
the solvent power of this agent is not at all confined to that class of
bodies; a great variety of solid substances are dissolved by heat:
thus metals, which are insoluble in water, can be dissolved by intense
heat, being first fused or converted into a liquid, and then rarefied
into an invisible vapour. Many other bodies, such as salt, gums, &c.
yield to either of these solvents.
CAROLINE.
And that, no doubt, is the reason why hot water will melt them so
much better than cold water?
MRS. B.
It is so. Caloric may, indeed, be considered as having, in every
instance, some share in the solution of a body by water, since water,
however low its temperature may be, always contains more or less
caloric.
98
EMILY.
Then, perhaps, water owes its solvent power merely to the caloric
contained in it?
MRS. B.
That, probably, would be carrying the speculation too far;
I should rather think that water and caloric unite their efforts to
dissolve a body, and that the difficulty or facility of effecting this,
depend both on the degree of attraction of aggregation to be overcome,
and on the arrangement of the particles which are more or less disposed
to be divided and penetrated by the solvent.
EMILY.
But have not all liquids the same solvent power as water?
MRS. B.
The solvent power of other liquids varies according to their nature,
and that of the substances submitted to their action. Most of these
solvents, indeed, differ essentially from water, as they do not merely
separate the integrant particles of the bodies which they dissolve, but
attack their constituent principles by the power of chemical attraction,
thus producing a true decomposition. These more complicated operations
we must consider in another place, and confine our attention
99
at present to the solutions by water and caloric.
CAROLINE.
But there are a variety of substances which, when dissolved in water,
make it thick and muddy, and destroy its transparency.
MRS. B.
In this case it is not a solution, but simply a mixture. I shall
show you the difference between a solution and a mixture, by
putting some common salt into one glass of water, and some powder of
chalk into another; both these substances are white, but their effect on
the water will be very different.
CAROLINE.
Very different indeed! The salt entirely disappears and leaves the
water transparent, whilst the chalk changes it into an opaque liquid
like milk.
EMILY.
And would lumps of chalk and salt produce similar effects on
water?
MRS. B.
Yes, but not so rapidly; salt is, indeed, soon melted though in a
lump; but chalk, which does not mix so readily with water, would require
a
100
much greater length of time; I therefore preferred showing you the
experiment with both substances reduced to powder, which does not in any
respect alter their nature, but facilitates the operation merely by
presenting a greater quantity of surface to the water.
I must not forget to mention a very curious circumstance respecting
solutions, which is, that a fluid is not nearly so much increased in
bulk by holding a body in solution, as it would by mere mixture with the
body.
CAROLINE.
That seems impossible; for two bodies cannot exist together in the
same space.
MRS. B.
Two bodies may, by condensation, occupy less space when in union than
when separate, and this I can show you by an easy experiment.
This phial, which contains some salt, I shall fill with water,
pouring it in quickly, so as not to dissolve much of the salt; and when
it is quite full I cork it.—If I now shake the phial till the salt
is dissolved, you will observe that it is no longer full.
CAROLINE.
I shall try to add a little more salt.—But now, you see,
Mrs. B., the water runs over.
101
MRS. B.
Yes; but observe that the last quantity of salt you put in remains
solid at the bottom, and displaces the water; for it has already melted
all the salt it is capable of holding in solution. This is called the
point of saturation; and the water in this case is said to be
saturated with salt.
EMILY.
I think I now understand the solution of a solid body by water
perfectly: but I have not so clear an idea of the solution of a liquid
by caloric.
MRS. B.
It is probably of a similar nature; but as caloric is an invisible
fluid, its action as a solvent is not so obvious as that of water.
Caloric, we may conceive, dissolves water, and converts it into vapour
by the same process as water dissolves salt; that is to say, the
particles of water are so minutely divided by the caloric as to become
invisible. Thus, you are now enabled to understand why the vapour of
boiling water, when it first issues from the spout of a kettle, is
invisible; it is so, because it is then completely dissolved by caloric.
But the air with which it comes in contact, being much colder than the
vapour, the latter yields to it a quantity of its caloric. The particles
of vapour being thus in a great measure deprived
102
of their solvent, gradually collect, and become visible in the form of
steam, which is water in a state of imperfect solution; and if you were
further to deprive it of its caloric, it would return to its original
liquid state.
CAROLINE.
That I understand very well. If you hold a cold plate over a tea-urn,
the steam issuing from it will be immediately converted into drops of
water by parting with its caloric to the plate; but in what state is the
steam, when it becomes invisible by being diffused in the air?
MRS. B.
It is not merely diffused, but is again dissolved by the air.
EMILY.
The air, then, has a solvent power, like water and caloric?
MRS. B.
This was formerly believed to be the case. But it appears from more
recent enquiries that the solvent power of the atmosphere depends solely
upon the caloric contained in it. Sometimes the watery vapour diffused
in the atmosphere is but imperfectly dissolved, as is the case in the
formation of clouds and fogs; but if it gets into a region sufficiently
warm, it becomes perfectly invisible.
103
EMILY.
Can any water dissolve in the atmosphere without its being previously
converted into vapour by boiling?
MRS. B.
Unquestionably; and this constitutes the difference between
vaporization and evaporation. Water, when heated to the
boiling point, can no longer exist in the form of water, and must
necessarily be converted into vapour or steam, whatever may be the state
and temperature of the surrounding medium; this is called vaporization.
But the atmosphere, by means of the caloric it contains, can take up a
certain portion of water at any temperature, and hold it in a state of
solution. This is simply evaporation. Thus the atmosphere is continually
carrying off moisture from the surface of the earth, until it is
saturated with it.
CAROLINE.
That is the case, no doubt, when we feel the atmosphere damp.
MRS. B.
On the contrary, when the moisture is well dissolved it occasions no
humidity: it is only when in a state of imperfect solution and floating
in the atmosphere, in the form of watery vapour, that it produces
dampness. This happens more frequently
104
in winter than in summer; for the lower the temperature of the
atmosphere, the less water it can dissolve; and in reality it never
contains so much moisture as in a dry hot summer’s day.
CAROLINE.
You astonish me! But why, then, is the air so dry in frosty weather,
when its temperature is at the lowest?
EMILY.
This, I conjecture, proceeds not so much from the moisture being
dissolved, as from its being frozen; is not that the case?
MRS. B.
It is; and the freezing of the watery vapour which the atmospheric
heat could not dissolve, produces what is called a hoar frost; for the
particles descend in freezing, and attach themselves to whatever they
meet with on the surface of the earth.
The tendency of free caloric to an equilibrium, together with its
solvent power, are likewise connected with the phenomena of rain, of
dew, &c. When moist air of a certain temperature happens to
pass through a colder region of the atmosphere, it parts with a portion
of its heat to the surrounding air; the quantity of caloric, therefore,
which served to keep the water in a state of
105
vapour, being diminished, the watery particles approach each other, and
form themselves into drops of water, which being heavier than the
atmosphere, descend to the earth. There are also other circumstances,
and particularly the variation in the weight of the atmosphere, which
may contribute to the formation of rain. This, however, is an intricate
subject, into which we cannot more fully enter at present.
EMILY.
In what manner do you account for the formation of dew?
MRS. B.
Dew is a deposition of watery particles or minute drops from the
atmosphere, precipitated by the coolness of the evening.
CAROLINE.
This precipitation is owing, I suppose, to the cooling of the
atmosphere, which prevents its retaining so great a quantity of watery
vapour in solution as during the heat of the day.
MRS. B.
Such was, from time immemorial, the generally received opinion
respecting the cause of dew; but it has been very recently proved by a
course of ingenious experiments of Dr. Wells, that the deposition
106
of dew is produced by the cooling of the surface of the earth, which he
has shown to take place previously to the cooling of the atmosphere; for
on examining the temperature of a plot of grass just before the
dew-fall, he found that it
was considerably colder than the air a few feet above it,
from which the dew was shortly after precipitated.
EMILY.
But why should the earth cool in the evening sooner than the
atmosphere?
MRS. B.
Because it parts with its heat more readily than the air; the earth
is an excellent radiator of caloric, whilst the atmosphere does not
possess that property, at least in any sensible degree. Towards evening,
therefore, when the solar heat declines, and when after sunset it
entirely ceases, the earth rapidly cools by radiating heat towards the
skies; whilst the air has no means of parting with its heat but by
coming into contact with the cooled surface of the earth, to which it
communicates its caloric. Its solvent power being thus reduced, it is
unable to retain so large a portion of watery vapour, and deposits those
pearly drops which we call dew.
EMILY.
If this be the cause of dew, we need not be apprehensive
107
of receiving any injury from it; for it can be deposited only on
surfaces that are colder than the atmosphere, which is never the case
with our bodies.
MRS. B.
Very true; yet I would not advise you for this reason to be too
confident of escaping all the ill effects which may arise from exposure
to the dew; for it may be deposited on your clothes, and chill you
afterwards by its evaporation from them. Besides, whenever the dew is
copious, there is a chill in the atmosphere which it is not always safe
to encounter.
CAROLINE.
Wind, then, must promote the deposition of dew, by bringing a more
rapid succession of particles of air in contact with the earth, just as
it promotes the cooling of the earth and warming of the atmosphere
during the heat of the day?
MRS. B.
Yes; provided the wind be unattended with clouds, for these
accumulations of moisture not only prevent the free radiation of the
earth towards the upper regions, but themselves radiate towards the
earth; under these circumstances much less dew is formed than on fine
clear nights, when the radiation of the earth passes without obstacle
through the atmosphere to the distant regions of space, whence it
108
receives no caloric in exchange. The dew continues to be deposited
during the night, and is generally most abundant towards morning, when
the contrast between the temperature of the earth and that of the air is
greatest. After sunrise the equilibrium of temperature between these two
bodies is gradually restored by the solar rays passing freely through
the atmosphere to the earth; and later in the morning the temperature of
the earth gains the ascendency, and gives out caloric to the air by
contact, in the same manner as it receives it from the air during the
night.—Can you tell me, now, why a bottle of wine taken fresh from
the cellar (in summer particularly), will soon be covered with dew; and
even the glasses into which the wine is poured will be moistened with a
similar vapour?
EMILY.
The bottle being colder than the surrounding air, must absorb caloric
from it; the moisture therefore which that air contained becomes
visible, and forms the dew which is deposited on the bottle.
MRS. B.
Very well, Emily. Now, Caroline, can you inform me why, in a warm
room, or close carriage, the contrary effect takes place; that is to
say, that the inside of the windows is covered with vapour?
109
CAROLINE.
I have heard that it proceeds from the breath of those within the
room or the carriage; and I suppose it is occasioned by the windows
which, being colder than the breath, deprive it of part of its caloric,
and by this means convert it into watery vapour.
MRS. B.
You have both explained it extremely well. Bodies attract dew in
proportion as they are good radiators of caloric, as it is this quality
which reduces their temperature below that of the atmosphere; hence we
find that little or no dew is deposited on rocks, sand, water; while
grass and living vegetables, to which it is so highly beneficial,
attract it in abundance—another remarkable instance of the wise
and bountiful dispensations of Providence.
EMILY.
And we may again observe it in the abundance of dew in summer, and in
hot climates, when its cooling effects are so much required; but I do
not understand what natural cause increases the dew in hot weather?
MRS. B.
The more caloric the earth receives during the day, the more it will
radiate afterwards, and consequently the more rapidly its temperature
will be reduced in the evening, in comparison to that of the
110
atmosphere. In the West-Indies especially, where the intense heat of the
day is strongly contrasted with the coolness of the evening, the dew is
prodigiously abundant. During a drought, the dew is less plentiful, as
the earth is not sufficiently supplied with moisture to be able to
saturate the atmosphere.
CAROLINE.
I have often observed, Mrs. B., that when I walk out in frosty
weather, with a veil over my face, my breath freezes upon it. Pray what
is the reason of that?
MRS. B.
It is because the cold air immediately seizes on the caloric of your
breath, and, by robbing it of its solvent, reduces it to a denser fluid,
which is the watery vapour that settles on your veil, and there it
continues parting with its caloric till it is brought down to the
temperature of the atmosphere, and assumes the form of ice.
You may, perhaps, have observed that the breath of animals, or rather
the moisture contained in it, is visible in damp weather, or during a
frost. In the former case, the atmosphere being over-saturated with
moisture, can dissolve no more. In the latter, the cold condenses it
into visible vapour; and for the same reason, the steam arising from
water that is warmer than the atmosphere,
111
becomes visible. Have you never taken notice of the vapour rising from
your hands after having dipped them into warm water?
CAROLINE.
Frequently, especially in frosty weather.
MRS. B.
We have already observed that pressure is an obstacle to evaporation:
there are liquids that contain so great a quantity of caloric, and whose
particles consequently adhere so slightly together, that they may be
rapidly converted into vapour without any elevation of temperature,
merely by taking off the weight of the atmosphere. In such liquids, you
perceive, it is the pressure of the atmosphere alone that connects their
particles, and keeps them in a liquid state.
CAROLINE.
I do not well understand why the particles of such fluids should be
disunited and converted into vapour, without any elevation of
temperature, in spite of the attraction of cohesion.
MRS. B.
It is because the degree of heat at which we usually observe these
fluids is sufficient to overcome their attraction of cohesion. Ether is
of this description;
112
it will boil and be converted into vapour, at the common temperature of
the air, if the pressure of the atmosphere be taken off.
EMILY.
I thought that ether would evaporate without either the pressure of
the atmosphere being taken away, or heat applied; and that it was for
that reason so necessary to keep it carefully corked up?
MRS. B.
It is true it will evaporate, but without ebullition; what I am now
speaking of is the vaporization of ether, or its conversion into vapour
by boiling. I am going to show you how suddenly the ether in this
phial will be converted into vapour, by means of the
air-pump.—Observe with what rapidity the bubbles ascend, as I take
off the pressure of the atmosphere.
CAROLINE.
It positively boils: how singular to see a liquid boil without
heat!
MRS. B.
Now I shall place the phial of ether in this glass, which it nearly
fits, so as to leave only a small space, which I fill with water; and in
this state I put it again under the receiver.
113
(Plate IV. Fig. 1.)* You will
observe, as I exhaust the air from it, that whilst the ether boils, the
water freezes.
Plate IV.
Vol. I. p. 84.
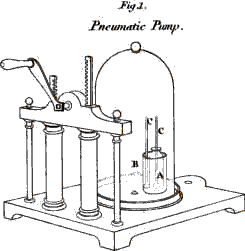
Fig. 1. Ether evaporated & water frozen in the air pump.
A Phial of Ether.
B Glass vessel containing water.
C.C Thermometers, one
in the Ether, the other in the water.
Larger view
(complete Plate)
CAROLINE.
It is indeed wonderful to see water freeze in contact with a boiling
fluid!
EMILY.
I am at a loss to conceive how the ether can pass to the state of
vapour without an addition of caloric. Does it not contain more caloric
in a state of vapour, than in a state of liquidity?
MRS. B.
It certainly does; for though it is the pressure of the atmosphere
which condenses it into a liquid, it is by forcing out the caloric that
belongs to it when in an aëriform state.
114
EMILY.
You have, therefore, two difficulties to explain,
Mrs. B.—First, from whence the ether obtains the caloric
necessary to convert it into vapour when it is relieved from the
pressure of the atmosphere; and, secondly, what is the reason that the
water, in which the bottle of ether stands, is frozen?
CAROLINE.
Now, I think, I can answer both these questions. The ether
obtains the addition of caloric required, from the water in the glass;
and the loss of caloric, which the latter sustains, is the occasion of
its freezing.
MRS. B.
You are perfectly right; and if you look at the thermometer which I
have placed in the water, whilst I am working the pump, you will see
that every time bubbles of vapour are produced, the mercury descends;
which proves that the heat of the water diminishes in proportion as the
ether boils.
EMILY.
This I understand now very well; but if the water freezes in
consequence of yielding its caloric to the ether, the equilibrium of
heat must, in this case, be totally destroyed. Yet you have told us,
that the exchange of caloric between two bodies of
115
equal temperature, was always equal; how, then, is it that the water,
which was originally of the same temperature as the ether, gives out
caloric to it, till the water is frozen, and the ether made to boil?
MRS. B.
I suspected that you would make these objections; and, in order to
remove them, I enclosed two thermometers in the air-pump; one which
stands in the glass of water, the other in the phial of ether; and you
may see that the equilibrium of temperature is not destroyed; for as the
thermometer descends in the water, that in the ether sinks in the same
manner; so that both thermometers indicate the same temperature, though
one of them is in a boiling, the other in a freezing liquid.
EMILY.
The ether, then, becomes colder as it boils? This is so contrary to
common experience, that I confess it astonishes me exceedingly.
CAROLINE.
It is, indeed, a most extraordinary circumstance. But pray, how do
you account for it?
MRS. B.
I cannot satisfy your curiosity at present; for before we can attempt
to explain this apparent
116
paradox, it is necessary to become acquainted with the subject of LATENT HEAT: and that, I think, we must
defer till our next interview.
CAROLINE.
I believe, Mrs. B., that you are glad to put off the explanation; for
it must be a very difficult point to account for.
MRS. B.
I hope, however, that I shall do it to your complete
satisfaction.
EMILY.
But before we part, give me leave to ask you one question. Would not
water, as well as ether, boil with less heat, if deprived of the
pressure of the atmosphere?
MRS. B.
Undoubtedly. You must always recollect that there are two forces to
overcome, in order to make a liquid boil or evaporate; the attraction of
aggregation, and the weight of the atmosphere. On the summit of a high
mountain (as Mr. De Saussure ascertained on Mount Blanc) much less heat
is required to make water boil, than in the plain, where the weight of
the atmosphere is
117
greater.*
Indeed if the weight of the atmosphere be entirely removed by means of a
good air-pump, and if water be placed in the exhausted receiver, it will
evaporate so fast, however cold it maybe, as to give it the appearance
of boiling from the surface. But without the assistance of the air-pump,
I can show you a very pretty experiment, which proves the effect of
the pressure of the atmosphere in this respect.
Observe, that this Florence flask is about half full of water, and
the upper half of invisible vapour, the water being in the act of
boiling.—I take it from the lamp, and cork it
carefully—the water, you see, immediately ceases
boiling.—I shall now dip the flask into a bason of cold
water.†
CAROLINE.
But look, Mrs. B., the hot water begins to boil again, although the
cold water must rob it more and more of its caloric! What can be the
reason of that?
118
MRS. B.
Let us examine its temperature. You see the thermometer immersed in
it remains stationary at 180 degrees, which is about 30 degrees below
the boiling point. When I took the flask from the lamp, I observed
to you that the upper part of it was filled with vapour; this being
compelled to yield its caloric to the cold water, was again condensed
into water—What, then, filled the upper part of the flask?
EMILY.
Nothing; for it was too well corked for the air to gain admittance,
and therefore the upper part of the flask must be a vacuum.
MRS. B.
The water below, therefore, no longer sustains the pressure of the
atmosphere, and will consequently boil at a much lower temperature.
Thus, you see, though it had lost many degrees of heat, it began boiling
again the instant the vacuum was formed above it. The boiling has now
ceased, the temperature of the water being still farther reduced; if it
had been ether, instead of water, it would have continued boiling much
longer, for ether boils, under the usual atmospheric pressure, at a
temperature as low as 100 degrees; and in a vacuum it boils at almost
any temperature; but
119
water being a more dense fluid, requires a more considerable quantity of
caloric to make it evaporate quickly, even when the pressure of the
atmosphere is removed.
EMILY.
What proportion of vapour can the atmosphere contain in a state of
solution?
MRS. B.
I do not know whether it has been exactly ascertained by experiment;
but at any rate this proportion must vary, both according to the
temperature and the weight of the atmosphere; for the lower the
temperature, and the greater the pressure, the smaller must be the
proportion of vapour that the atmosphere can contain.
To conclude the subject of free caloric, I should mention
Ignition, by which is meant that emission of light which is
produced in bodies at a very high temperature, and which is the effect
of accumulated caloric.
EMILY.
You mean, I suppose, that light which is produced by a burning
body?
MRS. B.
No: ignition is quite independent of combustion. Clay, chalk, and
indeed all incombustible
120
substances, may be made red hot. When a body burns, the light emitted is
the effect of a chemical change which takes place, whilst ignition is
the effect of caloric alone, and no other change than that of
temperature is produced in the ignited body.
All solid bodies, and most liquids, are susceptible of ignition, or,
in other words, of being heated so as to become luminous; and it is
remarkable that this takes place pretty nearly at the same temperature
in all bodies, that is, at about 800 degrees of Fahrenheit’s scale.
EMILY.
But how can liquids attain so high a temperature, without being
converted into vapour?
MRS. B.
By means of confinement and pressure. Water confined in a strong iron
vessel (called Papin’s digester) can have its temperature raised to
upwards of 400 degrees. Sir James Hall has made some very curious
experiments on the effects of heat assisted by pressure; by means of
strong gun-barrels, he succeeded in melting a variety of substances
which were considered as infusible: and it is not unlikely that, by
similar methods, water itself might be heated to redness.
121
EMILY.
I am surprised at that: for I thought that the force of steam was
such as to destroy almost all mechanical resistance.
MRS. B.
The expansive force of steam is prodigious; but in order to subject
water to such high temperatures, it is prevented by confinement from
being converted into steam, and the expansion of heated water is
comparatively trifling.—But we have dwelt so long on the subject
of free caloric, that we must reserve the other modifications of that
agent to our next meeting, when we shall endeavour to proceed more
rapidly.
122
CONVERSATION IV.
ON COMBINED CALORIC, COMPREHENDING SPECIFIC AND
LATENT HEAT.

MRS. B.
We are now to examine the other
modifications of caloric.
CAROLINE.
I am very curious to know of what nature they can be; for I have no
notion of any kind of heat that is not perceptible to the senses.
MRS. B.
In order to enable you to understand them, it will be necessary to
enter into some previous explanations.
It has been discovered by modern chemists, that bodies of a different
nature, heated to the same temperature, do not contain the same quantity
of caloric.
CAROLINE.
How could that be ascertained? Have you not told us that it is
impossible to discover the absolute quantity of caloric which bodies
contain?
123
MRS. B.
True; but at the same time I said that we were enabled to form a
judgment of the proportions which bodies bore to each other in this
respect. Thus it is found that, in order to raise the temperature of
different bodies the same number of degrees, different quantities of
caloric are required for each of them. If, for instance, you place a
pound of lead, a pound of chalk, and a pound of milk, in a hot
oven, they will be gradually heated to the temperature of the oven; but
the lead will attain it first, the chalk next, and the milk last.
CAROLINE.
That is a natural consequence of their different bulks; the lead
being the smallest body, will be heated soonest, and the milk, which is
the largest, will require the longest time.
MRS. B.
That explanation will not do, for if the lead be the least in bulk,
it offers also the least surface to the caloric, the quantity of heat
therefore which can enter into it in the same space of time is
proportionally smaller.
EMILY.
Why, then, do not the three bodies attain the temperature of the oven
at the same time?
124
MRS. B.
It is supposed to be on account of the different capacity of these
bodies for caloric.
CAROLINE.
What do you mean by the capacity of a body for caloric?
MRS. B.
I mean a certain disposition of bodies to require more or less
caloric for raising their temperature to any degree of heat. Perhaps the
fact may be thus explained:
Let us put as many marbles into this glass as it will contain, and
pour some sand over them—observe how the sand penetrates and
lodges between them. We shall now fill another glass with pebbles of
various forms—you see that they arrange themselves in a more
compact manner than the marbles, which, being globular, can touch each
other by a single point only. The pebbles, therefore, will not admit so
much sand between them; and consequently one of these glasses will
necessarily contain more sand than the other, though both of them be
equally full.
CAROLINE.
This I understand perfectly. The marbles and the pebbles represent
two bodies of different kinds, and the sand the caloric contained in
them;
125
it appears very plain, from this comparison, that one body may admit of
more caloric between its particles than another.
MRS. B.
You can no longer be surprised, therefore, that bodies of a different
capacity for caloric should require different proportions of that fluid
to raise their temperatures equally.
EMILY.
But I do not conceive why the body that contains the most caloric
should not be of the highest temperature; that is to say, feel hot in
proportion to the quantity of caloric it contains?
MRS. B.
The caloric that is employed in filling the capacity of a body, is
not free caloric; but is imprisoned as it were in the body, and is
therefore imperceptible: for we can feel only the caloric which the body
parts with, and not that which it retains.
CAROLINE.
It appears to me very extraordinary that heat should be confined in a
body in such a manner as to be imperceptible.
126
MRS. B.
If you lay your hand on a hot body, you feel only the caloric which
leaves it, and enters your hand; for it is impossible that you should be
sensible of that which remains in the body. The thermometer, in the same
manner, is affected only by the free caloric which a body transmits to
it, and not at all by that which it does not part with.
CAROLINE.
I begin to understand it: but I confess that the idea of insensible
heat is so new and strange to me, that it requires some time to render
it familiar.
MRS. B.
Call it insensible caloric, and the difficulty will appear much less
formidable. It is indeed a sort of contradiction to call it heat, when
it is so situated as to be incapable of producing that sensation. Yet
this modification of caloric is commonly called SPECIFIC HEAT.
CAROLINE.
But it certainly would have been more correct to have called it
specific caloric.
EMILY.
I do not understand how the term specific applies to this
modification of caloric?
127
MRS. B.
It expresses the relative quantity of caloric which different
species of bodies of the same weight and temperature are capable
of containing. This modification is also frequently called heat of
capacity, a term perhaps preferable, as it explains better its
own meaning.
You now understand, I suppose, why the milk and chalk required a
longer portion of time than the lead to raise their temperature to that
of the oven?
EMILY.
Yes: the milk and chalk having a greater capacity for caloric than
the lead, a greater proportion of that fluid became insensible in
those bodies: and the more slowly, therefore, their temperature was
raised.
CAROLINE.
But might not this difference proceed from the different conducting
powers of heat in these three bodies, since that which is the best
conductor must necessarily attain the temperature of the oven first?
MRS. B.
Very well observed, Caroline. This objection would be insurmountable,
if we could not, by reversing the experiment, prove that the milk, the
chalk, and the lead, actually absorbed different
128
quantities of caloric, and we know that if the different time they took
in heating, proceeded merely from their different conducting powers,
they would each have acquired an equal quantity of caloric.
CAROLINE.
Certainly. But how can you reverse this experiment?
MRS. B.
It may be done by cooling the several bodies to the same degree in an
apparatus adapted to receive and measure the caloric which they give
out. Thus, if you plunge them into three equal quantities of water, each
at the same temperature, you will be able to judge of the relative
quantity of caloric which the three bodies contained, by that, which, in
cooling, they communicated to their respective portions of water: for
the same quantity of caloric which they each absorbed to raise their
temperature, will abandon them in lowering it; and on examining the
three vessels of water, you will find the one in which you immersed the
lead to be the least heated; that which held the chalk will be the next;
and that which contained the milk will be heated the most of all. The
celebrated Lavoisier has invented a machine to estimate, upon this
principle, the specific heat of bodies in a more perfect manner; but I
cannot
129
explain it to you, till you are acquainted with the next modification of
caloric.
EMILY.
The more dense a body is, I suppose, the less is its capacity for
caloric?
MRS. B.
This is not always the case with bodies of different nature; iron,
for instance, contains more specific heat than tin, though it is more
dense. This seems to show that specific heat does hot merely depend upon
the interstices between the particles; but, probably, also upon some
peculiar constitution of the bodies which we do not comprehend.
EMILY.
But, Mrs. B., it would appear to me more proper to compare bodies by
measure, rather than by weight, in order to estimate their
specific heat. Why, for instance, should we not compare pints of
milk, of chalk, and of lead, rather than pounds of those
substances; for equal weights may be composed of very different
quantities?
MRS. B.
You are mistaken, my dear; equal weight must contain equal quantities
of matter; and when we wish to know what is the relative quantity of
caloric,
130
which substances of various kinds are capable of containing under the
same temperature, we must compare equal weights, and not equal bulks of
those substances. Bodies of the same weight may undoubtedly be of very
different dimensions; but that does not change their real quantity of
matter. A pound of feathers does not contain one atom more than a
pound of lead.
CAROLINE.
I have another difficulty to propose. It appears to me, that if the
temperature of the three bodies in the oven did not rise equally, they
would never reach the same degree; the lead would always keep its
advantage over the chalk and milk, and would perhaps be boiling before
the others had attained the temperature of the oven. I think you
might as well say that, in the course of time, you and I should be of
the same age?
MRS. B.
Your comparison is not correct, Caroline. As soon as the lead reached
the temperature of the oven, it would remain stationary; for it would
then give out as much heat as it would receive. You should recollect
that the exchange of radiating heat, between two bodies of equal
temperature, is equal: it would be impossible, therefore, for the lead
to accumulate heat after having attained
131
the temperature of the oven; and that of the chalk and milk therefore
would ultimately arrive at the same standard. Now I fear that this will
not hold good with respect to our ages, and that, as long as I live,
I shall never cease to keep my advantage over you.
EMILY.
I think that I have found a comparison for specific heat, which is
very applicable. Suppose that two men of equal weight and bulk, but who
required different quantities of food to satisfy their appetites, sit
down to dinner, both equally hungry; the one would consume a much
greater quantity of provisions than the other, in order to be equally
satisfied.
MRS. B.
Yes, that is very fair; for the quantity of food necessary to satisfy
their respective appetites, varies in the same manner as the quantity of
caloric requisite to raise equally the temperature of different
bodies.
EMILY.
The thermometer, then, affords no indication of the specific heat of
bodies?
MRS. B.
None at all: no more than satiety is a test of the quantity of food
eaten. The thermometer, as
132
I have repeatedly said, can be affected only by free caloric, which
alone raises the temperature of bodies.
But there is another mode of proving the existence of specific heat,
which affords a very satisfactory illustration of that modification.
This, however, I did not enlarge upon before, as I thought it might
appear to you rather complicated.—If you mix two fluids of
different temperatures, let us say the one at 50 degrees, and the other
at 100 degrees, of what temperature do you suppose the mixture
will be?
CAROLINE.
It will be no doubt the medium between the two, that is to say, 75
degrees.
MRS. B.
That will be the case if the two bodies happen to have the same
capacity for caloric; but if not, a different result will be
obtained. Thus, for instance, if you mix together a pound of mercury,
heated at 50 degrees, and a pound of water heated at 100 degrees, the
temperature of the mixture, instead of being 75 degrees, will be 80 degrees; so that the
water will have lost only 12 degrees, whilst the mercury will have
gained 38 degrees; from which you will conclude that the capacity of
mercury for heat is less than that of water.
133
CAROLINE.
I wonder that mercury should have so little specific heat. Did we not
see it was a much better conductor of heat than water?
MRS. B.
And it is precisely on that account that its specific heat is less.
For since the conductive power of bodies depends, as we have observed
before, on their readiness to receive heat and part with it, it is
natural to expect that those bodies which are the worst conductors
should absorb the most caloric before they are disposed to part with it
to other bodies. But let us now proceed to LATENT HEAT.
CAROLINE.
And pray what kind of heat is that?
MRS. B.
It is another modification of combined caloric, which is so analogous
to specific heat, that most chemists make no distinction between them;
but Mr. Pictet, in his Essay on Fire, has so clearly discriminated them,
that I am induced to adopt his view of the subject. We therefore call
latent heat that portion of insensible caloric which is employed
in changing the state of bodies; that is to say, in converting solids
into liquids, or liquids; into vapour. When a body changes its state
from
134
solid to liquid, or from liquid to vapour, its expansion occasions a
sudden and considerable increase of capacity for heat, in consequence of
which it immediately absorbs a quantity of caloric, which becomes fixed
in the body which it has transformed; and, as it is perfectly concealed
from our senses, it has obtained the name of latent heat.
CAROLINE.
I think it would be much more correct to call this modification
latent caloric instead of latent heat, since it does not excite the
sensation of heat.
MRS. B.
This modification of heat was discovered and named by Dr. Black long
before the French chemists introduced the term caloric, and we must not
presume to alter it, as it is still used by much better chemists than
ourselves. And, besides, you are not to suppose that the nature of heat
is altered by being variously modified: for if latent heat and specific
heat do not excite the same sensations as free caloric, it is owing to
their being in a state of confinement, which prevents them from acting
upon our organs; and consequently, as soon as they are extricated from
the body in which they are imprisoned, they return to their state of
free caloric.
135
EMILY.
But I do not yet clearly see in what respect latent heat differs from
specific heat; for they are both of them imprisoned and concealed in
bodies.
MRS. B.
Specific heat is that which is employed in filling the capacity of a
body for caloric, in the state in which this body actually exists; while
latent heat is that which is employed only in effecting a change of
state, that is, in converting bodies from a solid to a liquid, or from a
liquid to an aëriform state. But I think that, in a general point of
view, both these modifications might be comprehended under the name of
heat of capacity, as in both cases the caloric is equally engaged
in filling the capacities of bodies.
I shall now show you an experiment, which I hope will give you a
clear idea of what is understood by latent heat.
The snow which you see in this phial has been cooled by certain
chemical means (which I cannot well explain to you at present), to 5 or
6 degrees below the freezing point, as you will find indicated by the
thermometer which is placed in it. We shall expose it to the heat of a
lamp, and you will see the thermometer gradually rise, till it reaches
the freezing point——
136
EMILY.
But there it stops, Mrs. B., and yet the lamp burns just as well as
before. Why is not its heat communicated to the thermometer?
CAROLINE.
And the snow begins to melt, therefore it must be rising above the
freezing point?
MRS. B.
The heat no longer affects the thermometer, because it is wholly
employed in converting the ice into water. As the ice melts, the caloric
becomes latent in the new-formed liquid, and therefore cannot
raise its temperature; and the thermometer will consequently remain
stationary, till the whole of the ice be melted.
CAROLINE.
Now it is all melted, and the thermometer begins to rise again.
MRS. B.
Because the conversion of the ice into water being completed, the
caloric no longer becomes latent; and therefore the heat which the water
now receives raises its temperature, as you find the thermometer
indicates.
137
EMILY.
But I do not think that the thermometer rises so quickly in the water
as it did in the ice, previous to its beginning to melt, though the lamp
burns equally well?
MRS. B.
That is owing to the different specific heat of ice and water. The
capacity of water for caloric being greater than that of ice, more heat
is required to raise its temperature, and therefore the thermometer
rises slower in the water than in the ice.
EMILY.
True; you said that a solid body always increased its capacity for
heat by becoming fluid; and this is an instance of it.
MRS. B.
Yes, and the latent heat is that which is absorbed in consequence of
the greater capacity which the water has for heat, in comparison to
ice.
I must now tell you a curious calculation founded on that
consideration. I have before observed to you that though the
thermometer shows us the comparative warmth of bodies, and enables us to
determine the same point at different times and places, it gives us no
idea of the absolute quantity of heat in any body. We cannot tell how
low it ought to fall by the privation of all heat, but an
138
attempt has been made to infer it in the following manner. It has been
found by experiment, that the capacity of water for heat, when compared
with that of ice, is as 10 to 9, so that, at the same temperature, ice
contains one tenth of caloric less than water. By experiment also it is
observed, that in order to melt ice, there must be added to it as much
heat, as would, if it did not melt it, raise its temperature 140
degrees. This quantity of heat is therefore absorbed when the ice, by
being converted into water, is made to contain one-ninth more caloric
than it did before. Therefore 140 degrees is a ninth part of the heat
contained in ice at 30 degrees; and the point of zero, or the absolute
privation of heat, must consequently be 1260 degrees below 32
degrees.
This mode of investigating so curious a question is ingenious, but
its correctness is not yet established by similar calculations for other
bodies. The points of absolute cold, indicated by this method in various
bodies, are very remote from each other; it is however possible, that
this may arise from some imperfection in the experiments.
CAROLINE.
It is indeed very ingenious—but we must now attend to our
present experiment. The water begins to boil, and the thermometer is
again stationary.
139
MRS. B.
Well, Caroline, it is your turn to explain the phenomenon.
CAROLINE.
It is wonderfully curious! The caloric is now busy in changing the
water into steam, in which it hides itself, and becomes insensible. This
is another example of latent heat, producing a change of form. At first
it converted a solid body into a liquid, and now it turns the liquid
into vapour!
MRS. B.
You see, my dear, how easily you have become acquainted with these
modifications of insensible heat, which at first appeared so
unintelligible. If, now, we were to reverse these changes, and condense
the vapour into water, and the water into ice, the latent heat would
re-appear entirely, in the form of free caloric.
EMILY.
Pray do let us see the effect of latent heat returning to its free
state.
MRS. B.
For the purpose of showing this, we need simply conduct the vapour
through this tube into this vessel of cold water, where it will part
with its latent heat and return to its liquid form.
140
EMILY.
How rapidly the steam heats the water!
MRS. B.
That is because it does not merely impart its free caloric to the
water, but likewise its latent heat. This method of heating liquids, has
been turned to advantage, in several economical establishments. The
steam-kitchens, which are getting into such general use, are upon the
same principle. The steam is conveyed through a pipe in a similar
manner, into the several vessels which contain the provisions to be
dressed, where it communicates to them its latent caloric, and returns
to the state of water. Count Rumford makes great use of this principle
in many of his fire-places: his grand maxim is to avoid all unnecessary
waste of caloric, for which purpose he confines the heat in such a
manner, that not a particle of it shall unnecessarily escape; and while
he economises the free caloric, he takes care also to turn the latent
heat to advantage. It is thus that he is enabled to produce a degree of
heat superior to that which is obtained in common fire-places, though he
employs less fuel.
EMILY.
When the advantages of such contrivances are so clear and plain,
I cannot understand why they are not universally used.
141
MRS. B.
A long time is always required before innovations, however useful,
can be reconciled with the prejudices of the vulgar.
EMILY.
What a pity it is that there should be a prejudice against new
inventions; how much more rapidly the world would improve, if such
useful discoveries were immediately and universally adopted!
MRS. B.
I believe, my dear, that there are as many novelties attempted to be
introduced, the adoption of which would be prejudicial to society, as
there are of those which would be beneficial to it. The well-informed,
though by no means exempt from error, have an unquestionable advantage
over the illiterate, in judging what is likely or not to prove
serviceable; and therefore we find the former more ready to adopt such
discoveries as promise to be really advantageous, than the latter, who
having no other test of the value of a novelty but time and experience,
at first oppose its introduction. The well-informed, however, are
frequently disappointed in their most sanguine expectations, and the
prejudices of the vulgar, though they often retard the progress of
knowledge, yet sometimes, it must be
142
admitted, prevent the propagation of error.—But we are deviating
from our subject.
We have converted steam into water, and are now to change water into
ice, in order to render the latent heat sensible, as it escapes from the
water on its becoming solid. For this purpose we must produce a degree
of cold that will make water freeze.
CAROLINE.
That must be very difficult to accomplish in this warm room.
MRS. B.
Not so much as you think. There are certain chemical mixtures which
produce a rapid change from the solid to the fluid state, or the
reverse, in the substances combined, in consequence of which change
latent heat is either extricated or absorbed.
EMILY.
I do not quite understand you.
MRS. B.
This snow and salt, which you see me mix together, are melting
rapidly; heat, therefore, must be absorbed by the mixture, and cold
produced.
CAROLINE.
It feels even colder than ice, and yet the snow is melted. This is
very extraordinary.
143
MRS. B.
The cause of the intense cold of the mixture is to be attributed to
the change from a solid to a fluid state. The union of the snow and salt
produces a new arrangement of their particles, in consequence of which
they become liquid; and the quantity of caloric, required to effect this
change, is seized upon by the mixture wherever it can be obtained. This
eagerness of the mixture for caloric, during its liquefaction, is such,
that it converts part of its own free caloric into latent heat, and it
is thus that its temperature is lowered.
EMILY.
Whatever you put in this mixture, therefore, would freeze?
MRS. B.
Yes; at least any fluid that is susceptible of freezing at that
temperature. I have prepared this mixture of salt and snow for the
purpose of freezing the water from which you are desirous of seeing the
latent heat escape. I have put a thermometer in the glass of water
that is to be frozen, in order that you may see how it cools.
CAROLINE.
The thermometer descends, but the heat which the water is now losing,
is its free, not its latent heat.
144
MRS. B.
Certainly; it does not part with its latent heat till it changes its
state and is converted into ice.
EMILY.
But here is a very extraordinary circumstance! The thermometer is
fallen below the freezing point, and yet the water is not frozen.
MRS. B.
That is always the case previous to the freezing of water when it is
in a state of rest. Now it begins to congeal, and you may observe that
the thermometer again rises to the freezing point.
CAROLINE.
It appears to me very strange that the thermometer should rise the
very moment that the water freezes; for it seems to imply that the water
was colder before it froze than when in the act of freezing.
MRS. B.
It is so; and after our long dissertation on this circumstance,
I did not think it would appear so surprising to you. Reflect a
little, and I think you will discover the reason of it.
CAROLINE.
It must be, no doubt, the extrications of latent heat, at the instant
the water freezes, that raises the temperature.
145
MRS. B.
Certainly; and if you now examine the thermometer, you will find that
its rise was but temporary, and lasted only during the disengagement of
the latent heat—now that all the water is frozen it falls again,
and will continue to fall till the ice and mixture are of an equal
temperature.
EMILY.
And can you show us any experiments in which liquids, by being mixed,
become solid, and disengage latent heat?
MRS. B.
I could show you several; but you are not yet sufficiently advanced
to understand them well. I shall, however, try one, which will
afford you a striking instance of the fact. The fluid which you see in
this phial consists of a quantity of a certain salt called muriat of
lime, dissolved in water. Now, if I pour into it a few drops of this
other fluid, called sulphuric acid, the whole, or very nearly the
whole, will be instantaneously converted into a solid mass.
EMILY.
How white it turns! I feel the latent heat escaping, for the bottle
is warm, and the fluid is changed to a solid white substance like
chalk!
146
CAROLINE.
This is, indeed, the most curious experiment we have seen yet. But
pray what is that white vapour that ascends from the mixture?
MRS. B.
You are not yet enough of a chemist to understand that.—But
take care, Caroline, do not approach too near it, for it has a very
pungent smell.
I shall show you another instance similar to that of the water, which
you observed to become warmer as it froze. I have in this phial a
solution of a salt called sulphat of soda or Glauber’s salt, made very
strong, and corked up when it was hot, and kept without agitation till
it became cold, as you may feel the phial is. Now when I take out the
cork and let the air fall upon it, (for being closed when boiling, there
was a vacuum in the upper part) observe that the salt will suddenly
crystallize. . . .
CAROLINE.
Surprising! how beautifully the needles of salt have shot through the
whole phial!
MRS. B.
Yes, it is very striking—but pray do not forget the object of
the experiment. Feel how warm the phial has become by the conversion of
part of the liquid into a solid.
147
EMILY.
Quite warm I declare! this is a most curious experiment of the
disengagement of latent heat.
MRS. B.
The slakeing of lime is another remarkable instance of the
extrication of latent heat. Have you never observed how quick-lime
smokes when water is poured upon it, and how much heat it produces?
CAROLINE.
Yes; but I do not understand what change of state takes place in the
lime that occasions its giving out latent heat; for the quick-lime,
which is solid, is (if I recollect right) reduced to powder, by this
operation, and is, therefore, rather expanded than condensed.
MRS. B.
It is from the water, not the lime, that the latent heat is set free.
The water incorporates with, and becomes solid in the lime; in
consequence of which, the heat, which kept it in a liquid state, is
disengaged, and escapes in a sensible form.
CAROLINE.
I always thought that the heat originated in the lime. It seems very
strange that water, and cold water too, should contain so much heat.
148
EMILY.
After this extrication of caloric, the water must exist in a state of
ice in the lime, since it parts with the heat which kept it liquid.
MRS. B.
It cannot properly be called ice, since ice implies a degree of cold,
at least equal to the freezing point. Yet as water, in combining with
lime, gives out more heat than in freezing, it must be in a state of
still greater solidity in the lime, than it is in the form of ice; and
you may have observed that it does not moisten or liquefy the lime in
the smallest degree.
EMILY.
But, Mrs. B., the smoke that rises is white; if it was only pure
caloric which escaped, we might feel, but could not see it.
MRS. B.
This white vapour is formed by some of the particles of lime, in a
state of fine dust, which are carried off by the caloric.
EMILY.
In all changes of state, then, a body either absorbs or disengages
latent heat?
149
MRS. B.
You cannot exactly say absorbs latent heat, as the heat
becomes latent only on being confined in the body; but you may say,
generally, that bodies, in passing from a solid to a liquid form, or
from the liquid state to that of vapour, absorb heat; and that when the
reverse takes place, heat is disengaged.*
EMILY.
We can now, I think, account for the ether boiling, and the water
freezing in vacuo, at the same temperature.†
MRS. B.
Let me hear how you explain it.
EMILY.
The latent heat, which the water gave out in freezing, was
immediately absorbed by the ether, during its conversion into vapour;
and therefore, from a latent state in one liquid, it passed into a
latent state in the other.
MRS. B.
But this only partly accounts for the result of the experiment; it
remains to be explained why the
150
temperature of the ether, while in a state of ebullition, is brought
down to the freezing temperature of the water.—It is because the
ether, during its evaporation, reduces its own temperature, in the same
proportion as that of the water, by converting its free caloric into
latent heat: so that, though one liquid boils, and the other freezes,
their temperatures remain in a state of equilibrium.
EMILY.
But why does not water, as well as ether, reduce its own temperature
by evaporating?
MRS. B.
The fact is that it does, though much less rapidly than ether. Thus,
for instance, you may often have observed, in the heat of summer, how
much any particular spot may be cooled by watering, though the water
used for that purpose be as warm as the air itself. Indeed so much cold
may be produced by the mere evaporation of water, that the inhabitants
of India, by availing themselves of the most favourable circumstances
for this process which their warm climate can afford, namely, the cool
of the night, and situations most exposed to the night breeze, succeed
in causing water to freeze, though the temperature of the air be as high
as 60 degrees. The water is put into shallow earthen trays, so as to
expose an extensive
151
surface to the process of evaporation, and in the morning, the water is
found covered with a thin cake of ice, which is collected in sufficient
quantity to be used for purposes of luxury.
CAROLINE.
How delicious it must be to drink liquids so cold in those tropical
climates! But, Mrs. B., could we not try that experiment?
MRS. B.
If we were in the country, I have no doubt but that we should be able
to freeze water, by the same means, and under similar circumstances. But
we can do it immediately, upon a small scale, in this very room, in
which the thermometer stands at 70 degrees. For this purpose we need
only place some water in a little cup under the receiver of the air-pump
(Plate V. fig. 1.), and
exhaust the air from it. What will be the consequence, Caroline?
Plate V.
Vol. I. page 138.
Fig. 1.
The air-pump & receiver for Mr. Leslie’s experiment.
C a saucer with sulphuric Acid.
B a glass or earthen cup containing Water.
D a stand for the cup with its legs made of Glass.
A a Thermometer.
Larger view
(complete Plate)
CAROLINE.
Of course the water will evaporate more quickly, since there will no
longer be any atmospheric pressure on its surface: but will this be
sufficient to make the water freeze?
152
MRS. B.
Probably not, because the vapour will not be carried off fast enough;
but this will be accomplished without difficulty if we introduce into
the receiver (fig. 1.), in a saucer, or other large shallow vessel,
some strong sulphuric acid, a substance which has a great
attraction for water, whether in the form of vapour, or in the liquid
state. This attraction is such that the acid will instantly absorb the
moisture as it rises from the water, so as to make room for the
formation of fresh vapour; this will of course hasten the process, and
the cold produced from the rapid evaporation of the water, will, in a
few minutes, be sufficient to freeze its surface.* We shall now exhaust the air
from the receiver.
EMILY.
Thousands of small bubbles already rise through the water from the
internal surface of the cup; what is the reason of this?
MRS. B.
These are bubbles of air which were partly attached to the vessel,
and partly diffused in the water itself; and they expand and rise in
consequence of the atmospheric pressure being removed.
153
CAROLINE.
See, Mrs. B.; the thermometer in the cup is sinking fast; it has
already descended to 40 degrees!
EMILY.
The water seems now and then violently agitated on the surface, as if
it was boiling; and yet the thermometer is descending fast!
MRS. B.
You may call it boiling, if you please, for this appearance
is, as well as boiling, owing to the rapid formation of vapour; but
here, as you have just observed, it takes place from the surface, for it
is only when heat is applied to the bottom of the vessel that the vapour
is formed there.—Now crystals of ice are actually shooting all
over the surface of the water.
CAROLINE.
How beautiful it is! The surface is now entirely frozen—but the
thermometer remains at 32 degrees.
MRS. B.
And so it will, conformably with our doctrine of latent heat, until
the whole of the water is frozen; but it will then again begin to
descend lower and lower, in consequence of the evaporation which goes on
from the surface of the ice.
154
EMILY.
This is a most interesting experiment; but it would be still more
striking if no sulphuric acid were required.
MRS. B.
I will show you a freezing instrument, contrived by Dr. Wollaston,
upon the same principle as Mr. Leslie’s experiment, by which water may
be frozen by its own evaporation alone, without the assistance of
sulphuric acid.
This tube, which, as you see (Plate V. fig. 2.), is terminated at each extremity
by a bulb, one of which is half full of water, is internally perfectly
exhausted of air; the consequence of this is, that the water in the bulb
is always much disposed to evaporate. This evaporation, however, does
not proceed sufficiently fast to freeze the water; but if the empty ball
be cooled by some artificial means, so as to condense quickly the vapour
which rises from the water, the process may be thus so much promoted as
to cause the water to freeze in the other ball. Dr. Wollaston has called
this instrument Cryophorus.
Plate V.
Vol. I. page 138.
Fig. 2. Dr. Wollaston’s Cryophorus.
Fig. 5. Dr. Marcet’s mode of using the Cryophorus.
Fig. 3. & 4. the different parts of Fig. 5. seen
separate.
Larger view
(complete Plate)
CAROLINE.
So that cold seems to perform here the same part which the sulphuric
acid acted in Mr. Leslie’s experiment?
155
MRS. B.
Exactly so; but let us try the experiment.
EMILY.
How will you cool the instrument? You have neither ice nor snow.
MRS. B.
True: but we have other means of effecting this.* You recollect what an
intense cold can be produced by the evaporation of ether in an exhausted
receiver. We shall inclose the bulb in this little bag of fine flannel
(fig. 3.), then soke it in ether, and introduce it into the
receiver of the air-pump. (Fig. 5.) For this purpose we shall find
it more convenient to use a cryophorus of this shape (fig. 4.), as
its elongated bulb passes easily through a brass plate which closes the
top of the receiver. If we now exhaust the receiver quickly, you will
see, in less than a minute, the water freeze in the other bulb, out of
the receiver.
EMILY.
The bulb already looks quite dim, and small drops of water are
condensing on its surface.
156
CAROLINE.
And now crystals of ice shoot all over the water. This is, indeed,
a very curious experiment!
MRS. B.
You will see, some other day, that, by a similar method, even
quicksilver may be frozen.—But we cannot at present indulge in any
further digression.
Having advanced so far on the subject of heat, I may now give
you an account of the calorimeter, an instrument invented by Lavoisier,
upon the principles just explained, for the purpose of estimating the
specific heat of bodies. It consists of a vessel, the inner surface of
which is lined with ice, so as to form a sort of hollow globe of ice, in
the midst of which the body, whose specific heat is to be ascertained,
is placed. The ice absorbs caloric from this body, till it has brought
it down to the freezing point; this caloric converts into water a
certain portion of the ice which runs out through an aperture at the
bottom of the machine; and the quantity of ice changed to water is a
test of the quantity of caloric which the body has given out in
descending from a certain temperature to the freezing point.
CAROLINE.
In this apparatus, I suppose, the milk, chalk,
157
and lead, would melt different quantities of ice, in proportion to their
different capacities for caloric?
MRS. B.
Certainly: and thence we are able to ascertain, with precision, their
respective capacities for heat. But the calorimeter affords us no more
idea of the absolute quantity of heat contained in a body, than the
thermometer; for though by means of it we extricate both the free and
combined caloric, yet we extricate them only to a certain degree, which
is the freezing point; and we know not how much they contain of either
below that point.
EMILY.
According to the theory of latent heat, it appears to me that the
weather should be warm when it freezes, and cold in a thaw: for latent
heat is liberated from every substance that it freezes, and such a large
supply of heat must warm the atmosphere; whilst, during a thaw, that
very quantity of free heat must be taken from the atmosphere, and return
to a latent state in the bodies which it thaws.
MRS. B.
Your observation is very natural; but consider that in a frost the
atmosphere is so much colder than the earth, that all the caloric which
it takes
158
from the freezing bodies is insufficient to raise its temperature above
the freezing point; otherwise the frost must cease. But if the quantity
of latent heat extricated does not destroy the frost, it serves to
moderate the suddenness of the change of temperature of the atmosphere,
at the commencement both of frost, and of a thaw. In the first instance,
its extrication diminishes the severity of the cold; and, in the latter,
its absorption moderates the warmth occasioned by a thaw: it even
sometimes produces a discernible chill, at the breaking up of a
frost.
CAROLINE.
But what are the general causes that produce those sudden changes in
the weather, especially from hot to cold, which we often experience?
MRS. B.
This question would lead us into meteorological discussions, to which
I am by no means competent. One circumstance, however, we can easily
understand. When the air has passed over cold countries, it will
probably arrive here at a temperature much below our own, and then it
must absorb heat from every object it meets with, which will produce a
general fall of temperature.
CAROLINE.
But pray, now that we know so much of the
159
effects of heat, will you inform us whether it is really a distinct
body, or, as I have heard, a peculiar kind of motion produced in
bodies?
MRS. B.
As I before told you, there is yet much uncertainty as to the nature
of these subtle agents. But I am inclined to consider heat not as mere
motion, but as a separate substance. Late experiments too appear to make
it a compound body, consisting of the two electricities, and in our next
conversation I shall inform you of the principal facts on which that
opinion is founded.
160
CONVERSATION V.
ON THE CHEMICAL AGENCIES OF
ELECTRICITY.

MRS. B.
Before we proceed further it will be
necessary to give you some account of certain properties of electricity,
which have of late years been discovered to have an essential connection
with the phenomena of chemistry.
CAROLINE.
It is ELECTRICITY, if I recollect
right, which comes next in our list of simple substances?
MRS. B.
I have placed electricity in that list, rather from the necessity of
classing it somewhere, than from any conviction that it has a right to
that situation, for we are as yet so ignorant of its intimate nature,
that we are unable to determine, not only whether it is simple or
compound, but whether it is in fact a material agent; or, as Sir H. Davy
has hinted, whether it may not be merely a property inherent
161
in matter. As, however, it is necessary to adopt some hypothesis for the
explanation of the discoveries which this agent has enabled us to make,
I have chosen the opinion, at present most prevalent, which
supposes the existence of two kinds of electricity, distinguished by the
names of positive and negative electricity.
CAROLINE.
Well, I must confess, I do not feel nearly so interested in a
science in which so much uncertainty prevails, as in those which rest
upon established principles; I never was fond of electricity,
because, however beautiful and curious the phenomena it exhibits may be,
the theories, by which they were explained, appeared to me so various,
so obscure and inadequate, that I always remained dissatisfied.
I was in hopes that the new discoveries in electricity had thrown
so great a light on the subject, that every thing respecting it would
now have been clearly explained.
MRS. B.
That is a point which we are yet far from having attained. But, in
spite of the imperfection of our theories, you will be amply repaid by
the importance and novelty of the subject. The number of new facts which
have already been ascertained, and the immense prospect of discovery
162
which has lately been opened to us, will, I hope, ultimately lead
to a perfect elucidation of this branch of natural science; but at
present you must be contented with studying the effects, and in some
degree explaining the phenomena, without aspiring to a precise knowledge
of the remote cause of electricity.
You have already obtained some notions of electricity: in our present
conversation, therefore, I shall confine myself to that part of the
science which is of late discovery, and is more particularly connected
with chemistry.
It was a trifling and accidental circumstance which first gave rise
to this new branch of physical science. Galvani, a professor of
natural philosophy at Bologna, being engaged (about twenty years ago) in
some experiments on muscular irritability, observed, that when a piece
of metal was laid on the nerve of a frog, recently dead, whilst the limb
supplied by that nerve rested upon some other metal, the limb suddenly
moved, on a communication being made between the two pieces of
metal.
EMILY.
How is this communication made?
MRS. B.
Either by bringing the two metals into contact, or by connecting them
by means of a metallic conductor.
163
But without subjecting a frog to any cruel experiments, I can
easily make you sensible of this kind of electric action. Here is a
piece of zinc, (one of the metals I mentioned in the list of elementary
bodies)—put it under your tongue, and this piece of silver
upon your tongue, and let both the metals project a little beyond
the tip of the tongue—very well—now make the projecting
parts of the metals touch each other, and you will instantly perceive a
peculiar sensation.
EMILY.
Indeed I did, a singular taste, and I think a degree of heat: but I
can hardly describe it.
MRS. B.
The action of these two pieces of metal on the tongue is,
I believe, precisely similar to that made on the nerve of a frog.
I shall not detain you by a detailed account of the theory by which
Galvani attempted to account for this fact, as his explanation was soon
overturned by subsequent experiments, which proved that Galvanism
(the name this new power had obtained) was nothing more than
electricity. Galvani supposed that the virtue of this new agent resided
in the nerves of the frog, but Volta, who prosecuted this subject with
much greater success, shewed that the phenomena did not depend on the
organs of the frog, but upon
164
the electrical agency of the metals, which is excited by the moisture of
the animal, the organs of the frog being only a delicate test of the
presence of electric influence.
CAROLINE.
I suppose, then, the saliva of the mouth answers the same purpose as
the moisture of the frog, in exciting the electricity of the pieces of
silver and zinc with which Emily tried the experiment on her tongue.
MRS. B.
Precisely. It does not appear, however, necessary that the fluid used
for this purpose should be of an animal nature. Water, and acids very
much diluted by water, are found to be the most effectual in promoting
the developement of electricity in metals; and, accordingly, the
original apparatus which Volta first constructed for this purpose,
consisted of a pile or succession of plates of zinc and copper, each
pair of which was connected by pieces of cloth or paper impregnated with
water; and this instrument, from its original inconvenient structure and
limited strength, has gradually arrived at its present state of power
and improvement, such as is exhibited in the Voltaic battery. In this
apparatus, a specimen of which you see before you (Plate VI. fig. 1.), the plates of
zinc and copper are soldered together in pairs, each pair being placed
at regular
165
distances in wooden troughs
and the interstices being filled with fluid.
Plate VI.
p. 151.
Fig. 1. 2. & 4. Voltaic Batteries
Larger view
(complete Plate)
CAROLINE.
Though you will not allow us to enquire into the precise cause of
electricity, may we not ask in what manner the fluid acts on the metals
so as to produce it?
MRS. B.
The action of the fluid on the metals, whether water or acid be used,
is entirely of a chemical nature. But whether electricity is excited by
this chemical action, or whether it is produced by the contact of the
two metals, is a point upon which philosophers do not yet perfectly
agree.
EMILY.
But can the mere contact of two metals, without any intervening
fluid, produce electricity?
MRS. B.
Yes, if they are afterwards separated. It is an established fact,
that when two metals are put in contact, and afterwards separated, that
which has the strongest attraction for oxygen exhibits signs of
positive, the other of negative electricity.
CAROLINE.
It seems then but reasonable to infer that the
166
power of the Voltaic battery should arise from the contact of the plates
of zinc and copper.
MRS. B.
It is upon this principle that Volta and Sir H. Davy explain the
phenomena of the pile; but notwithstanding these two great authorities,
many philosophers entertain doubts on the truth of this theory. The
principal difficulty which occurs in explaining the phenomena of the
Voltaic battery on this principle, is, that two such plates show no
signs of different states of electricity whilst in contact, but only on
being separated after contact. Now in the Voltaic battery, those plates
that are in contact always continue so, being soldered together: and
they cannot therefore receive a succession of charges. Besides, if we
consider the mere disturbance of the balance of electricity by the
contact of the plates, as the sole cause of the production of Voltaic
electricity, it remains to be explained how this disturbed balance
becomes an inexhaustible source of electrical energy, capable of pouring
forth a constant and copious supply of electrical fluid, though without
any means of replenishing itself from other sources. This subject, it
must be owned, is involved in too much obscurity to enable us to speak
very decidedly in favour of any theory. But, in order to avoid
perplexing you with different explanations,
167
I shall confine myself to one which appears to me to be least encumbered
with difficulties, and most likely to accord with truth.*
This theory supposes the electricity to be excited by the chemical
action of the acid on the zinc; but you are yet such novices in
chemistry, that I think it will be necessary to give you some previous
explanation of the nature of this action.
All metals have a strong attraction for oxygen, and this element is
found in great abundance both in water and in acids. The action of the
diluted acid on the zinc consists therefore in its oxygen combining with
it, and dissolving its surface.
CAROLINE.
In the same manner I suppose as we saw an acid dissolve copper?
MRS. B.
Yes; but in the Voltaic battery the diluted acid is not strong enough
to produce so complete
168
an effect; it acts only on the surface of the zinc, to which it yields
its oxygen, forming upon it a film or crust, which is a compound of the
oxygen and the metal.
EMILY.
Since there is so strong a chemical attraction between oxygen and
metals, I suppose they are naturally in different states of
electricity?
MRS. B.
Yes; it appears that all metals are united with the positive, and
that oxygen is the grand source of the negative electricity.
CAROLINE.
Does not then the acid act on the plates of copper, as well as on
those of zinc?
MRS. B.
No; for though copper has an affinity for oxygen, it is less strong
than that of zinc; and therefore the energy of the acid is only exerted
upon the zinc.
It will be best, I believe, in order to render the action of the
Voltaic battery more intelligible, to confine our attention at first to
the effect produced on two plates only. (Plate VI. fig. 2.)
If a plate of zinc be placed opposite to one of copper, or any other
metal less attractive of oxygen,
169
and the space between them (suppose of half an inch in thickness), be
filled with an acid or any fluid capable of oxydating the zinc, the
oxydated surface will have its capacity for electricity diminished, so
that a quantity of electricity will be evolved from that surface. This
electricity will be received by the contiguous fluid, by which it will
be transmitted to the opposite metallic surface, the copper, which is
not oxydated, and is therefore disposed to receive it; so that the
copper plate will thus become positive, whilst the zinc plate will be in
the negative state.
This evolution of electrical fluid however will be very limited; for
as these two plates admit of but very little accumulation of
electricity, and are supposed to have no communication with other
bodies, the action of the acid, and further developement of electricity,
will be immediately stopped.
EMILY.
This action, I suppose, can no more continue to go on, than that of a
common electrical machine, which is not allowed to communicate with
other bodies?
MRS. B.
Precisely; the common electrical machine, when excited by the
friction of the rubber, gives out both the positive and negative
electricities.—(Plate
VI. Fig. 3.) The positive, by the rotation
170
of the glass cylinder, is conveyed into the conductor, whilst the
negative goes into the rubber. But unless there is a communication made
between the rubber and the ground, but a very inconsiderable quantity of
electricity can be excited; for the rubber, like the plates of the
battery, has too small a capacity to admit of an accumulation of
electricity. Unless therefore the electricity can pass out of the
rubber, it will not continue to go into it, and consequently no
additional accumulation will take place. Now as one kind of electricity
cannot be given out without the other, the developement of the positive
electricity is stopped as well as that of the negative, and the
conductor therefore cannot receive a succession of charges.
Plate VI.
p. 151.
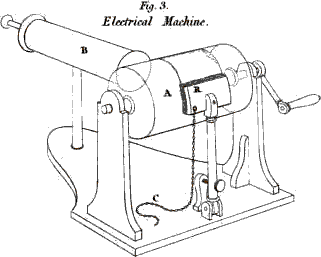
Fig. 3.
A the Cylinder.
B the Conductor.
R the Rubber.
C the Chain.
Larger view
(complete Plate)
CAROLINE.
But does not the conductor, as well as the rubber, require a
communication with the earth, in order to get rid of its
electricity?
MRS. B.
No; for it is susceptible of receiving and containing a considerable
quantity of electricity, as it is much larger than the rubber, and
therefore has a greater capacity; and this continued accumulation of
electricity in the conductor is what is called a charge.
171
EMILY.
But when an electrical machine is furnished with two conductors to
receive the two electricities, I suppose no communication with the
earth is required?
MRS. B.
Certainly not, until the two are fully charged; for the two
conductors will receive equal quantities of electricity.
CAROLINE.
I thought the use of the chain had been to convey the electricity
from the ground into the machine?
MRS. B.
That was the idea of Dr. Franklin, who supposed that there was but
one kind of electricity, and who, by the terms positive and negative
(which he first introduced), meant only different quantities of the same
kind of electricity. The chain was in that case supposed to convey
electricity from the ground through the rubber into the
conductor. But as we have adopted the hypothesis of two electricities,
we must consider the chain as a vehicle to conduct the negative
electricity into the earth.
EMILY.
And are both kinds of electricity produced whenever electricity is
excited?
172
MRS. B.
Yes, invariably. If you rub a tube of glass with a woollen cloth, the
glass becomes positive, and the cloth negative. If, on the contrary, you
excite a stick of sealing-wax by the same means, it is the rubber which
becomes positive, and the wax negative.
But with regard to the Voltaic battery, in order that the acid may
act freely on the zinc, and the two electricities be given out without
interruption, some method must be devised, by which the plates may part
with their electricities as fast as they receive them.—Can you
think of any means by which this might be effected?
EMILY.
Would not two chains or wires, suspended from either plate to the
ground, conduct the electricities into the earth, and thus answer the
purpose?
MRS. B.
It would answer the purpose of carrying off the electricity,
I admit; but recollect, that though it is necessary to find a vent
for the electricity, yet we must not lose it, since it is the power
which we are endeavouring to obtain. Instead, therefore, of conducting
it into the ground, let us make the wires, from either plate, meet: the
two electricities will thus be brought together, and will combine
173
and neutralize each other; and as long as this communication continues,
the two plates having a vent for their respective electricities, the
action of the acid will go on freely and uninterruptedly.
EMILY.
That is very clear, so far as two plates only are concerned; but I
cannot say I understand how the energy of the succession of plates, or
rather pairs of plates, of which the Galvanic trough is composed, is
propagated and accumulated throughout a battery?
MRS. B.
In order to shew you how the intensity of the electricity is
increased by increasing the number of plates, we will examine the action
of four plates; if you understand these, you will readily comprehend
that of any number whatever. In this figure (Plate VI. Fig. 4.), you will observe that the two
central plates are united; they are soldered together, (as we observed
in describing the Voltaic trough,) so as to form but one plate which
offers two different surfaces, the one of copper, the other of zinc.
Now you recollect that, in explaining the action of two plates, we
supposed that a quantity of electricity was evolved from the surface of
the first zinc plate, in consequence of the action of the acid, and was
conveyed by the interposed fluid to the copper
174
plate, No. 2, which thus became positive. This copper plate
communicates its electricity to the contiguous zinc plate, No. 3,
in which, consequently, some accumulation of electricity takes place.
When, therefore, the fluid in the next cell acts upon the zinc plate,
electricity is extricated from it in larger quantity, and in a more
concentrated form, than before. This concentrated electricity is again
conveyed by the fluid to the next pair of plates, No. 4 and 5, when
it is farther increased by the action of the fluid in the third cell,
and so on, to any number of plates of which the battery may consist; so
that the electrical energy will continue to accumulate in proportion to
the number of double plates, the first zinc plate of the series being
the most negative, and the last copper plate the most positive.
CAROLINE.
But does the battery become more and more strongly charged, merely by
being allowed to stand undisturbed?
MRS. B.
No, for the action will soon stop, as was explained before, unless a
vent be given to the accumulated electricities. This is easily done,
however, by establishing a communication by means of the wires
(Fig. 1.), between the two ends of the battery: these being brought
into contact, the two
175
electricities meet and neutralize each other, producing the shock and
other effects of electricity; and the action goes on with renewed
energy, being no longer obstructed by the accumulation of the two
electricities which impeded its progress.
EMILY.
Is it the union of the two electricities which produces the electric
spark?
MRS. B.
Yes; and it is, I believe, this circumstance which gave rise to Sir
H. Davy’s opinion that caloric may be a compound of the two
electricities.
CAROLINE.
Yet surely caloric is very different from the electrical spark?
MRS. B.
The difference may consist probably only in intensity: for the heat
of the electric spark is considerably more intense, though confined to a
very minute spot, than any heat we can produce by other means.
EMILY.
Is it quite certain that the electricity of the Voltaic battery is
precisely of the same nature as that of the common electrical
machine?
176
MRS. B.
Undoubtedly; the shock given to the human body, the spark, the
circumstance of the same substances which are conductors of the one
being also conductors of the other, and of those bodies, such as glass
and sealing-wax, which are non-conductors of the one, being also
non-conductors of the other, are striking proofs of it. Besides, Sir H.
Davy has shewn in his Lectures, that a Leyden jar, and a common electric
battery, can be charged with electricity obtained from a Voltaic
battery, the effect produced being perfectly similar to that obtained by
a common machine.
Dr. Wollaston has likewise proved that similar chemical
decompositions are effected by the electric machine and by the Voltaic
battery; and has made other experiments which render it highly probable,
that the origin of both electricities is essentially the same, as they
show that the rubber of the common electrical machine, like the zinc in
the Voltaic battery, produces the two electricities by combining with
oxygen.
CAROLINE.
But I do not see whence the rubber obtains oxygen, for there is
neither acid nor water used in the common machine, and I always
understood that the electricity was excited by the friction.
177
MRS. B.
It appears that by friction the rubber obtains oxygen from the
atmosphere, which is partly composed of that element. The oxygen
combines with the amalgam of the rubber, which is of a metallic nature,
much in the same way as the oxygen of the acid combines with the zinc in
the Voltaic battery, and it is thus that the two electricities are
disengaged.
CAROLINE.
But, if the electricities of both machines are similar, why not use
the common machine for chemical decompositions?
MRS. B.
Though its effects are similar to those of the Voltaic battery, they
are incomparably weaker. Indeed Dr. Wollaston, in using it for chemical
decompositions, was obliged to act upon the most minute quantities of
matter, and though the result was satisfactory in proving the similarity
of its effects to those of the Voltaic battery, these effects were too
small in extent to be in any considerable degree applicable to chemical
decomposition.
CAROLINE.
How terrible, then, the shock must be from a Voltaic battery, since
it is so much more powerful than an electrical machine!
178
MRS. B.
It is not nearly so formidable as you think; at least it is by no
means proportional to the chemical effect. The great superiority of the
Voltaic battery consists in the large quantity of electricity
that passes; but in regard to the rapidity or intensity of
the charge, it is greatly surpassed by the common electrical machine. It
would seem that the shock or sensation depends chiefly upon the
intensity; whilst, on the contrary, for chemical purposes, it is
quantity which is required. In the Voltaic battery, the electricity,
though copious, is so weak as not to be able to force its way through
the fluid which separates the plates, whilst that of a common machine
will pass through any space of water.
CAROLINE.
Would not it be possible to increase the intensity of the Voltaic
battery till it should equal that of the common machine?
MRS. B.
It can actually be increased till it imitates a weak electrical
machine, so as to produce a visible spark when accumulated in a Leyden
jar. But it can never be raised sufficiently to pass through any
considerable extent of air, because of the ready communication through
the fluids employed.
By increasing the number of plates of a battery,
179
you increase its intensity, whilst, by enlarging the dimensions
of the plates, you augment its quantity; and, as the superiority
of the battery over the common machine consists entirely in the quantity
of electricity produced, it was at first supposed that it was the size,
rather than the number of plates that was essential to the augmentation
of power. It was, however, found upon trial, that the quantity of
electricity produced by the Voltaic battery, even when of a very
moderate size, was sufficiently copious, and that the chief advantage in
this apparatus was obtained by increasing the intensity, which, however,
still falls very short of that of the common machine.
I should not omit to mention, that a very splendid, and, at the same
time, most powerful battery, was, a few years ago, constructed
under the direction of Sir H. Davy, which he repeatedly exhibited in his
course of electro-chemical lectures. It consists of two thousand double
plates of zinc and copper, of six square inches in dimensions, arranged
in troughs of Wedgwood-ware, each of which contains twenty of these
plates. The troughs are furnished with a contrivance for lifting the
plates out of them in a very convenient and expeditious manner.*
180
CAROLINE.
Well, now that we understand the nature of the action of the Voltaic battery,
I long to hear an account of the discoveries to which it has given
rise.
MRS. B.
You must restrain your impatience, my dear, for I cannot with any
propriety introduce the subject of these discoveries till we come to
them in the regular course of our studies. But, as almost every
substance in nature has already been exposed to the influence of the
Voltaic battery, we shall very soon have occasion to notice its
effects.
Plate XIII.
Vol. II. page 16.
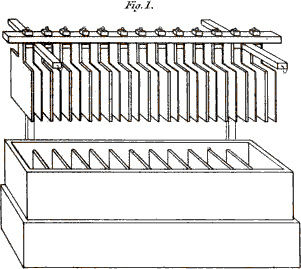
Fig. 1. Voltaic Battery of improved construction with the Plates
out of the Cells.
Larger view
(complete Plate)
181

MRS. B.
To-day we shall examine the chemical
properties of the ATMOSPHERE.
CAROLINE.
I thought that we were first to learn the nature of Oxygen, which come
next in our table of simple bodies?
MRS. B.
And so you shall; the atmosphere being composed of two principles,
Oxygen and Nitrogen, we shall proceed to analyse it, and
consider its component parts separately.
EMILY.
I always thought that the atmosphere had been a very complicated
fluid, composed of all the variety of exhalations from the earth.
MRS. B.
Such substances may be considered rather as heterogeneous
182
and accidental, than as forming any of its component parts; and the
proportion they bear to the whole mass is quite inconsiderable.
Atmospherical air is composed of two
gasses, known by the names of OXYGEN
GAS and NITROGEN or AZOTIC GAS.
EMILY.
Pray what is a gas?
MRS. B.
The name of gas is given to any fluid capable of existing constantly
in an aeriform state, under the pressure and at the temperature of the
atmosphere.
CAROLINE.
Is not water, or any other substance, when evaporated by heat, called
gas?
MRS. B.
No, my dear; vapour is, indeed, an elastic fluid, and bears a strong
resemblance to a gas; there are, however, several points in which they
essentially differ, and by which you may always distinguish them. Steam,
or vapour, owes its elasticity merely to a high temperature, which is
equal to that of boiling water. And it differs from boiling water only
by being united with more caloric, which, as we before explained, is in
a latent state. When
183
steam is cooled, it instantly returns to the form of water; but air, or
gas, has never yet been rendered liquid or solid by any degree of
cold.
EMILY.
But does not gas, as well as vapour, owe its elasticity to
caloric?
MRS. B.
It was the prevailing opinion; and the difference of gas or vapour
was thought to depend on the different manner in which caloric was
united with the basis of these two kinds of elastic fluids. In vapour,
it was considered as in a latent state; in gas, it was said to be
chemically combined. But the late researches of Sir H. Davy have given
rise to a new theory respecting gasses; and there is now reason to
believe that these bodies owe their permanently elastic state, not
solely to caloric, but likewise to the prevalence of either the one or
the other of the two electricities.
EMILY.
When you speak, then, of the simple bodies oxygen and nitrogen, you
mean to express those substances which are the basis of the two
gasses?
MRS. B.
Yes, in strict propriety, for they can properly be called gasses only
when brought to an aeriform state.
184
CAROLINE.
In what proportions are they combined in the atmosphere?
MRS. B.
The oxygen gas constitutes a little more than one-fifth, and the
nitrogen gas a little less than four-fifths. When separated, they are
found to possess qualities totally different from each other. For oxygen
gas is essential both to respiration and combustion, while neither of
these processes can be performed in nitrogen gas.
CAROLINE.
But if nitrogen gas is unfit for respiration, how does it happen that
the large proportion of it which enters into the composition of the
atmosphere is not a great impediment to breathing?
MRS. B.
We should breathe more freely than our lungs could bear, if we
respired oxygen gas alone. The nitrogen is no impediment to respiration,
and probably, on the contrary, answers some useful purpose, though we do
not know in what manner it acts in that process.
EMILY.
And by what means can the two gasses, which compose the atmospheric
air, be separated?
185
MRS. B.
There are many ways of analysing the atmosphere: the two gasses may
be separated first by combustion.
EMILY.
You surprise me! how is it possible that combustion should separate
them?
MRS. B.
I should previously remind you that oxygen is supposed to be the only
simple body naturally combined with negative electricity. In all the
other elements the positive electricity prevails, and they have
consequently, all of them, an attraction for oxygen.*
CAROLINE.
Oxygen the only negatively electrified body! that surprises me
extremely; how then are the combinations of the other bodies performed,
if, according to your explanation of chemical attraction, bodies are
supposed only to combine in virtue of their opposite states of
electricity?
186
MRS. B.
Observe that I said, that oxygen was the only simple body,
naturally negative. Compound bodies, in which oxygen prevails over the
other component parts, are also negative, but their negative energy is
greater or less in proportion as the oxygen predominates. Those
compounds into which oxygen enters in less proportion than the other
constituents, are positive, but their positive energy is diminished in
proportion to the quantity of oxygen which enters into their
composition.
All bodies, therefore, that are not already combined with oxygen,
will attract it, and, under certain circumstances, will absorb it from
the atmosphere, in which case the nitrogen gas will remain alone, and
may thus be obtained in its separate state.
CAROLINE.
I do not understand how a gas can be absorbed?
MRS. B.
It is only the oxygen, or basis of the gas, which is absorbed; and
the two electricities escaping, that is to say, the negative from the
oxygen, the positive from the burning body, unite and produce
caloric.
EMILY.
And what becomes of this caloric?
187
MRS. B.
We shall make this piece of dry wood attract oxygen from the
atmosphere, and you will see what becomes of the caloric.
CAROLINE.
You are joking, Mrs. B—; you do not mean to decompose the
atmosphere with a piece of dry stick?
MRS. B.
Not the whole body of the atmosphere, certainly; but if we can make
this piece of wood attract any quantity of oxygen from it,
a proportional quantity of atmospherical air will be
decomposed.
CAROLINE.
If wood has so strong an attraction for oxygen, why does it not
decompose the atmosphere spontaneously?
MRS. B.
It is found by experience, that an elevation of temperature is
required for the commencement of the union of the oxygen and the
wood.
This elevation of temperature was formerly thought to be necessary,
in order to diminish the cohesive attraction of the wood, and enable the
oxygen to penetrate and combine with it more readily. But since the
introduction of the new theory of chemical combination, another cause
has
188
been assigned, and it is now supposed that the high temperature, by
exalting the electrical energies of bodies, and consequently their force
of attraction, facilitates their combination.
EMILY.
If it is true, that caloric is composed of the two electricities, an
elevation of temperature must necessarily augment the electric energies
of bodies.
MRS. B.
I doubt whether that would be a necessary consequence; for, admitting
this composition of caloric, it is only by its being decomposed that
electricity can be produced. Sir H. Davy, however, in his numerous
experiments, has found it to be an almost invariable rule that the
electrical energies of bodies are increased by elevation of
temperature.
What means then shall we employ to raise the temperature of the wood,
so as to enable it to attract oxygen from the atmosphere?
CAROLINE.
Holding it near the fire, I should think, would answer the
purpose.
MRS. B.
It may, provided you hold it sufficiently close
189
to the fire; for a very considerable elevation of temperature is
required.
CAROLINE.
It has actually taken fire, and yet I did not let it touch the coals,
but I held it so very close that I suppose it caught fire merely from
the intensity of the heat.
MRS. B.
Or you might say, in other words, that the caloric which the wood
imbibed, so much elevated its temperature, and exalted its electric
energy, as to enable it to attract oxygen very rapidly from the
atmosphere.
EMILY.
Does the wood absorb oxygen while it is burning?
MRS. B.
Yes, and the heat and light are produced by the union of the two
electricities which are set at liberty, in consequence of the oxygen
combining with the wood.
CAROLINE.
You astonish me! the heat of a burning body proceeds then as much
from the atmosphere as from the body itself?
MRS. B.
It was supposed that the caloric, given out
190
during combustion, proceeded entirely, or nearly so, from the
decomposition of the oxygen gas; but, according to Sir H. Davy’s new
view of the subject, both the oxygen gas, and the combustible body,
concur in supplying the heat and light, by the union of their opposite
electricities.
EMILY.
I have not yet met with any thing in chemistry that has surprised or
delighted me so much as this explanation of combustion. I was at
first wondering what connection there could be between the affinity of a
body for oxygen and its combustibility; but I think I understand it now
perfectly.
MRS. B.
Combustion then, you see, is nothing more than the rapid combination
of a body with oxygen, attended by the disengagement of light and
heat.
EMILY.
But are there no combustible bodies whose attraction for oxygen is so
strong, that they will combine with it, without the application of
heat?
CAROLINE.
That cannot be; otherwise we should see bodies burning
spontaneously.
191
MRS. B.
But there are some instances of this kind, such as phosphorus,
potassium, and some compound bodies, which I shall hereafter make you
acquainted with. These bodies, however, are prepared by art, for in
general, all the combustions that could occur spontaneously, at the
temperature of the atmosphere, have already taken place; therefore new
combustions cannot happen without the temperature of the body being
raised. Some bodies, however, will burn at a much lower temperature than
others.
CAROLINE.
But the common way of burning a body is not merely to approach it to
one already on fire, but rather to put the one in actual contact with
the other, as when I burn this piece of paper by holding it in the flame
of the fire.
MRS. B.
The closer it is in contact with the source of caloric, the sooner
will its temperature be raised to the degree necessary for it to burn.
If you hold it near the fire, the same effect will be produced; but more
time will be required, as you found to be the case with the piece of
stick.
EMILY.
But why is it not necessary to continue applying
192
caloric throughout the process of combustion, in order to keep up the
electric energy of the wood, which is required to enable it to combine
with the oxygen?
MRS. B.
The caloric which is gradually produced by the two electricities
during combustion, keeps up the temperature of the burning body; so that
when once combustion has begun, no further application of caloric is
required.
CAROLINE.
Since I have learnt this wonderful theory of combustion,
I cannot take my eyes from the fire; and I can scarcely conceive
that the heat and light, which I always supposed to proceed entirely
from the coals, are really produced as much by the atmosphere.
EMILY.
When you blow the fire, you increase the combustion, I suppose,
by supplying the coals with a greater quantity of oxygen gas?
MRS. B.
Certainly; but of course no blowing will produce combustion, unless
the temperature of the coals be first raised. A single spark,
however, is sometimes sufficient to produce that effect; for, as I said
before, when once combustion has commenced,
193
the caloric disengaged is sufficient to elevate the temperature of the
rest of the body, provided that there be a free access of oxygen. It
however sometimes happens that if a fire be ill made, it will be
extinguished before all the fuel is consumed, from the very circumstance
of the combustion being so slow that the caloric disengaged is
insufficient to keep up the temperature of the fuel. You must recollect
that there are three things required in order to produce combustion;
a combustible body, oxygen, and a temperature at which the one will
combine with the other.
EMILY.
You said that combustion was one method of decomposing the
atmosphere, and obtaining the nitrogen gas in its simple state; but how
do you secure this gas, and prevent it from mixing with the rest of the
atmosphere?
MRS. B.
It is necessary for this purpose to burn the body within a close
vessel, which is easily done.—We shall introduce a small lighted
taper (Plate VII.
Fig. 1.) under this glass receiver, which stands in a bason over
water, to prevent all communication with the external air.
Plate VII.
Vol. I. p. 181.
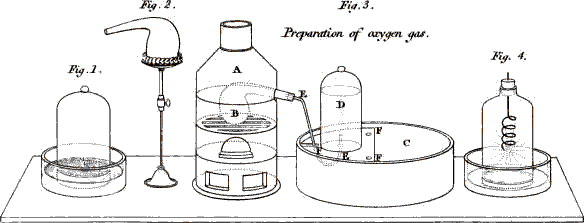
Fig. 1. Combustion of a taper under a receiver.
Fig. 2. A Retort on a stand.
Fig. 3.
A Furnace.
B Earthen Retort in the furnace.
C Water bath.
D Receiver.
E.E Tube conveying the gas from the Retort through the water into
the Receiver.
F.F.F Shelf perforated on which the Receiver stands.
Fig. 4. Combustion of iron wire in oxygen gas.
Larger
view
194
CAROLINE.
How dim the light burns already!—It is now extinguished.
MRS. B.
Can you tell us why it is extinguished?
CAROLINE.
Let me consider.—The receiver was full of atmospherical air;
the taper, in burning within it, must have combined with the oxygen
contained in that air, and the caloric that was disengaged produced the
light of the taper. But when the whole of the oxygen was absorbed, the
whole of its electricity was disengaged; consequently no more caloric
could be produced, the taper ceased to burn, and the flame was
extinguished.
MRS. B.
Your explanation is perfectly correct.
EMILY.
The two constituents of the oxygen gas being thus disposed of, what
remains under the receiver must be pure nitrogen gas?
MRS. B.
There are some circumstances which prevent the nitrogen gas, thus
obtained, from being perfectly pure; but we may easily try whether the
195
oxygen has disappeared, by putting another lighted taper under
it.—You see how instantaneously the flame is extinguished, for
want of oxygen to supply the negative electricity required for the
formation of caloric; and were you to put an animal under the receiver,
it would immediately be suffocated. But that is an experiment which I do
not think your curiosity will tempt you to try.
EMILY.
Certainly not.—But look, Mrs. B., the receiver is full of a
thick white smoke. Is that nitrogen gas?
MRS. B.
No, my dear; nitrogen gas is perfectly transparent and invisible,
like common air. This cloudiness proceeds from a variety of exhalations,
which arise from the burning taper, and the nature of which you cannot
yet understand.
CAROLINE.
The water within the receiver has now risen a little above its level
in the bason. What is the reason of this?
MRS. B.
With a moment’s reflection, I dare say, you would have explained it
yourself. The water rises in consequence of the oxygen gas within it
196
having been destroyed, or rather decomposed, by the combustion of the
taper.
CAROLINE.
Then why did not the water rise immediately when the oxygen gas was
destroyed?
MRS. B.
Because the heat of the taper, whilst burning, produced a dilatation
of the air in the vessel, which at first counteracted this effect.
Another means of decomposing the atmosphere is the oxygenation
of certain metals. This process is very analogous to combustion; it is,
indeed, only a more general term to express the combination of a body
with oxygen.
CAROLINE.
In what respect, then, does it differ from combustion?
MRS. B.
The combination of oxygen in combustion is always accompanied by a
disengagement of light and heat; whilst this circumstance is not a
necessary consequence of simple oxygenation.
CAROLINE.
But how can a body absorb oxygen without the combination of the two
electricities which produce caloric?
197
MRS. B.
Oxygen does not always present itself in a gaseous state; it is a
constituent part of a vast number of bodies, both solid and liquid, in
which it exists in a much denser state than in the atmosphere; and from
these bodies it may be obtained without much disengagement of caloric.
It may likewise, in some cases, be absorbed from the atmosphere without
any sensible production of light and heat; for, if the process be slow,
the caloric is disengaged in such small quantities, and so gradually,
that it is not capable of producing either light or heat. In this case
the absorption of oxygen is called oxygenation or
oxydation, instead of combustion, as the production of
sensible light and heat is essential to the latter.
EMILY.
I wonder that metals can unite with oxygen; for, as they are so
dense, their attraction of aggregation must be very great; and I should
have thought that oxygen could never have penetrated such bodies.
MRS. B.
Their strong attraction for oxygen counterbalances this obstacle.
Most metals, however, require to be made red-hot before they are capable
of attracting oxygen in any considerable quantity.
198
By this combination they lose most of their metallic properties, and
fall into a kind of powder, formerly called calx, but now much
more properly termed an oxyd; thus we have oxyd of lead,
oxyd of iron, &c.
EMILY.
And in the Voltaic battery, it is, I suppose, an oxyd of zinc, that
is formed by the union of the oxygen with that metal?
MRS. B.
Yes, it is.
CAROLINE.
The word oxyd, then, simply means a metal combined with oxygen?
MRS. B.
Yes; but the term is not confined to metals, though chiefly applied
to them. Any body whatever, that has combined with a certain quantity of
oxygen, either by means of oxydation or combustion, is called an
oxyd, and is said to be oxydated or oxygenated.
EMILY.
Metals, when converted into oxyds, become, I suppose,
negative?
MRS. B.
Not in general; because in most oxyds the positive energy of the
metal more than counterbalances
199
the native energy of the oxygen with which it combines.
This black powder is an oxyd of manganese, a metal which has so
strong an affinity for oxygen, that it attracts that substance from the
atmosphere at any known temperature: it is therefore never found in its
metallic form, but always in that of an oxyd, in which state, you see,
it has very little of the appearance of a metal. It is now heavier than
it was before oxydation, in consequence of the additional weight of the
oxygen with which it has combined.
CAROLINE.
I am very glad to hear that; for I confess I could not help having
some doubts whether oxygen was really a substance, as it is not to be
obtained in a simple and palpable state; but its weight is,
I think, a decisive proof of its being a real body.
MRS. B.
It is easy to estimate its weight, by separating it from the
manganese, and finding how much the latter has lost.
EMILY.
But if you can take the oxygen from the metal, shall we not then have
it in its palpable simple state?
MRS. B.
No; for I can only separate the oxygen from
200
the manganese, by presenting to it some other body, for which it has a
greater affinity than for the manganese. Caloric affording the two
electricities is decomposed, and one of them uniting with the oxygen,
restores it to the aëriform state.
EMILY.
But you said just now, that manganese would attract oxygen from the
atmosphere in which it is combined with the negative electricity; how,
therefore, can the oxygen have a superior affinity for that electricity,
since it abandons it to combine with the manganese?
MRS. B.
I give you credit for this objection, Emily; and the only answer I
can make to it is, that the mutual affinities of metals for oxygen, and
of oxygen for electricity, vary at different temperatures;
a certain degree of heat will, therefore, dispose a metal to
combine with oxygen, whilst, on the contrary, the former will be
compelled to part with the latter, when the temperature is further
increased. I have put some oxyd of manganese into a retort, which
is an earthen vessel with a bent neck, such as you see here. (Plate VII. Fig. 2.)—The
retort containing the manganese you cannot see, as I have enclosed it in
this furnace, where it is now red-hot. But, in order to
201
make you sensible of the escape of the gas, which is itself invisible,
I have connected the neck of the retort with this bent tube, the
extremity of which is immersed in this vessel of water. (Plate VII. Fig. 3.)—Do you
see the bubbles of air rise through the water?
CAROLINE.
Perfectly. This, then, is pure oxygen gas; what a pity it should be
lost! Could you not preserve it?
MRS. B.
We shall collect it in this receiver.—For this purpose, you
observe, I first fill it with water, in order to exclude the
atmospherical air; and then place it over the bubbles that issue from
the retort, so as to make them rise through the water to the upper part
of the receiver.
EMILY.
The bubbles of oxygen gas rise, I suppose, from their specific
levity?
MRS. B.
Yes; for though oxygen forms rather a heavy gas, it is light compared
to water. You see how it gradually displaces the water from the
receiver. It is now full of gas, and I may leave it inverted in water on
this shelf, where I can keep the gas
202
as long as I choose, for future experiments. This apparatus (which is
indispensable in all experiments in which gases are concerned) is called
a water-bath.
CAROLINE.
It is a very clever contrivance, indeed; equally simple and useful.
How convenient the shelf is for the receiver to rest upon under water,
and the holes in it for the gas to pass into the receiver! I long
to make some experiments with this apparatus.
MRS. B.
I shall try your skill that way, when you have a little more
experience. I am now going to show you an experiment, which proves,
in a very striking manner, how essential oxygen is to combustion. You
will see that iron itself will burn in this gas, in the most rapid and
brilliant manner.
CAROLINE.
Really! I did not know that it was possible to burn iron.
EMILY.
Iron is a simple body, and you know, Caroline, that all simple bodies
are naturally positive, and therefore must have an affinity for
oxygen.
MRS. B.
Iron will, however, not burn in atmospherical
203
air without a very great elevation of temperature; but it is eminently
combustible in pure oxygen gas; and what will surprise you still more,
it can be set on fire without any considerable rise of temperature. You
see this spiral iron wire—I fasten it at one end to this
cork, which is made to fit an opening at the top of the glass-receiver.
(Plate VII.
Fig. 4.)
EMILY.
I see the opening in the receiver; but it is carefully closed by a
ground glass-stopper.
MRS. B.
That is in order to prevent the gas from escaping; but I shall take
out the stopper, and put in the cork, to which the wire hangs.—Now
I mean to burn this wire in the oxygen gas, but I must fix a small piece
of lighted tinder to the extremity of it, in order to give the first
impulse to combustion; for, however powerful oxygen is in promoting
combustion, you must recollect that it cannot take place without some
elevation of temperature. I shall now introduce the wire into the
receiver, by quickly changing the stoppers.
CAROLINE.
Is there no danger of the gas escaping while you change the
stoppers?
204
MRS. B.
Oxygen gas is a little heavier than atmospherical air, therefore it
will not mix with it very rapidly; and, if I do not leave the opening
uncovered, we shall not lose any——
CAROLINE.
Oh, what a brilliant and beautiful flame!
EMILY.
It is as white and dazzling as the sun!—Now a piece of the
melted wire drops to the bottom: I fear it is extinguished; but no,
it burns again as bright as ever.
MRS. B.
It will burn till the wire is entirely consumed, provided the oxygen
is not first expended: for you know it can burn only while there is
oxygen to combine with it.
CAROLINE.
I never saw a more beautiful light. My eyes can hardly bear it! How
astonishing to think that all this caloric was contained in the small
quantity of gas and iron that was enclosed in the receiver; and that,
without producing any sensible heat!
CAROLINE.
How wonderfully quick combustion goes on in pure oxygen gas! But
pray, are these drops of burnt iron as heavy as the wire was before?
205
MRS. B.
They are even heavier; for the iron, in burning, has acquired exactly
the weight of the oxygen which has disappeared, and is now combined with
it. It has become an oxyd of iron.
CAROLINE.
I do not know what you mean by saying that the oxygen has
disappeared, Mrs. B., for it was always invisible.
MRS. B.
True, my dear; the expression was incorrect. But though you could not
see the oxygen gas, I believe you had no doubt of its presence, as
the effect it produced on the wire was sufficiently evident.
CAROLINE.
Yes, indeed; yet you know it was the caloric, and not the oxygen gas
itself, that dazzled us so much.
MRS. B.
You are not quite correct in your turn, in saying the caloric dazzled
you; for caloric is invisible; it affects only the sense of feeling; it
was the light which dazzled you.
CAROLINE.
True; but light and caloric are such constant companions, that it is
difficult to separate them, even in idea.
206
MRS. B.
The easier it is to confound them, the more careful you should be in
making the distinction.
CAROLINE.
But why has the water now risen, and filled part of the receiver?
MRS. B.
Indeed, Caroline, I did not suppose you would have asked such a
question! I dare say, Emily, you can answer it.
EMILY.
Let me reflect . . . . . . The oxygen has combined with the wire; the
caloric has escaped; consequently nothing can remain in the receiver,
and the water will rise to fill the vacuum.
CAROLINE.
I wonder that I did not think of that. I wish that we had weighed the
wire and the oxygen gas before combustion; we might then have found
whether the weight of the oxyd was equal to that of both.
MRS. B.
You might try the experiment if you particularly wished it; but I can
assure you, that, if accurately performed, it never fails to show that
the additional weight of the oxyd is precisely equal to that
207
of the oxygen absorbed, whether the process has been a real combustion,
or a simple oxygenation.
CAROLINE.
But this cannot be the case with combustions in general; for when any
substance is burnt in the common air, so far from increasing in weight,
it is evidently diminished, and sometimes entirely consumed.
MRS. B.
But what do you mean by the expression consumed? You cannot
suppose that the smallest particle of any substance in nature can be
actually destroyed. A compound body is decomposed by combustion;
some of its constituent parts fly off in a gaseous form, while others
remain in a concrete state; the former are called the volatile,
the latter the fixed products of combustion. But if we collect
the whole of them, we shall always find that they exceed the weight of
the combustible body, by that of the oxygen which has combined with them
during combustion.
EMILY.
In the combustion of a coal fire, then, I suppose that the ashes
are what would be called the fixed product, and the smoke the volatile
product?
208
MRS. B.
Yet when the fire burns best, and the quantity of volatile products
should be the greatest, there is no smoke; how can you account for
that?
EMILY.
Indeed I cannot; therefore I suppose that I was not right in my
conjecture.
MRS. B.
Not quite: ashes, as you supposed, are a fixed product of combustion;
but smoke, properly speaking, is not one of the volatile products, as it
consists of some minute undecomposed particles of the coals that are
carried off by the heated air without being burnt, and are either
deposited in the form of soot, or dispersed by the wind. Smoke,
therefore, ultimately, becomes one of the fixed products of
combustion. And you may easily conceive that the stronger the fire is,
the less smoke is produced, because the fewer particles escape
combustion. On this principle depends the invention of Argand’s Patent
Lamps; a current of air is made to pass through the cylindrical
wick of the lamp, by which means it is so plentifully supplied with
oxygen, that scarcely a particle of oil escapes combustion, nor is there
any smoke produced.
EMILY.
But what then are the volatile products of combustion?
209
MRS. B.
Various new compounds, with which you are not yet acquainted, and
which being converted by caloric either into vapour or gas, are
invisible; but they can be collected, and we shall examine them at some
future period.
CAROLINE.
There are then other gases, besides the oxygen and nitrogen
gases.
MRS. B.
Yes, several: any substance that can assume and maintain the form of
an elastic fluid at the temperature of the atmosphere, is called a gas.
We shall examine the several gases in their respective places; but we
must now confine our attention to those that compose the atmosphere.
I shall show you another method of decomposing the atmosphere, which
is very simple. In breathing, we retain a portion of the oxygen, and
expire the nitrogen gas; so that if we breathe in a closed vessel, for a
certain length of time, the air within it will be deprived of its oxygen
gas. Which of you will make the experiment?
CAROLINE.
I should be very glad to try it.
210
MRS. B.
Very well; breathe several times through this glass tube into the
receiver with which it is connected, until you feel that your breath is
exhausted.
CAROLINE.
I am quite out of breath already!
MRS. B.
Now let us try the gas with a lighted taper.
EMILY.
It is very pure nitrogen gas, for the taper is immediately
extinguished.
MRS. B.
That is not a proof of its being pure, but only of the absence of
oxygen, as it is that principle alone which can produce combustion,
every other gas being absolutely incapable of it.
EMILY.
In the methods which you have shown us, for decomposing the
atmosphere, the oxygen always abandons the nitrogen; but is there no way
of taking the nitrogen from the oxygen, so as to obtain the latter pure
from the atmosphere?
MRS. B.
You must observe, that whenever oxygen is
211
taken from the atmosphere, it is by decomposing the oxygen gas; we
cannot do the same with the nitrogen gas, because nitrogen has a
stronger affinity for caloric than for any other known principle: it
appears impossible therefore to separate it from the atmosphere by the
power of affinities. But if we cannot obtain the oxygen gas, by this
means, in its separate state, we have no difficulty (as you have seen)
to procure it in its gaseous form, by taking it from those substances
that have absorbed it from the atmosphere, as we did with the oxyd of
manganese.
EMILY.
Can atmospherical air be recomposed, by mixing due proportions of
oxygen and nitrogen gases?
MRS. B.
Yes: if about one part of oxygen gas be mixed with about four parts
of nitrogen gas, atmospherical air is produced.*
EMILY.
The air, then, must be an oxyd of nitrogen?
MRS. B.
No, my dear; for there must be a chemical
212
combination between oxygen and nitrogen in order to produce an oxyd;
whilst in the atmosphere these two substances are separately combined
with caloric, forming two distinct gases, which are simply mixed in the
formation of the atmosphere.
I shall say nothing more of oxygen and nitrogen at present, as we
shall continually have occasion to refer to them in our future
conversations. They are both very abundant in nature; nitrogen is the
most plentiful in the atmosphere, and exists also in all animal
substances; oxygen forms a constituent part, both of the animal and
vegetable kingdoms, from which it may be obtained by a variety of
chemical means. But it is now time to conclude our lesson. I am
afraid you have learnt more to-day than you will be able to
remember.
CAROLINE.
I assure you that I have been too much interested in it, ever to
forget it. In regard to nitrogen there seems to be but little to
remember; it makes a very insignificant figure in comparison to oxygen,
although it composes a much larger portion of the atmosphere.
MRS. B.
Perhaps this insignificance you complain of may arise from the
compound nature of nitrogen, for though I have hitherto considered it as
a simple
213
body, because it is not known in any natural process to be decomposed,
yet from some experiments of Sir H. Davy, there appears to be reason for
suspecting that nitrogen is a compound body, as we shall see afterwards.
But even in its simple state, it will not appear so insignificant when
you are better acquainted with it; for though it seems to perform but a
passive part in the atmosphere, and has no very striking properties,
when considered in its separate state, yet you will see by-and-bye what
a very important agent it becomes, when combined with other bodies. But
no more of this at present; we must reserve it for its proper place.
214

CAROLINE.
The next simple bodies we come to are CHLORINE and IODINE. Pray what kinds of substances are these; are
they also invisible?
MRS. B.
No; for chlorine, in the state of gas, has a distinct greenish
colour, and is therefore visible; and iodine, in the same state, has a
beautiful claret-red colour. The knowledge of these two bodies, however,
and the explanation of their properties, imply various considerations,
which you would not yet be able to understand; we shall therefore defer
their examination to some future conversation, and we shall pass on to
the next simple substance, Hydrogen,
which we cannot, any more than oxygen, obtain in a visible or palpable
form. We are acquainted with it only in its gaseous state, as we are
with oxygen and nitrogen.
CAROLINE.
But in its gaseous state it cannot be called a
215
simple substance, since it is combined with heat and electricity?
MRS. B.
True, my dear; but as we do not know in nature of any substance which
is not more or less combined with caloric and electricity, we are apt to
say that a substance is in its pure state when combined with those
agents only.
Hydrogen was formerly called inflammable air, as it is
extremely combustible, and burns with a great flame. Since the invention
of the new nomenclature, it has obtained the name of hydrogen, which is
derived from two Greek words, the meaning of which is, to produce
water.
EMILY.
And how does hydrogen produce water?
MRS. B.
By its combustion. Water is composed of eighty-five parts, by weight,
of oxygen, combined with fifteen parts of hydrogen; or of two parts, by
bulk of hydrogen gas, to one part of oxygen gas.
CAROLINE.
Really! is it possible that water should be a combination of two
gases, and that one of these
216
should be inflammable air! Hydrogen must be a most extraordinary gas
that will produce both fire and water.
EMILY.
But I thought you said that combustion could take place in no gas but
oxygen?
MRS. B.
Do you recollect what the process of combustion consists in?
EMILY.
In the combination of a body with oxygen, with disengagement of light
and heat.
MRS. B.
Therefore when I say that hydrogen is combustible, I mean that
it has an affinity for oxygen; but, like all other combustible
substances, it cannot burn unless supplied with oxygen, and also heated
to a proper temperature.
CAROLINE.
The simply mixing fifteen parts of hydrogen, with eighty-five parts
of oxygen gas, will not, therefore, produce water?
MRS. B.
No; water being a much denser fluid than gases, in order to reduce
these gases to a liquid, it is
217
necessary to diminish the quantity of caloric or electricity which
maintains them in an elastic form.
EMILY.
That I should think might be done by combining the oxygen and
hydrogen together; for in combining they would give out their respective
electricities in the form of caloric, and by this means would be
condensed.
CAROLINE.
But you forget, Emily, that in order to make the oxygen and hydrogen
combine, you must begin by elevating their temperature, which increases,
instead of diminishing, their electric energies.
MRS. B.
Emily is, however, right; for though it is necessary to raise their
temperature, in order to make them combine, as that combination affords
them the means of parting with their electricities, it is eventually the
cause of the diminution of electric energy.
CAROLINE.
You love to deal in paradoxes to-day, Mrs. B.—Fire, then,
produces water?
MRS. B.
The combustion of hydrogen gas certainly does;
218
but you do not seem to have remembered the theory of combustion so well
as you thought you would. Can you tell me what happens in the combustion
of hydrogen gas?
CAROLINE.
The hydrogen combines with the oxygen, and their opposite
electricities are disengaged in the form of caloric.—Yes,
I think I understand it now—by the loss of this caloric, the
gases are condensed into a liquid.
EMILY.
Water, then, I suppose, when it evaporates and incorporates with the
atmosphere, is decomposed and converted into hydrogen and oxygen
gases?
MRS. B.
No, my dear—there you are quite mistaken: the decomposition of
water is totally different from its evaporation; for in the latter case
(as you should recollect) water is only in a state of very minute
division; and is merely suspended in the atmosphere, without any
chemical combination, and without any separation of its constituent
parts. As long as these remain combined, they form WATER, whether in a state of liquidity, or in that
of an elastic fluid, as vapour, or under the solid form of ice.
In our experiments on latent heat, you may recollect
219
that we caused water successively to pass through these three forms,
merely by an increase or diminution of caloric, without employing any
power of attraction, or effecting any decomposition.
CAROLINE.
But are there no means of decomposing water?
MRS. B.
Yes, several: charcoal, and metals, when heated red hot, will attract
the oxygen from water, in the same manner as they will from the
atmosphere.
CAROLINE.
Hydrogen, I see, is like nitrogen, a poor dependant friend of
oxygen, which is continually forsaken for greater favourites.
MRS. B.
The connection, or friendship, as you choose to call it, is much more
intimate between oxygen and hydrogen, in the state of water, than
between oxygen and nitrogen, in the atmosphere; for, in the first case,
there is a chemical union and condensation of the two substances; in the
latter, they are simply mixed together in their gaseous state. You will
find, however, that, in some cases, nitrogen is quite as intimately
connected with oxygen, as hydrogen is.—But this is foreign to our
present subject.
220
EMILY.
Water, then, is an oxyd, though the atmospherical air is not?
MRS. B.
It is not commonly called an oxyd, though, according to our
definition, it may, no doubt, be referred to that class of bodies.
CAROLINE.
I should like extremely to see water decomposed.
MRS. B.
I can gratify your curiosity by a much more easy process than the
oxydation of charcoal or metals: the decomposition of water by these
latter means takes up a great deal of time, and is attended with much
trouble; for it is necessary that the charcoal or metal should be made
red hot in a furnace, that the water should pass over them in a state of
vapour, that the gas formed should be collected over the water-bath,
&c. In short, it is a very complicated affair. But the same effect
may be produced with the greatest facility, by the action of the Voltaic
battery, which this will give me an opportunity of exhibiting.
CAROLINE.
I am very glad of that, for I longed to see the power of this
apparatus in decomposing bodies.
221
MRS. B.
For this purpose I fill this piece of glass-tube (Plate VIII. fig. 1.) with
water, and cork it up at both ends; through one of the corks I introduce
that wire of the battery which conveys the positive electricity; and the
wire which conveys the negative electricity is made to pass through the
other cork, so that the two wires approach each other sufficiently near
to give out their respective electricities.
Plate VIII.
Vol. I. p. 206

Fig. 1. Apparatus for the decomposition of water by the
Voltaic Battery.
Larger view
(complete Plate)
CAROLINE.
It does not appear to me that you approach the wires so near as you
did when you made the battery act by itself.
MRS. B.
Water being a better conductor of electricity than air, the two wires
will act on each other at a greater distance in the former than in the
latter.
EMILY.
Now the electrical effect appears: I see small bubbles of air emitted
from each wire.
MRS. B.
Each wire decomposes the water, the positive by combining with its
oxygen which is negative, the negative by combining with its hydrogen
which is positive.
222
CAROLINE.
That is wonderfully curious! But what are the small bubbles of
air?
MRS. B.
Those that appear to proceed from the positive wire, are the result
of the decomposition of the water by that wire. That is to say, the
positive electricity having combined with some of the oxygen of the
water, the particles of hydrogen which were combined with that portion
of oxygen are set at liberty, and appear in the form of small bubbles of
gas or air.
EMILY.
And I suppose the negative fluid having in the same manner combined
with some of the hydrogen of the water, the particles of oxygen that
were combined with it, are set free, and emitted in a gaseous form.
MRS. B.
Precisely so. But I should not forget to observe, that the wires used
in this experiment are made of platina, a metal which is not
capable of combining with oxygen; for otherwise the wire would combine
with the oxygen, and the hydrogen alone would be disengaged.
223
CAROLINE.
But could not water be decomposed without the electric circle being
completed? If, for instance, you immersed only the positive wire in the
water, would it not combine with the oxygen, and the hydrogen gas be
given out?
MRS. B.
No; for as you may recollect, the battery cannot act unless the
circle be completed; since the positive wire will not give out its
electricity, unless attracted by that of the negative wire.
CAROLINE.
I understand it now.—But look, Mrs. B., the decomposition of
the water which has now been going on for some time, does not sensibly
diminish its quantity—what is the reason of that?
MRS. B.
Because the quantity decomposed is so extremely small. If you compare
the density of water with that of the gases into which it is resolved,
you must be aware that a single drop of water is sufficient to produce
thousands of such small bubbles as those you now perceive.
CAROLINE.
But in this experiment, we obtain the oxygen
224
and hydrogen gases mixed together. Is there any means of procuring the
two gases separately?
MRS. B.
They can be collected separately with great ease, by modifying a
little the experiment. Thus if instead of one tube, we employ two, as
you see here, (c, d, Plate VIII. fig. 2.) both tubes being closed at one end, and
open at the other; and if after filling these tubes with water, we place
them standing in a glass of water (e), with their open end
downwards, you will see that the moment we connect the wires (a, b)
which proceed upwards from the interior of each tube, the one with one
end of the battery, and the other with the other end, the water in the
tubes will be decomposed; hydrogen will be given out round the wire in
the tube connected with the positive end of the battery, and oxygen in
the other; and these gases will be evolved, exactly in the proportions
which I have before mentioned, namely, two measures of hydrogen for one
of oxygen. We shall now begin the experiment, but it will be some time
before any sensible quantity of the gases can be collected.
Plate VIII.
Vol. I. p. 206
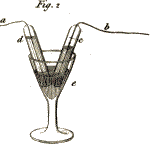
Fig. 2. Apparatus for decomposing water by Voltaic
Electricity & obtaining the gasses separate.
Larger view
(complete Plate)
EMILY.
The decomposition of water in this way, slow as it is, is certainly
very striking; but I confess that I should be still more gratified, if
you could shew it us on a larger scale, and by a quicker process.
225
I am sorry that the decomposition of water by charcoal or metals is
attended with so much inconvenience.
MRS. B.
Water may be decomposed by means of metals without any difficulty;
but for this purpose the intervention of an acid is required. Thus, if
we add some sulphuric acid (a substance with the nature of which
you are not yet acquainted) to the water which the metal is to
decompose, the acid disposes the metal to combine with the oxygen of the
water so readily and abundantly, that no heat is required to hasten the
process. Of this I am going to shew you an instance. I put into
this bottle the water that is to be decomposed, as also the metal that
is to effect that decomposition by combining with the oxygen, and the
acid which is to facilitate the combination of the metal and the oxygen.
You will see with what violence these will act on each other.
CAROLINE.
But what metal is it that you employ for this purpose?
MRS. B.
It is iron; and it is used in the state of filings, as these present
a greater surface to the acid than a solid piece of metal. For as it is
the surface of the metal which is acted upon by the acid, and is
disposed to receive the oxygen produced by the
226
decomposition of the water, it necessarily follows that the greater is
the surface, the more considerable is the effect. The bubbles which are
now rising are hydrogen gas——
CAROLINE.
How disagreeably it smells!
MRS. B.
It is indeed unpleasant, though, I believe, not particularly hurtful.
We shall not, however, suffer any more to escape, as it will be wanted
for experiments. I shall, therefore, collect it in a
glass-receiver, by making it pass through this bent tube, which will
conduct it into the water-bath. (Plate VIII. fig. 3.)
Plate VIII.
Vol. I. p. 206
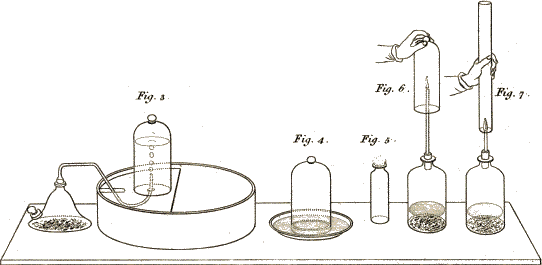
Fig. 3. Apparatus for preparing & collecting hydrogen
gas.
Fig. 4. Receiver full of hydrogen gas inverted over
water.
Fig. 5. Slow combustion of hydrogen gas.
Fig. 6. Apparatus for illustrating the formation of water by
the combustion of hydrogen gas.
Fig. 7. Apparatus for producing harmonic sounds by the
combustion of hydrogen gas.
Larger view
(complete Plate)
EMILY.
How very rapidly the gas escapes! it is perfectly transparent, and
without any colour whatever.—Now the receiver is
full——
MRS. B.
We shall, therefore, remove it, and substitute another in its place.
But you must observe, that when the receiver is full, it is necessary to
keep it inverted with the mouth under water, otherwise the gas would
escape. And in order that it may not be in the way, I introduce
within the bath, under the water, a saucer, into which I slide the
receiver, so that it can be taken out of the bath
227
and conveyed any where, the water in the saucer being equally effectual
in preventing its escape as that in the bath. (Plate VIII. fig. 4.)
EMILY.
I am quite surprised to see what a large quantity of hydrogen gas can
be produced by such a small quantity of water, especially as oxygen is
the principal constituent of water.
MRS. B.
In weight it is; but not in volume. For though the proportion, by
weight, is nearly six parts of oxygen to one of hydrogen, yet the
proportion of the volume of the gases, is about one part of oxygen to
two of hydrogen; so much heavier is the former than the latter.
CAROLINE.
But why is the vessel in which the water is decomposed so hot? As the
water changes from a liquid to a gaseous form, cold should be produced
instead of heat.
MRS. B.
No; for if one of the constituents of water is converted into a gas,
the other becomes solid in combining with the metal.
EMILY.
In this case, then, neither heat nor cold should be produced?
228
MRS. B.
True: but observe that the sensible heat which is disengaged in this
operation, is not owing to the decomposition of the water, but to an
extrication of heat produced by the mixture of water and sulphuric acid.
I will mix some water and sulphuric acid together in this glass,
that you may feel the surprising quantity of heat that is disengaged by
their union—now take hold of the glass——
CAROLINE.
Indeed I cannot; it feels as hot as boiling water. I should have
imagined there would have been heat enough disengaged to have rendered
the liquid solid.
MRS. B.
As, however, it does not produce that effect, we cannot refer this
heat to the modification called latent heat. We may, however,
I think, consider it as heat of capacity, as the liquid is
condensed by its loss; and if you were to repeat the experiment, in a
graduated tube, you would find that the two liquids, when mixed, occupy
considerably less space than they did separately.—But we will
reserve this to another opportunity, and attend at present to the
hydrogen gas which we have been producing.
If I now set the hydrogen gas, which is contained in this receiver,
at liberty all at once, and
229
kindle it as soon as it comes in contact with the atmosphere, by
presenting it to a candle, it will so suddenly and rapidly decompose the
oxygen gas, by combining with its basis, that an explosion, or a
detonation (as chemists commonly call it), will be produced. For
this purpose, I need only take up the receiver, and quickly present
its open mouth to the candle——so . . . .
CAROLINE.
It produced only a sort of hissing noise, with a vivid flash of
light. I had expected a much greater report.
MRS. B.
And so it would have been, had the gases been closely confined at the
moment they were made to explode. If, for instance, we were to put in
this bottle a mixture of hydrogen gas and atmospheric air; and if, after
corking the bottle, we should kindle the mixture by a very small
orifice, from the sudden dilatation of the gases at the moment of their
combination, the bottle must either fly to pieces, or the cork be blown
out with considerable violence.
CAROLINE.
But in the experiment which we have just seen, if you did not kindle
the hydrogen gas, would it not equally combine with the oxygen?
230
MRS. B.
Certainly not; for, as I have just explained to you, it is necessary
that the oxygen and hydrogen gases be burnt together, in order to
combine chemically and produce water.
CAROLINE.
That is true; but I thought this was a different combination, for I
see no water produced.
MRS. B.
The water resulting from this detonation was so small in quantity,
and in such a state of minute division, as to be invisible. But water
certainly was produced; for oxygen is incapable of combining with
hydrogen in any other proportions than those that form water; therefore
water must always be the result of their combination.
If, instead of bringing the hydrogen gas into sudden contact with the
atmosphere (as we did just now) so as to make the whole of it explode
the moment it is kindled, we allow but a very small surface of gas to
burn in contact with the atmosphere, the combustion goes on quietly and
gradually at the point of contact, without any detonation, because the
surfaces brought together are too small for the immediate union of
gases. The experiment is a very easy one. This phial, with a narrow
neck, (Plate VIII.
fig. 5.) is full
231
of hydrogen gas, and is carefully corked. If I take out the cork without
moving the phial, and quickly approach the candle to the orifice, you
will see how different the result will be——
EMILY.
How prettily it burns, with a blue flame! The flame is gradually
sinking within the phial—now it has entirely disappeared. But does
not this combustion likewise produce water?
MRS. B.
Undoubtedly. In order to make the formation of the water sensible to
you, I shall procure a fresh supply of hydrogen gas, by putting
into this bottle (Plate
VIII. fig. 6.) iron filings, water, and sulphuric acid,
materials similar to those which we have just used for the same purpose.
I shall then cork up the bottle, leaving only a small orifice in
the cork, with a piece of glass-tube fixed to it, through which the gas
will issue in a continued rapid stream.
CAROLINE.
I hear already the hissing of the gas through the tube, and I can
feel a strong current against my hand.
MRS. B.
This current I am going to kindle with the candle—see how
vividly it burns——
232
EMILY.
It burns like a candle with a long flame. But why does this
combustion last so much longer than in the former experiment?
MRS. B.
The combustion goes on uninterruptedly as long as the new gas
continues to be produced. Now if I invert this receiver over the flame,
you will soon perceive its internal surface covered with a very fine
dew, which is pure water——
CAROLINE.
Yes, indeed; the glass is now quite dim with moisture! How glad I am
that we can see the water produced by this combustion.
EMILY.
It is exactly what I was anxious to see; for I confess I was a little
incredulous.
MRS. B.
If I had not held the glass-bell over the flame, the water would have
escaped in the state of vapour, as it did in the former experiment. We
have here, of course, obtained but a very small quantity of water; but
the difficulty of procuring a proper apparatus, with sufficient
quantities of
233
gases, prevents my showing it you on a larger scale.
The composition of water was discovered about the same period, both
by Mr. Cavendish, in this country, and by the celebrated French chemist
Lavoisier. The latter invented a very perfect and ingenious apparatus to
perform, with great accuracy, and upon a large scale, the formation of
water by the combination of oxygen and hydrogen gases. Two tubes,
conveying due proportions, the one of oxygen, the other of hydrogen gas,
are inserted at opposite sides of a large globe of glass, previously
exhausted of air; the two streams of gas are kindled within the globe,
by the electrical spark, at the point where they come in contact; they
burn together, that is to say, the hydrogen combines with the oxygen,
the caloric is set at liberty, and a quantity of water is produced
exactly equal, in weight, to that of the two gases introduced into the
globe.
CAROLINE.
And what was the greatest quantity of water ever formed in this
apparatus?
MRS. B.
Several ounces; indeed, very nearly a pound, if I recollect right;
but the operation lasted many days.
234
EMILY.
This experiment must have convinced all the world of the truth of the
discovery. Pray, if improper proportions of the gases were mixed and set
fire to, what would be the result?
MRS. B.
Water would equally be formed, but there would be a residue of either
one or other of the gases, because, as I have already told you, hydrogen
and oxygen will combine only in the proportions requisite for the
formation of water.
EMILY.
Look, Mrs. B., our experiment with the Voltaic battery (Plate VIII. fig. 2.) has made
great progress; a quantity of gas has been formed in each tube, but
in one of them there is twice as much gas as in the other.
MRS. B.
Yes; because, as I said before, water is composed of two volumes of
hydrogen to one of oxygen—and if we should now mix these gases
together and set fire to them by an electrical spark, both gases would
entirely disappear, and a small quantity of water would be formed.
There is another curious effect produced by the combustion of
hydrogen gas, which I shall show
235
you, though I must acquaint you first, that I cannot well explain the
cause of it. For this purpose, I must put some materials into our
apparatus, in order to obtain a stream of hydrogen gas, just as we have
done before. The process is already going on, and the gas is rushing
through the tube—I shall now kindle it with the
taper——
EMILY.
It burns exactly as it did before——What is the curious
effect which you were mentioning?
MRS. B.
Instead of the receiver, by means of which we have just seen the
drops of water form, we shall invert over the flame this piece of tube,
which is about two feet in length, and one inch in diameter (Plate VIII. fig. 7.); but you
must observe that it is open at both ends.
EMILY.
What a strange noise it makes! something like the Æolian harp, but
not so sweet.
CAROLINE.
It is very singular, indeed; but I think rather too powerful to be
pleasing. And is not this sound accounted for?
236
MRS. B.
That the percussion of glass, by a rapid stream of gas, should
produce a sound, is not extraordinary: but the sound here is so
peculiar, that no other gas has a similar effect. Perhaps it is owing to
a brisk vibratory motion of the glass, occasioned by the successive
formation and condensation of small drops of water on the sides of the
glass tube, and the air rushing in to replace the vacuum formed.*
CAROLINE.
How very much this flame resembles the burning of a candle.
MRS. B.
The burning of a candle is produced by much the same means.
A great deal of hydrogen is contained in candles, whether of tallow
or wax. This hydrogen being converted into gas by the heat of the
candle, combines with the oxygen of the atmosphere, and flame and water
result from this combination. So that, in fact, the flame of a candle is
owing to the combustion of hydrogen gas. An elevation of temperature,
such as is produced by a lighted match or taper, is required to give the
first impulse to the combustion; but afterwards
237
it goes on of itself, because the candle finds a supply of caloric in
the successive quantities of heat which results from the union of the
two electricities given out by the gases during their combustion. But
there are other circumstances connected with the combustion of candles
and lamps, which I cannot explain to you till you are acquainted with
carbon, which is one of their constituent parts. In general,
however, whenever you see flame, you may infer that it is owing to the
formation and burning of hydrogen gas*; for flame is the peculiar mode of
burning hydrogen gas, which, with only one or two apparent exceptions,
does not belong to any other combustible.
EMILY.
You astonish me! I understood that flame was the caloric produced by
the union of the two electricities, in all combustions whatever?
MRS. B.
Your error proceeded from your vague and incorrect idea of flame; you
have confounded it with light and caloric in general. Flame always
implies caloric, since it is produced by the combustion of hydrogen gas;
but all caloric does not
238
imply flame. Many bodies burn with intense heat without producing flame.
Coals, for instance, burn with flame until all the hydrogen which they
contain is evaporated; but when they afterwards become red hot, much
more caloric is disengaged than when they produce flame.
CAROLINE.
But the iron wire, which you burnt in oxygen gas, appeared to me to
emit flame; yet, as it was a simple metal, it could contain no
hydrogen?
MRS. B.
It produced a sparkling dazzling blaze of light, but no real
flame.
EMILY.
And what is the cause of the regular shape of the flame of a
candle?
MRS. B.
The regular stream of hydrogen gas which exhales from its combustible
matter.
CAROLINE.
But the hydrogen gas must, from its great levity, ascend into the
upper regions of the atmosphere; why therefore does not the flame
continue to accompany it?
239
MRS. B.
The combustion of the hydrogen gas is completed at the point where
the flame terminates; it then ceases to be hydrogen gas, as it is
converted by its combination with oxygen into watery vapour; but in a
state of such minute division as to be invisible.
CAROLINE.
I do not understand what is the use of the wick of a candle, since
the hydrogen gas burns so well without it?
MRS. B.
The combustible matter of the candle must be decomposed in order to
emit the hydrogen gas, and the wick is instrumental in effecting this
decomposition. Its combustion first melts the combustible matter, and .
. . .
CAROLINE.
But in lamps the combustible matter is already fluid, and yet they
also require wicks?
MRS. B.
I am going to add that, afterwards, the burning wick (by the power of
capillary attraction) gradually draws up the fluid to the point where
combustion
240
takes place; for you must have observed that the wick does not burn
quite to the bottom.
CAROLINE.
Yes; but I do not understand why it does not.
MRS. B.
Because the air has not so free an access to that part of the wick
which is immediately in contact with the candle, as to the part just
above, so that the heat there is not sufficient to produce its
decomposition; the combustion therefore begins a little above this
point.
CAROLINE.
But, Mrs. B., in those beautiful lights, called gas-lights,
which are now seen in many streets, and will, I hope, be soon
adopted every where,
I can perceive no wick at all. How are these lights
managed?
MRS. B.
I am glad you have put me in mind of saying a few words on this very
useful and interesting improvement. In this mode of lighting, the gas is
conveyed to the extremity of a tube, where it is kindled, and burns as
long as the supply continues. There is, therefore, no occasion for a
wick, or any other fuel whatever.
241
EMILY.
But how is all this gas procured in such large quantities?
MRS. B.
It is obtained from coal, by distillation.—Coal, when exposed
to heat in a close vessel, is decomposed; and hydrogen, which is one of
its constituents, rises in the state of gas, combined with another of
its component parts, carbon, forming a compound gas, called
Hydrocarbonat, the nature of which we shall again have an
opportunity of noticing when we treat of carbon. This gas, like
hydrogen, is perfectly transparent, invisible, and highly inflammable;
and in burning it emits that vivid light which you have so often
observed.
CAROLINE.
And does the process for procuring it require nothing but heating the
coals, and conveying the gas through tubes?
MRS. B.
Nothing else; except that the gas must be made to pass, immediately
at its formation, through two or three large vessels of water, in which
it deposits some other ingredients, and especially water, tar, and oil,
which also arise from the distillation of coals. The gas-light
apparatus, therefore, consists simply in a large iron vessel, in which
the coals are exposed to the heat of a furnace,—some reservoirs
242
of water, in which the gas deposits its impurities,—and tubes that
convey it to the desired spot, being propelled with uniform velocity
through the tubes by means of a certain degree of pressure which is made
upon the reservoir.
EMILY.
What an admirable contrivance! Do you not think, Mrs. B., that
it will soon get into universal use?
MRS. B.
Most probably, as to the lighting of streets, offices, and public
places, as it far surpasses any former invention for that purpose; but
as to the interior of private houses, this mode of lighting has not yet
been sufficiently tried to know whether it will be found generally
desirable, either in regard to economy or convenience. It may, however,
be considered as one of the happiest applications of chemistry to the
comforts of life; and there is every reason to suppose that it will
answer the full extent of public, expectation.
I have another experiment to show you with hydrogen gas, which I
think will entertain you. Have you ever blown bubbles with soap and
water?
EMILY.
Yes, often, when I was a child; and I used to make them float in the
air by blowing them upwards.
243
MRS. B.
We shall fill some such bubbles with hydrogen gas, instead of
atmospheric air, and you will see with what ease and rapidity they will
ascend, without the assistance of blowing, from the lightness of the
gas.—Will you mix some soap and water whilst I fill this bladder
with the gas contained in the receiver which stands on the shelf in the
water-bath?
CAROLINE.
What is the use of the brass-stopper and turn-cock at the top of the
receiver?
MRS. B.
It is to afford a passage to the gas when required. There is, you
see, a similar stop-cock fastened to this bladder, which is made to
fit that on the receiver. I screw them one on the other, and now
turn the two cocks, to open a communication between the receiver and the
bladder; then, by sliding the receiver off the shelf, and gently sinking
it into the bath, the water rises in the receiver and forces the gas
into the bladder. (Plate
IX. fig. 1.)
Plate IX.
Vol. I. p. 228
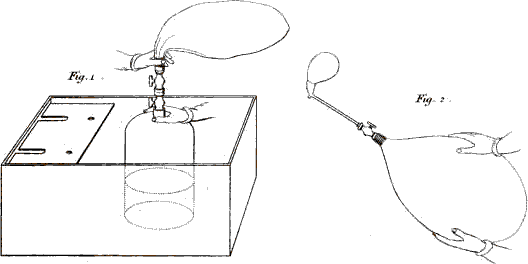
Fig. 1. Apparatus for transferring gases from a Receiver into a
bladder.
Fig. 2. Apparatus for blowing Soap bubbles.
Larger
view
CAROLINE.
Yes, I see the bladder swell as the water rises in the receiver.
244
MRS. B.
I think that we have already a sufficient quantity in the bladder for
our purpose; we must be careful to stop both the cocks before we
separate the bladder from the receiver, lest the gas should
escape.—Now I must fix a pipe to the stopper of the bladder, and
by dipping its mouth into the soap and water, take up a few
drops—then I again turn the cock, and squeeze the bladder in order
to force the gas into the soap and water at the mouth of the pipe. (Plate IX. fig. 2.)
EMILY.
There is a bubble—but it bursts before it leaves the mouth of
the pipe.
MRS. B.
We must have patience and try again; it is not so easy to blow
bubbles by means of a bladder, as simply with the breath.
CAROLINE.
Perhaps there is not soap enough in the water; I should have had
warm water, it would have dissolved the soap better.
EMILY.
Does not some of the gas escape between the bladder and the pipe?
245
MRS. B.
No, they are perfectly air tight; we shall succeed presently,
I dare say.
CAROLINE.
Now a bubble ascends; it moves with the rapidity of a balloon. How
beautifully it refracts the light!
EMILY.
It has burst against the ceiling—you succeed now wonderfully;
but why do they all ascend and burst against the ceiling?
MRS. B.
Hydrogen gas is so much lighter than atmospherical air, that it
ascends rapidly with its very light envelope, which is burst by the
force with which it strikes the ceiling.
Air-balloons are filled with this gas, and if they carried no other
weight than their covering, would ascend as rapidly as these
bubbles.
CAROLINE.
Yet their covering must be much heavier than that of these
bubbles?
MRS. B.
Not in proportion to the quantity of gas they contain. I do not
know whether you have ever
246
been present at the filling of a large balloon. The apparatus for that
purpose is very simple. It consists of a number of vessels, either jars
or barrels, in which the materials for the formation of the gas are
mixed, each of these being furnished with a tube, and communicating with
a long flexible pipe, which conveys the gas into the balloon.
EMILY.
But the fire-balloons which were first invented, and have been since
abandoned, on account of their being so dangerous, were constructed,
I suppose, on a different principle.
MRS. B.
They were filled simply with atmospherical air, considerably rarefied
by heat; and the necessity of having a fire underneath the balloon, in
order to preserve the rarefaction of the air within it, was the
circumstance productive of so much danger.
If you are not yet tired of experiments, I have another to show
you. It consists in filling soap-bubbles with a mixture of hydrogen and
oxygen gases, in the proportions that form water; and afterwards setting
fire to them.
EMILY.
They will detonate, I suppose?
247
MRS. B.
Yes, they will. As you have seen the method of transferring the gas
from the receiver into the bladder, it is not necessary to repeat it.
I have therefore provided a bladder which contains a due proportion
of oxygen and hydrogen gases, and we have only to blow bubbles
with it.
CAROLINE.
Here is a fine large bubble rising—shall I set fire to it with
the candle?
MRS. B.
If you please . . . .
CAROLINE.
Heavens, what an explosion!—It was like the report of a gun:
I confess it frightened me much. I never should have imagined
it could be so loud.
EMILY.
And the flash was as vivid as lightning.
MRS. B.
The combination of the two gases takes place during that instant of
time that you see the flash, and hear the detonation.
248
EMILY.
This has a strong resemblance to thunder and lightning.
MRS. B.
These phenomena, however, are generally of an electrical nature. Yet
various meteorological effects may be attributed to accidental
detonations of hydrogen gas in the atmosphere; for nature abounds with
hydrogen: it constitutes a very considerable portion of the whole mass
of water belonging to our globe, and from that source almost every other
body obtains it. It enters into the composition of all animal
substances, and of a great number of minerals; but it is most abundant
in vegetables. From this immense variety of bodies, it is often
spontaneously disengaged; its great levity makes it rise into the
superior regions of the atmosphere; and when, either by an electrical
spark, or any casual elevation of temperature, it takes fire, it may
produce such meteors or luminous appearances as are occasionally seen in
the atmosphere. Of this kind are probably those broad flashes which we
often see on a summer-evening, without hearing any detonation.
EMILY.
Every flash, I suppose, must produce a quantity of water?
249
CAROLINE.
And this water, naturally, descends in the form of rain?
MRS. B.
That probably is often the case, though it is not a necessary
consequence; for the water may be dissolved by the atmosphere, as it
descends towards the lower regions, and remain there in the form of
clouds.
The application of electrical attraction to chemical phenomena is
likely to lead to many very interesting discoveries in meteorology; for
electricity evidently acts a most important part in the atmosphere. This
subject however, is, as yet, not sufficiently developed for me to
venture enlarging upon it. The phenomena of the atmosphere are far from
being well understood; and even with the little that is known, I am
but imperfectly acquainted.
But before we take leave of hydrogen, I must not omit to mention to you
a most interesting discovery of Sir H. Davy, which is connected with
this subject.
CAROLINE.
You allude, I suppose, to the new miner’s lamp, which has of late
been so much talked of? I have long been desirous of knowing what
that discovery was, and what purpose it was intended to answer.
250
MRS. B.
It often happens in coal-mines, that quantities of the gas, called by
chemists hydro-carbonat, or by the miners fire-damp, (the
same from which the gas-lights are obtained,) ooze out from fissures in
the beds of coal, and fill the cavities in which the men are at work;
and this gas being inflammable, the consequence is, that when the men
approach those places with a lighted candle, the gas takes fire, and
explosions happen which destroy the men and horses employed in that part
of the colliery, sometimes in great numbers.
EMILY.
What tremendous accidents these must be! But whence does that gas
originate?
MRS. B.
Being the chief product of the combustion of coal, no wonder that
inflammable gas should occasionally appear in situations in which this
mineral abounds, since there can be no doubt that processes of
combustion are frequently taking place at a great depth under the
surface of the earth; and therefore those accumulations of gas may arise
either from combustions actually going on, or from former combustions,
the gas having perhaps been confined there for ages.
251
CAROLINE.
And how does Sir H. Davy’s lamp prevent those dreadful
explosions?
MRS. B.
By a contrivance equally simple and ingenious; and one which does no
less credit to the philosophical views from which it was deduced, than
to the philanthropic motives from which the enquiry sprung. The
principle of the lamp is shortly this: It was ascertained, two or three
years ago, both by Mr. Tennant and by Sir Humphry himself, that the
combustion of inflammable gas could not be propagated through small
tubes; so that if a jet of an inflammable gaseous mixture, issuing from
a bladder or any other vessel, through a small tube, be set fire to, it
burns at the orifice of the tube, but the flame never penetrates into
the vessel. It is upon this fact that Sir Humphry’s safety-lamp is
founded.
EMILY.
But why does not the flame ever penetrate through the tube into the
vessel from which the gas issues, so as to explode at once the whole of
the gas?
MRS. B.
Because, no doubt, the inflamed gas is so much cooled in its passage
through a small tube as to
252
cease to burn before the combustion reaches the reservoir.
CAROLINE.
And how can this principle be applied to the construction of a
lamp?
MRS. B.
Nothing easier. You need only suppose a lamp enclosed all round in
glass or horn, but having a number of small open tubes at the bottom,
and others at the top, to let the air in and out. Now, if such a lamp or
lanthorn be carried into an atmosphere capable of exploding, an
explosion or combustion of the gas will take place within the lamp; and
although the vent afforded by the tubes will save the lamp from
bursting, yet, from the principle just explained, the combustion will
not be propagated to the external air through the tubes, so that no
farther consequence will ensue.
EMILY.
And is that all the mystery of that valuable lamp?
MRS. B.
No; in the early part of the enquiry a lamp of this kind was actually
proposed; but it was but a rude sketch compared to its present state of
improvement. Sir H. Davy, after a succession of trials, by which he
brought his lamp nearer and nearer
253
to perfection, at last conceived the happy idea that if the lamp were
surrounded with a wire-work or wire-gauze, of a close texture, instead
of glass or horn, the tubular contrivance I have just described would be
entirely superseded, since each of the interstices of the gauze would
act as a tube in preventing the propagation of explosions; so that this
pervious metallic covering would answer the various purposes of
transparency, of permeability to air, and of protection against
explosion. This idea, Sir Humphry immediately submitted to the test of
experiment, and the result has answered his most sanguine expectations,
both in his laboratory and in the collieries, where it has already been
extensively tried. And he has now the happiness of thinking that his
invention will probably be the means of saving every year a number of
lives, which would have been lost in digging out of the bowels of the
earth one of the most valuable necessaries of life. Here is one of these
lamps, every part of which you will at once comprehend. (See Plate X. fig. 1.)
Plate X.
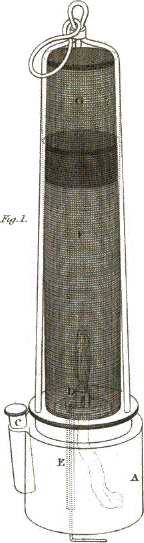
Fig. 1.
A. the cistern containing the Oil
B. the rim or screw by which the gauze cage is fixed to the cistern.
C. apperture for supplying Oil.
E. a wire for trimming the wick.
D. F. the wire gauze cylinder.
G. a double top.
Larger view
(complete Plate)
CAROLINE.
How very simple and ingenious! But I do not yet well see why an
explosion taking place within the lamp should not communicate to the
external air around it, through the interstices of the wire?
254
MRS. B.
This has been and is still a subject of wonder, even to philosophers;
and the only mode they have of explaining it is, that flame or ignition
cannot pass through a fine wire-work, because the metallic wire cools
the flame sufficiently to extinguish it in passing through the gauze.
This property of the wire-gauze is quite similar to that of the tubes
which I mentioned on introducing the subject; for you may consider each
interstice of the gauze as an extremely short tube of a very small
diameter.
EMILY.
But I should expect the wire would often become red-hot, by the
burning of the gas within the lamp?
MRS. B.
And this is actually the case, for the top of the lamp is very apt to
become red-hot. But, fortunately, inflammable gaseous mixtures cannot be
exploded by red-hot wire, the intervention of actual flame being
required for that purpose; so that the wire does not set fire to the
explosive gas around it.
EMILY.
I can understand that; but if the wire be red-hot, how can it cool
the flame within, and prevent its passing through the gauze?
255
MRS. B.
The gauze, though red-hot, is not so hot as the flame by which it has
been heated; and as metallic wire is a good conductor, the heat does not
much accumulate in it, as it passes off quickly to the other parts of
the lamp, as well as to any contiguous bodies.
CAROLINE.
This is indeed a most interesting discovery, and one which shows at
once the immense utility with which science may be practically applied
to some of the most important purposes.
256

MRS. B.
Sulphur is the next substance that
comes under our consideration. It differs in one essential point from
the preceding, as it exists in a solid form at the temperature of the
atmosphere.
CAROLINE.
I am glad that we have at last a solid body to examine; one that we
can see and touch. Pray, is it not with sulphur that the points of
matches are covered, to make them easily kindle?
MRS. B.
Yes, it is; and you therefore already know that sulphur is a very combustible
substance. It is seldom discovered in nature in a pure unmixed state; so
great is its affinity for other substances, that it is almost constantly
found combined with some of them. It is most commonly united with
257
metals, under various forms, and is separated from them by a very simple
process. It exists likewise in many mineral waters, and some vegetables
yield it in various proportions, especially those of the cruciform
tribe. It is also found in animal matter; in short, it may be discovered
in greater or less quantity, in the mineral, vegetable, and animal
kingdoms.
EMILY.
I have heard of flowers of sulphur, are they the produce of
any plant?
MRS. B.
By no means: they consist of nothing more than common sulphur,
reduced to a very fine powder by a process called
sublimation.—You see some of it in this phial; it is
exactly the same substance as this lump of sulphur, only its colour is a
paler yellow, owing to its state of very minute division.
EMILY.
Pray what is sublimation?
MRS. B.
It is the evaporation, or, more properly speaking, the volatilisation
of solid substances, which, in cooling, condense again in a concrete
form.
258
The process, in this instance, must be performed in a closed vessel,
both to prevent combustion, which would take place if the access of air
were not carefully precluded, and likewise in order to collect the
substance after the operation. As it is rather a slow process, we shall
not try the experiment now; but you will understand it perfectly if I
show you the apparatus used for the purpose. (Plate XI. fig. 1.) Some lumps of sulphur are
put into a receiver of this kind, which is called a cucurbit. Its
shape, you see, somewhat resembles that of a pear, and is open at the
top, so as to adapt itself exactly to a kind of conical receiver of this
sort, called the head. The cucurbit, thus covered with its head, is
placed over a sand-bath; this is nothing more than a vessel full of
sand, which is kept heated by a furnace, such as you see here, so as to
preserve the apparatus in a moderate and uniform temperature. The
sulphur then soon begins to melt, and immediately after this,
a thick white smoke rises, which is gradually deposited within the
head, or upper part of the apparatus, where it condenses against the
sides, somewhat in the form of a vegetation, whence it has obtained the
name of flowers of sulphur. This apparatus, which is called an
alembic, is highly useful in all kinds of distillations, as you
will see when we come to treat of those operations. Alembics are not
commonly
259
made of glass, like this, which is applicable only to distillations upon
a very small scale. Those used in manufactures are generally made of
copper, and are, of course, considerably larger. The principal
construction, however, is always the same, although their shape admits
of some variation.
Plate XI.
Vol. I. p. 237.
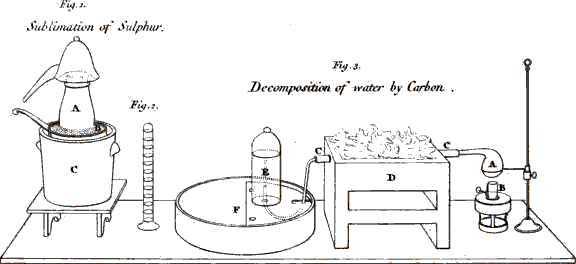
Fig. 1.
A Alembic.
B Sand-bath.
C Furnace.
Fig. 2. Eudiometer.
Fig. 3.
A Retort containing water.
B Lamp to heat the water.
C.C Porcelain tube containing Carbone.
D Furnace through which the tube passes.
E Receiver for the gas produced.
F Water bath.
Larger
view
CAROLINE.
What is the use of that neck, or tube, which bends down from the
upper piece of the apparatus?
MRS. B.
It is of no use in sublimations; but in distillations (the general
object of which is to evaporate, by heat, in closed vessels, the
volatile parts of a compound body, and to condense them again into a
liquid,) it serves to carry off the condensed fluid, which otherwise
would fall back into the cucurbit. But this is rather foreign to our
present subject. Let us return to the sulphur. You now perfectly
understand, I suppose, what is meant by sublimation?
EMILY.
I believe I do. Sublimation appears to consist in destroying, by
means of heat, the attraction of aggregation of the particles of a solid
body, which are thus volatilised; and as soon as they lose the
260
caloric which produced that effect, they are deposited in the form of a
fine powder.
CAROLINE.
It seems to me to be somewhat similar to the transformation of water
into vapour, which returns to its liquid state when deprived of
caloric.
EMILY.
There is this difference, however, that the sulphur does not return
to its former state, since, instead of lumps, it changes to a fine
powder.
MRS. B.
Chemically speaking, it is exactly the same substance, whether in the
form of lump or powder. For if this powder be melted again by heat, it
will, in cooling, be restored to the same solid state in which it was
before its sublimation.
CAROLINE.
But if there be no real change, produced by the sublimation of the
sulphur, what is the use of that operation?
MRS. B.
It divides the sulphur into very minute parts, and thus disposes it
to enter more readily into combination with other bodies. It is used
also as a means of purification.
261
CAROLINE.
Sublimation appears to me like the beginning of combustion, for the
completion of which one circumstance only is wanting, the absorption of
oxygen.
MRS. B.
But that circumstance is every thing. No essential alteration is
produced in sulphur by sublimation; whilst in combustion it combines
with the oxygen, and forms a new compound totally different in every
respect from sulphur in its pure state.—We shall now burn
some sulphur, and you will see how very different the result will be.
For this purpose I put a small quantity of flowers of sulphur into this
cup, and place it in a dish, into which I have poured a little water:
I now set fire to the sulphur with the point of this hot wire; for
its combustion will not begin unless its temperature be considerably
raised.—You see that it burns with a faint blueish flame; and as I
invert over it this receiver, white fumes arise from the sulphur, and
fill the vessel.—You will soon perceive that the water is rising
within the receiver, a little above its level in the
plate.—Well, Emily, can you account for this?
EMILY.
I suppose that the sulphur has absorbed the oxygen from the
atmospherical air within the receiver, and that we shall find some
oxygenated
262
sulphur in the cup. As for the white smoke, I am quite at a loss to
guess what it may be.
MRS. B.
Your first conjecture is very right: but you are mistaken in the
last; for nothing will be left in the cup. The white vapour is the
oxygenated sulphur, which assumes the form of an elastic fluid of a
pungent and offensive smell, and is a powerful acid. Here you see a
chemical combination of oxygen and sulphur, producing a true gas, which
would continue such under the pressure and at the temperature of the
atmosphere, if it did not unite with the water in the plate, to which it
imparts its acid taste, and all its acid properties.—You see, now,
with what curious effects the combustion of sulphur is attended.
CAROLINE.
This is something quite new; and I confess that I do not perfectly
understand why the sulphur turns acid.
MRS. B.
It is because it unites with oxygen, which is the acidifying
principle. And, indeed, the word oxygen is derived from two Greek
words signifying to produce an acid.
CAROLINE.
Why, then, is not water, which contains such a quantity of oxygen,
acid?
263
MRS. B.
Because hydrogen, which is the other constituent of water, is not
susceptible of acidification.—I believe it will be necessary,
before we proceed further, to say a few words of the general nature of
acids, though it is rather a deviation from our plan of examining the
simple bodies separately, before we consider them in a state of
combination.
Acids may be considered as a peculiar class of burnt bodies,
which during their combustion, or combination with oxygen, have acquired
very characteristic properties. They are chiefly discernible by their
sour taste, and by turning red most of the blue vegetable colours. These
two properties are common to the whole class of acids; but each of them
is distinguished by other peculiar qualities. Every acid consists of
some particular substance, (which constitutes its basis, and is
different in each,) and of oxygen, which is common to them all.
EMILY.
But I do not clearly see the difference between acids and oxyds.
MRS. B.
Acids were, in fact, oxyds, which, by the addition of a sufficient
quantity of oxygen, have been converted into acids. For acidification,
you must observe, always implies previous oxydation, as a body must have
combined with the quantity of
264
oxygen requisite to constitute it an oxyd, before it can combine with
the greater quantity that is necessary to render it an acid.
CAROLINE.
Are all oxyds capable of being converted into acids?
MRS. B.
Very far from it; it is only certain substances which will enter into
that peculiar kind of union with oxygen that produces acids, and the
number of these is proportionally very small; but all burnt bodies may
be considered as belonging either to the class of oxyds, or to that of
acids. At a future period, we shall enter more at large into this
subject. At present, I have but one circumstance further to point
out to your observation respecting acids: it is, that most of them are
susceptible of two degrees of acidification, according to the different
quantities of oxygen with which their basis combines.
EMILY.
And how are these two degrees of acidification distinguished?
MRS. B.
By the peculiar properties which result from them. The acid we have
just made is the first or weakest degree of acidification, and is called
sulphureous acid; if it were fully saturated with oxygen,
265
it would be called sulphuric acid. You must therefore remember,
that in this, as in all acids, the first degree of acidification is
expressed by the termination in ous; the stronger, by the
termination in ic.
CAROLINE.
And how is the sulphuric acid made?
MRS. B.
By burning sulphur in pure oxygen gas, and thus rendering its
combustion much more complete. I have provided some oxygen gas for
this purpose; it is in that bottle, but we must first decant the gas
into the glass receiver which stands on the shelf in the bath, and is
full of water.
CAROLINE.
Pray, let me try to do it, Mrs. B.
MRS. B.
It requires some little dexterity—hold the bottle completely
under water, and do not turn the mouth upwards, till it is immediately
under the aperture in the shelf, through which the gas is to pass into
the receiver, and then turn it up gradually.—Very well, you have
only let a few bubbles escape, and that must be expected at a first
trial.—Now I shall put this piece of sulphur into the receiver,
through the opening at the top, and
266
introduce along with it a small piece of lighted tinder to set fire to
it.—This requires being done very quickly, lest the atmospherical
air should get in, and mix with the pure oxygen gas.
EMILY.
How beautifully it burns!
CAROLINE.
But it is already buried in the thick vapour. This, I suppose,
is sulphuric acid?
EMILY.
Are these acids always in a gaseous state?
MRS. B.
Sulphureous acid, as we have already observed, is a permanent gas,
and can be obtained in a liquid form only by condensing it in water. In
its pure state, the sulphureous acid is invisible, and it now appears in
the form of a white smoke, from its combining with the moisture. But the
vapour of sulphuric acid, which you have just seen to rise during the
combustion, is not a gas, but only a vapour, which condenses into liquid
sulphuric acid, by losing its caloric. But it appears from Sir H. Davy’s
experiments, that this formation and condensation of sulphuric acid
requires the presence of water, for which purpose the vapour is received
267
into cold water, which may afterwards be separated from the acid by
evaporation.
Sulphur has hitherto been considered as a simple substance; but Sir
H. Davy has suspected that it contains a small portion of hydrogen, and
perhaps also of oxygen.
On submitting sulphur to the action of the Voltaic battery, he
observed that the negative wire gave out hydrogen; and the existence of
hydrogen in sulphur was rendered still more probable by his observing
that a small quantity of water was produced during the combustion of
sulphur.
EMILY.
And pray of what nature is sulphur when perfectly pure?
MRS. B.
Sulphur has probably never been obtained perfectly free from
combination, so that its radical may possibly possess properties very
different from those of common sulphur. It has been suspected to be of a
metallic nature; but this is mere conjecture.
Before we quit the subject of sulphur, I must tell you that it is
susceptible of combining with a great variety of substances, and
especially with hydrogen, with which you are already acquainted.
Hydrogen gas can dissolve a small portion of it.
268
EMILY.
What! can a gas dissolve a solid substance?
MRS. B.
Yes; a solid substance may be so minutely divided by heat, as to
become soluble in a gas: and there are several instances of it. But you
must observe, that, in this case, a chemical union or combination
of the sulphur with the hydrogen gas is produced. In order to effect
this, the sulphur must be strongly heated in contact with the gas; the
heat reduces the sulphur to such a state of extreme division, and
diffuses it so thoroughly through the gas, that they combine and
incorporate together. And as a proof that there must be a chemical union
between the sulphur and the gas, it is sufficient to remark that they
are not separated when the sulphur loses the caloric by which it was
volatilized. Besides, it is evident, from the peculiar fetid smell of
this gas, that it is a new compound totally different from either of its
constituents; it is called sulphuretted hydrogen gas, and is
contained in great abundance in sulphureous mineral waters.
CAROLINE.
Are not the Harrogate waters of this nature?
MRS. B.
Yes; they are naturally impregnated with sulphuretted
269
hydrogen gas, and there are many other springs of the same kind, which
shows that this gas must often be formed in the bowels of the earth by
spontaneous processes of nature.
CAROLINE.
And could not such waters be made artificially by impregnating common
water with this gas?
MRS. B.
Yes; they can be so well imitated, as perfectly to resemble the
Harrogate waters.
Sulphur combines likewise with phosphorus, and with the alkalies, and
alkaline earths, substances with which you are yet unacquainted. We
cannot, therefore, enter into these combinations at present. In our next
lesson we shall treat of phosphorus.
EMILY.
May we not begin that subject to-day; this lesson has been so
short?
MRS. B.
I have no objection, if you are not tired. What do you say,
Caroline?
CAROLINE.
I am as desirous as Emily of prolonging the lesson to-day, especially
as we are to enter on a new
270
subject; for I confess that sulphur has not appeared to me so
interesting as the other simple bodies.
MRS. B.
Perhaps you may find phosphorus more entertaining. You must not,
however, be discouraged when you meet with some parts of a study less
amusing than others; it would answer no good purpose to select the most
pleasing parts, since, if we did not proceed with some method, in order
to acquire a general idea of the whole, we could scarcely expect to take
interest in any particular subjects.
PHOSPHORUS.
Phosphorus is considered as a simple
body; though, like sulphur, it has been suspected of containing
hydrogen. It was not known by the earlier chemists. It was first
discovered by Brandt, a chemist of Hamburgh, whilst employed in
researches after the philosopher’s stone; but the method of obtaining it
remained a secret till it was a second time discovered both by Kunckel
and Boyle, in the year 1680. You see a specimen of phosphorus in this
phial; it is generally moulded into small sticks of a yellowish colour,
as you find it here.
271
CAROLINE.
I do not understand in what the discovery consisted; there may be a
secret method of making an artificial composition, but how can you talk
of making a substance which naturally exists?
MRS. B.
A body may exist in nature so closely combined with other substances,
as to elude the observation of chemists, or render it extremely
difficult to obtain it in its separate state. This is the case with
phosphorus, which is always so intimately combined with other
substances, that its existence remained unnoticed till Brandt discovered
the means of obtaining it free from other combinations. It is found in
all animal substances, and is now chiefly extracted from bones, by a
chemical process. It exists also in some plants, that bear a strong
analogy to animal matter in their chemical composition.
EMILY.
But is it never found in its pure separate state?
MRS. B.
Never, and this is the reason that it has remained so long
undiscovered.
Phosphorus is eminently combustible; it melts and takes fire at the
temperature of one hundred
272
degrees, and absorbs in its combustion nearly once and a half its own
weight of oxygen.
CAROLINE.
What! will a pound of phosphorus consume a pound and half of
oxygen?
MRS. B.
So it appears from accurate experiments. I can show you with
what violence it combines with oxygen, by burning some of it in that
gas. We must manage the experiment in the same manner as we did the
combustion of sulphur. You see I am obliged to cut this little bit of
phosphorus under water, otherwise there would be danger of its taking
fire by the heat of my fingers. I now put into the receiver, and kindle it by means of a
hot wire.
EMILY.
What a blaze! I can hardly look at it. I never saw any thing so
brilliant. Does it not hurt your eyes, Caroline?
CAROLINE.
Yes; but still I cannot help looking at it. A prodigious
quantity of oxygen must indeed be absorbed, when so much light and
caloric are disengaged!
273
MRS. B.
In the combustion of a pound of phosphorus, a sufficient
quantity of caloric is set free to melt upwards of a hundred pounds of
ice; this has been computed by direct experiments with the
calorimeter.
EMILY.
And is the result of this combustion, like that of sulphur, an
acid?
MRS. B.
Yes; phosphoric acid. And had we duly proportioned the phosphorus and
the oxygen, they would have been completely converted into phosphoric
acid, weighing together, in this new state, exactly the sum of their
weights separately. The water would have ascended into the receiver, on
account of the vacuum formed, and would have filled it entirely. In this
case, as in the combustion of sulphur, the acid vapour formed is
absorbed and condensed in the water of the receiver. But when this
combustion is performed without any water or moisture being present, the
acid then appears in the form of concrete whitish flakes, which are,
however, extremely ready to melt upon the least admission of
moisture.
EMILY.
Does phosphorus, in burning in atmospherical
274
air, produce, like sulphur, a weaker sort of the same acid?
MRS. B.
No: for it burns in atmospherical air, nearly at the same temperature
as in pure oxygen gas; and it is in both cases so strongly disposed to
combine with the oxygen, that the combustion is perfect, and the product
similar; only in atmospherical air, being less rapidly supplied with
oxygen, the process is performed in a slower manner.
CAROLINE.
But is there no method of acidifying phosphorus in a slighter manner,
so as to form phosphorus acid?
MRS. B.
Yes, there is. When simply exposed to the atmosphere, phosphorus
undergoes a kind of slow combustion at any temperature above zero.
EMILY.
But is not the process in this case rather an oxydation than a
combustion? For if the oxygen is too slowly absorbed for a sensible
quantity of light and heat to be disengaged, it is not a true
combustion.
MRS. B.
The case is not as you suppose: a faint light is
275
emitted which is very discernible in the dark; but the heat evolved is
not sufficiently strong to be sensible: a whitish vapour arises
from this combustion, which, uniting with water, condenses into liquid
phosphorus acid.
CAROLINE.
Is it not very singular that phosphorus should burn at so low a
temperature in atmospherical air, whilst it does not burn in pure oxygen
without the application of heat?
MRS. B.
So it at first appears. But this circumstance seems to be owing to
the nitrogen gas of the atmosphere. This gas dissolves small particles
of phosphorus, which being thus minutely divided and diffused in the
atmospherical air, combines with the oxygen, and undergoes this slow
combustion. But the same effect does not take place in oxygen gas,
because it is not capable of dissolving phosphorus; it is therefore
necessary, in this case, that heat should be applied to effect that
division of particles, which, in the former instance, is produced by the
nitrogen.
EMILY.
I have seen letters written with phosphorus, which are invisible by
day-light, but may be read
276
in the dark by their own light. They look as if they were written with
fire; yet they do not seem to burn.
MRS. B.
But they do really burn; for it is by their slow combustion that the
light is emitted; and phosphorus acid is the result of this
combustion.
Phosphorus is sometimes used as a test to estimate the purity of
atmospherical air. For this purpose, it is burnt in a graduated tube,
called an Eudiometer (Plate XI. fig. 2.), and from the quantity of air
which the phosphorus absorbs, the proportion of oxygen in the air
examined is deduced; for the phosphorus will absorb all the oxygen, and
the nitrogen alone will remain.
EMILY.
And the more oxygen is contained in the atmosphere, the purer,
I suppose, it is esteemed?
MRS. B.
Certainly. Phosphorus, when melted, combines with a great variety of
substances. With sulphur it forms a compound so extremely combustible,
that it immediately takes fire on coming in contact with the air. It is
with this composition that phosphoric matches are prepared, which kindle
as soon as they are taken out of their case and are exposed to the
air.
277
EMILY.
I have a box of these curious matches; but I have observed, that in
very cold weather, they will not take fire without being previously
rubbed.
MRS. B.
By rubbing them you raise their temperature; for, you know, friction
is one of the means of extricating heat.
EMILY.
Will phosphorus combine with hydrogen gas, as sulphur does?
MRS. B.
Yes; and the compound gas which results from this combination has a
smell still more fetid than the sulphuretted hydrogen; it resembles that
of garlic.
The phosphoretted hydrogen gas has this remarkable
peculiarity, that it takes fire spontaneously in the atmosphere, at any
temperature. It is thus, probably, that are produced those transient
flames, or flashes of light, called by the vulgar Will-of-the
Whisp, or more properly Ignes-fatui, which are often seen in
church-yards, and places where the putrefactions of animal matter exhale
phosphorus and hydrogen gas.
CAROLINE.
Country people, who are so much frightened by
278
those appearances, would soon be reconciled to them, if they knew from
what a simple cause they proceed.
MRS. B.
There are other combinations of phosphorus that have also very
singular properties, particularly that which results from its union with
lime.
EMILY.
Is there any name to distinguish the combination of two substances,
like phosphorus and lime, neither of which are oxygen, and which cannot
therefore produce either an oxyd or an acid?
MRS. B.
The names of such combinations are composed from those of their
ingredients, merely by a slight change in their termination. Thus the
combination of sulphur with lime is called a sulphuret, and that
of phosphorus, a phosphuret of lime. This latter compound,
I was going to say, has the singular property of decomposing water,
merely by being thrown into it. It effects this by absorbing the oxygen
of water, in consequence of which bubbles of hydrogen gas ascend,
holding in solution a small quantity of phosphorus.
279
EMILY.
These bubbles then are phosphoretted hydrogen gas?
MRS. B.
Yes; and they produce the singular appearance of a flash of fire
issuing from water, as the bubbles kindle and detonate on the surface of
the water, at the instant that they come in contact with the
atmosphere.
CAROLINE.
Is not this effect nearly similar to that produced by the combination
of phosphorus and sulphur, or, more properly speaking, the phosphuret
of sulphur?
MRS. B.
Yes; but the phenomenon appears more extraordinary in this case, from
the presence of water, and from the gaseous form of the combustible
compound. Besides, the experiment surprises by its great simplicity. You
only throw a piece of phosphoret of lime into a glass of water, and
bubbles of fire will immediately issue from it.
CAROLINE.
Cannot we try the experiment?
280
MRS. B.
Very easily: but we must do it in the open air; for the smell of the
phosphorated hydrogen gas is so extremely fetid, that it would be
intolerable in the house. But before we leave the room, we may produce,
by another process, some bubbles of the same gas, which are much less
offensive.
There is in this little glass retort a solution of potash in water;
I add to it a small piece of phosphorus. We must now heat the
retort over the lamp, after having engaged its neck under
water—you see it begins to boil; in a few minutes bubbles will
appear, which take fire and detonate as they issue from the water.
CAROLINE.
There is one—and another. How curious it is!—But I do not
understand how this is produced.
MRS. B.
It is the consequence of a display of affinities too complicated,
I fear, to be made perfectly intelligible to you at present.
In a few words, the reciprocal action of the potash, phosphorus,
caloric, and water are such, that some of the water is decomposed, and
the hydrogen gas thereby formed carries off some minute particles of
phosphorus, with which it forms phosphoretted
281
hydrogen gas, a compound which spontaneously takes fire at almost
any temperature.
EMILY.
What is that circular ring of smoke which slowly rises from each
bubble after its detonation?
MRS. B.
It consists of water and phosphoric acid in vapour, which are
produced by the combustion of hydrogen and phosphorus.
282

CAROLINE.
To-day, Mrs. B., I believe we are to
learn the nature and properties of CARBON. This substance is quite new to me;
I never heard it mentioned before.
MRS. B.
Not so new as you imagine; for carbon is nothing more than charcoal
in a state of purity, that is to say, unmixed with any foreign
ingredients.
CAROLINE.
But charcoal is made by art, Mrs. B., and a body consisting of one
simple substance cannot be fabricated?
MRS. B.
You again confound the idea, of making a simple body, with that of
separating it from a compound. The chemical processes by which a simple
body is obtained in a state of purity, consist in unmaking the
compound in which it is contained,
283
in order to separate from it the simple substance in question. The
method by which charcoal is usually obtained, is, indeed, commonly
called making it; but, upon examination, you will find this
process to consist simply in separating it from other substances with
which it is found combined in nature.
Carbon forms a considerable part of the solid matter of all organised
bodies; but it is most abundant in the vegetable creation, and it is
chiefly obtained from wood. When the oil and water (which are other
constituents of vegetable matter) are evaporated, the black, porous,
brittle substance that remains, is charcoal.
CAROLINE.
But if heat be applied to the wood in order to evaporate the oil and
water, will not the temperature of the charcoal be raised so as to make
it burn; and if it combines with oxygen, can we any longer call it
pure?
MRS. B.
I was going to say, that, in this operation, the air must be
excluded.
CAROLINE.
How then can the vapour of the oil and water fly off?
284
MRS. B.
In order to produce charcoal in its purest state (which is, even
then, but a less imperfect sort of carbon), the operation should be
performed in an earthen retort. Heat being applied to the body of the
retort, the evaporable part of the wood will escape through its neck,
into which no air can penetrate as long as the heated vapour continues
to fill it. And if it be wished to collect these volatile products of
the wood, this can easily be done by introducing the neck of the retort
into the water-bath apparatus, with which you are acquainted. But the
preparation of common charcoal, such as is used in kitchens and
manufactures, is performed on a much larger scale, and by an easier and
less expensive process.
EMILY.
I have seen the process of making common charcoal. The wood is ranged
on the ground in a pile of a pyramidical form, with a fire underneath;
the whole is then covered with clay, a few holes only being left
for the circulation of air.
MRS. B.
These holes are closed as soon as the wood is fairly lighted, so that
the combustion is checked, or at least continues but in a very imperfect
manner; but the heat produced by it is sufficient to
285
force out and volatilize, through the earthy cover, most part of the
oily and watery principles of the wood, although it cannot reduce it to
ashes.
EMILY.
Is pure carbon as black as charcoal?
MRS. B.
The purest charcoal we can prepare is so; but chemists have never yet
been able to separate it entirely from hydrogen. Sir H. Davy says, that
the most perfect carbon that is prepared by art contains about five per
cent. of hydrogen; he is of opinion, that if we could obtain it quite
free from foreign ingredients, it would be metallic, in common with
other simple substances.
But there is a form in which charcoal appears, that I dare say will
surprise you.—This ring, which I wear on my finger, owes its
brilliancy to a small piece of carbon.
CAROLINE.
Surely, you are jesting, Mrs. B.?
EMILY.
I thought your ring was diamond?
MRS. B.
It is so. But diamond is nothing more than carbon in a crystallized
state.
286
EMILY.
That is astonishing! Is it possible to see two things apparently more
different than diamond and charcoal?
CAROLINE.
It is, indeed, curious to think that we adorn ourselves with jewels
of charcoal!
MRS. B.
There are many other substances, consisting chiefly of carbon, that
are remarkably white. Cotton, for instance, is almost wholly carbon.
CAROLINE.
That, I own, I could never have imagined!—But pray,
Mrs. B., since it is known of what substance diamond and cotton are
composed, why should they not be manufactured, or imitated, by some
chemical process, which would render them much cheaper, and more
plentiful than the present mode of obtaining them?
MRS. B.
You might as well, my dear, propose that we should make flowers and
fruit, nay, perhaps even animals, by a chemical process; for it is known
of what these bodies consist, since every thing which we are acquainted
with in nature is formed from the various simple substances that we have
287
enumerated. But you must not suppose that a knowledge of the component
parts of a body will in every case enable us to imitate it. It is much
less difficult to decompose bodies, and discover of what materials they
are made, than it is to recompose them. The first of these processes is
called analysis, the last synthesis. When we are able to
ascertain the nature of a substance by both these methods, so that the
result of one confirms that of the other, we obtain the most complete
knowledge of it that we are capable of acquiring. This is the case with
water, with the atmosphere, with most of the oxyds, acids, and neutral
salts, and with many other compounds. But the more complicated
combinations of nature, even in the mineral kingdom, are in general
beyond our reach, and any attempt to imitate organised bodies must ever
prove fruitless; their formation is a secret that rests in the bosom of
the Creator. You see, therefore, how vain it would be to attempt to make
cotton by chemical means. But, surely, we have no reason to regret our
inability in this instance, when nature has so clearly pointed out a
method of obtaining it in perfection and abundance.
CAROLINE.
I did not imagine that the principle of life could be imitated by the
aid of chemistry; but it did not appear to me ridiculous to suppose that
chemists
288
might attain a perfect imitation of inanimate nature.
MRS. B.
They have succeeded in this point in a variety of instances; but, as
you justly observe, the principle of life, or even the minute and
intimate organisation of the vegetable kingdom, are secrets that have
almost entirely eluded the researches of philosophers; nor do I imagine
that human art will ever be capable of investigating them with complete
success.
EMILY.
But diamond, since it consists of one simple unorganised substance,
might be, one would think, perfectly imitable by art?
MRS. B.
It is sometimes as much beyond our power to obtain a simple body in a
state of perfect purity, as it is to imitate a complicated combination;
for the operations by which nature separates bodies are frequently as
inimitable as those which she uses for their combination. This is the
case with carbon; all the efforts of chemists to separate it entirely
from other substances have been fruitless, and in the purest state in
which it can be obtained by art, it still retains a portion of hydrogen,
and probably of some other foreign ingredients. We are ignorant
289
of the means which nature employs to crystallize it. It may probably be
the work of ages, to purify, arrange, and unite the particles of carbon
in the form of diamond. Here is some charcoal in the purest state we can
procure it: you see that it is a very black, brittle, light, porous
substance, entirely destitute of either taste or smell. Heat, without
air, produces no alteration in it, as it is not volatile; but, on the
contrary, it invariably remains at the bottom of the vessel after all
the other parts of the vegetable are evaporated.
EMILY.
Yet carbon is, no doubt, combustible, since you say that charcoal
would absorb oxygen if air were admitted during its preparation?
CAROLINE.
Unquestionably. Besides, you know, Emily, how much it is used in
cooking. But pray what is the reason that charcoal burns without smoke,
whilst a wood fire smokes so much?
MRS. B.
Because, in the conversion of wood into charcoal, the volatile
particles of the former have been evaporated.
CAROLINE.
Yet I have frequently seen charcoal burn with
290
flame; therefore it must, in that case, contain some hydrogen.
MRS. B.
Very true; but you must recollect that charcoal, especially that
which is used for common purposes, is not perfectly pure. It generally
retains some remains of the various other component parts of vegetables,
and hydrogen particularly, which accounts for the flame in question.
CAROLINE.
But what becomes of the carbon itself during its combustion?
MRS. B.
It gradually combines with the oxygen of the atmosphere, in the same
way as sulphur and phosphorus, and, like those substances, it is
converted into a peculiar acid, which flies off in a gaseous form. There
is this difference, however, that the acid is not, in this instance, as
in the two cases just mentioned, a mere condensable vapour, but a
permanent elastic fluid, which always remains in the state of gas, under
any pressure and at any temperature. The nature of this acid was first
ascertained by Dr. Black, of Edinburgh; and, before the introduction of
the new nomenclature, it was called fixed air. It is now
distinguished by the more appropriate name of carbonic acid
gas.
291
EMILY.
Carbon, then, can be volatilized by burning, though, by heat alone,
no such effect is produced?
MRS. B.
Yes; but then it is no longer simple carbon, but an acid of which
carbon forms the basis. In this state, carbon retains no more appearance
of solidity or corporeal form, than the basis of any other gas. And you
may, I think, from this instance, derive a more clear idea of the
basis of the oxygen, hydrogen, and nitrogen gases, the existence of
which, as real bodies, you seemed to doubt, because they were not to be
obtained simply in a solid form.
EMILY.
That is true; we may conceive the basis of the oxygen, and of the
other gases, to be solid, heavy substances, like carbon; but so much
expanded by caloric as to become invisible.
CAROLINE.
But does not the carbonic acid gas partake of the blackness of
charcoal?
MRS. B.
Not in the least. Blackness, you know, does not appear to be
essential to carbon, and it is pure carbon, and not charcoal, that we
must consider
292
as the basis of carbonic acid. We shall make some carbonic acid, and, in
order to hasten the process, we shall burn the carbon in oxygen gas.
EMILY.
But do you mean then to burn diamond?
MRS. B.
Charcoal will answer the purpose still better, being softer and more
easy to inflame; besides the experiments on diamond are rather
expensive.
CAROLINE.
But is it possible to burn diamond?
MRS. B.
Yes, it is; and in order to effect this combustion, nothing more is
required than to apply a sufficient degree of heat by means of the
blow-pipe, and of a stream of oxygen gas. Indeed it is by burning
diamond that its chemical nature has been ascertained. It has long been
known as a combustible substance, but it is within these few years only
that the product of its combustion has been proved to be pure carbonic
acid. This remarkable discovery is due to Mr. Tennant.
Now let us try to make some carbonic acid.—Will you, Emily,
decant some oxygen gas from this large jar into the receiver in which we
are to
293
burn the carbon;
and I shall introduce this small piece of charcoal, with a little
lighted tinder, which will be necessary to give the first impulse to the
combustion.
EMILY.
I cannot conceive how so small a piece of tinder, and that but just
lighted, can raise the temperature of the carbon sufficiently to set
fire to it; for it can produce scarcely any sensible heat, and it hardly
touches the carbon.
MRS. B.
The tinder thus kindled has only heat enough to begin its own
combustion, which, however, soon becomes so rapid in the oxygen gas, as
to raise the temperature of the charcoal sufficiently for this to burn
likewise, as you see is now the case.
EMILY.
I am surprised that the combustion of carbon is not more brilliant;
it does not give out near so much light or caloric as phosphorus, or
sulphur. Yet since it combines with so much oxygen, why is not a
proportional quantity of light and heat disengaged from the
decomposition of the oxygen gas, and the union of its electricity with
that of the charcoal?
MRS. B.
It is not surprising that less light and heat should be liberated in
this than in almost any other combustion,
294
since the oxygen, instead of entering into a solid or liquid
combination, as it does in the phosphoric and sulphuric acids, is
employed in forming another elastic fluid; it therefore parts with less
of its caloric.
EMILY.
True; and, on second consideration, it appears, on the contrary,
surprising that the oxygen should, in its combination with carbon,
retain a sufficient portion of caloric to maintain both substances in a
gaseous state.
CAROLINE.
We may then judge of the degree of solidity in which oxygen is
combined in a burnt body, by the quantity of caloric liberated during
its combustion?
MRS. B.
Yes; provided that you take into the account the quantity of oxygen
absorbed by the combustible body, and observe the proportion which the
caloric bears to it.
CAROLINE.
But why should the water, after the combustion of carbon, rise in the
receiver, since the gas within it retains an aëriform state?
MRS. B.
Because the carbonic acid gas is gradually absorbed
295
by the water; and this effect would be promoted by shaking the
receiver.
EMILY.
The charcoal is now extinguished, though it is not nearly consumed;
it has such an extraordinary avidity for oxygen, I suppose, that
the receiver did not contain enough to satisfy the whole.
MRS. B.
That is certainly the case; for if the combustion were performed in
the exact proportions of 28 parts of carbon to 72 of oxygen, both these
ingredients would disappear, and 100 parts of carbonic acid would be
produced.
CAROLINE.
Carbonic acid must be a very strong acid, since it contains so great
a proportion of oxygen?
MRS. B.
That is a very natural inference; yet it is erroneous. For the
carbonic is the weakest of all the acids. The strength of an acid seems
to depend upon the nature of its basis, and its mode of combination, as
well as upon the proportion of the acidifying principle. The same
quantity of oxygen that will convert some bodies into strong acids, will
only be sufficient simply to oxydate others.
296
CAROLINE.
Since this acid is so weak, I think chemists should have called it
the carbonous, instead of the carbonic acid.
EMILY.
But, I suppose, the carbonous acid is still weaker, and is formed by
burning carbon in atmospherical air.
MRS. B.
It has been lately discovered, that carbon may be converted into a
gas, by uniting with a smaller proportion of oxygen; but as this gas
does not possess any acid properties, it is no more than an oxyd; it is
called gaseous oxyd of carbon.
CAROLINE.
Pray is not carbonic acid a very wholesome gas to breathe, as it
contains so much oxygen?
MRS. B.
On the contrary, it is extremely pernicious. Oxygen, when in a state
of combination with other substances, loses, in almost every instance,
its respirable properties, and the salubrious effects which it has on
the animal economy when in its unconfined state. Carbonic acid is not
only unfit for respiration, but extremely deleterious if taken into the
lungs.
297
EMILY.
You know, Caroline, how very unwholesome the fumes of burning
charcoal are reckoned.
CAROLINE.
Yes; but, to confess the truth, I did not consider that a charcoal
fire produced carbonic acid gas.—Can this gas be condensed into a
liquid?
MRS. B.
No: for, as I told you before, it is a permanent elastic fluid. But
water can absorb a certain quantity of this gas, and can even be
impregnated with it, in a very strong degree, by the assistance of
agitation and pressure, as I am going to show you. I shall decant
some carbonic acid gas into this bottle, which I fill first with water,
in order to exclude the atmospherical air; the gas is then introduced
through the water, which you see it displaces, for it will not mix with
it in any quantity, unless strongly agitated, or allowed to stand over
it for some time. The bottle is now about half full of carbonic acid
gas, and the other half is still occupied by the water. By corking the
bottle, and then violently shaking it, in this way, I can mix the
gas and water together.—Now will you taste it?
EMILY.
It has a distinct acid taste.
298
CAROLINE.
Yes, it is sensibly sour, and appears full of little bubbles.
MRS. B.
It possesses likewise all the other properties of acids, but, of
course, in a less degree than the pure carbonic acid gas, as it is so
much diluted by water.
This is a kind of artificial Seltzer water. By analysing that which
is produced by nature, it was found to contain scarcely any thing more
than common water impregnated with a certain proportion of carbonic acid
gas. We are, therefore, able to imitate it, by mixing those proportions
of water and carbonic acid. Here, my dear, is an instance, in which, by
a chemical process, we can exactly copy the operations of nature; for
the artificial Seltzer waters can be made in every respect similar to
those of nature; in one point, indeed, the former have an advantage,
since they may
be prepared stronger, or weaker, as occasion requires.
CAROLINE.
I thought I had tasted such water before. But what renders it so
brisk and sparkling?
MRS. B.
This sparkling, or effervescence, as it is called, is always
occasioned by the action of an elastic fluid escaping from a liquid; in
the artificial Seltzer
299
water, it is produced by the carbonic acid, which being lighter than the
water in which it was strongly condensed, flies off with great rapidity
the instant the bottle is uncorked; this makes it necessary to drink it
immediately. The bubbling that took place in this bottle was but
trifling, as the water was but very slightly impregnated with carbonic
acid. It requires a particular apparatus to prepare the gaseous
artificial mineral waters.
EMILY.
If, then, a bottle of Seltzer water remains for any length of time
uncorked, I suppose it returns to the state of common water?
MRS. B.
The whole of the carbonic acid gas, or very nearly so, will soon
disappear; but there is likewise in Seltzer water a very small quantity
of soda, and of a few other saline or earthy ingredients, which will
remain in the water, though it should be kept uncorked for any length of
time.
CAROLINE.
I have often heard of people drinking soda-water. Pray what sort of
water is that?
MRS. B.
It is a kind of artificial Seltzer water, holding
300
in solution, besides the gaseous acid, a particular saline
substance, called soda, which imparts to the water certain medicinal
qualities.
CAROLINE.
But how can these waters be so wholesome, since carbonic acid is so
pernicious?
MRS. B.
A gas, we may conceive, though very prejudicial to breathe, may be
beneficial to the stomach.—But it would be of no use to attempt
explaining this more fully at present.
CAROLINE.
Are waters never impregnated with other gases?
MRS. B.
Yes; there are several kinds of gaseous waters. I forgot to tell
you that waters have, for some years past, been prepared, impregnated
both with oxygen and hydrogen gases. These are not an imitation of
nature, but are altogether obtained by artificial means. They have been
lately used medicinally, particularly on the continent, where,
I understand, they have acquired some reputation.
EMILY.
If I recollect right, Mrs. B., you told us that
301
carbon was capable of decomposing water; the affinity between oxygen and
carbon must, therefore, be greater than between oxygen and hydrogen?
MRS. B.
Yes; but this is not the case unless their temperature be raised to a
certain degree. It is only when carbon is red-hot, that it is capable of
separating the oxygen from the hydrogen. Thus, if a small quantity of
water be thrown on a red-hot fire, it will increase rather than
extinguish the combustion; for the coals or wood (both of which contain
a quantity of carbon) decompose the water, and thus supply the fire both
with oxygen and hydrogen gases. If, on the contrary, a large mass
of water be thrown over the fire, the diminution of heat thus produced
is such, that the combustible matter loses the power of decomposing the
water, and the fire is extinguished.
EMILY.
I have heard that fire-engines sometimes do more harm than good, and
that they actually increase the fire when they cannot throw water enough
to extinguish it. It must be owing, no doubt, to the decomposition of
the water by the carbon during the conflagration.
302
MRS. B.
Certainly.—The apparatus which you see here (Plate XI. fig. 3.), may be used
to exemplify what we have just said. It consists in a kind of open
furnace, through which a porcelain tube, containing charcoal, passes. To
one end of the tube is adapted a glass retort with water in it; and the
other end communicates with a receiver placed on the water-bath.
A lamp being applied to the retort, and the water made to boil, the
vapour is gradually conveyed through the red-hot charcoal, by which it
is decomposed; and the hydrogen gas which results from this
decomposition is collected in the receiver. But the hydrogen thus
obtained is far from being pure; it retains in solution a minute portion
of carbon, and contains also a quantity of carbonic acid. This renders
it heavier than pure hydrogen gas, and gives it some peculiar
properties; it is distinguished by the name of carbonated hydrogen
gas.
CAROLINE.
And whence does it obtain the carbonic acid that is mixed
with it?
EMILY.
I believe I can answer that question, Caroline.—From the union
of the oxygen (proceeding from
303
the decomposed water) with the carbon, which, you know, makes carbonic
acid.
CAROLINE.
True; I should have recollected that.—The product of the
decomposition of water by red-hot charcoal, therefore, is carbonated
hydrogen gas, and carbonic acid gas.
MRS. B.
You are perfectly right now.
Carbon is frequently found combined with hydrogen in a state of
solidity, especially in coals, which owe their combustible nature to
these two principles.
EMILY.
Is it the hydrogen, then, that produces the flame of coals?
MRS. B.
It is so; and when all the hydrogen is consumed, the carbon continues
to burn without flame. But again, as I mentioned when speaking of the
gas-lights, the hydrogen gas produced by the burning of coals is not
pure; for, during the combustion, particles of carbon are successively
volatilized with the hydrogen, with which they form what is called a
hydro-carbonat, which is the principal product of this
combustion.
Carbon is a very bad conductor of heat; for
304
this reason, it is employed (in conjunction with other ingredients) for
coating furnaces and other chemical apparatus.
EMILY.
Pray what is the use of coating furnaces?
MRS. B.
In most cases, in which a furnace is used, it is necessary to produce
and preserve a great degree of heat, for which purpose every possible
means are used to prevent the heat from escaping by communicating with
other bodies, and this object is attained by coating over the inside of
the furnace with a kind of plaster, composed of materials that are bad
conductors of heat.
Carbon, combined with a small quantity of iron, forms a compound
called plumbago, or black-lead, of which pencils are made. This
substance, agreeably to the nomenclature, is a carburet of
iron.
EMILY.
Why, then, is it called black-lead?
MRS. B.
It is an ancient name given to it by ignorant people, from its
shining metallic appearance; but it is certainly a most improper name
for it, as there is not a particle of lead in the composition.
305
There is only one mine of this mineral, which is in Cumberland. It is
supposed to approach as nearly to pure carbon as the best prepared
charcoal does, as it contains only five parts of iron, unadulterated by
any other foreign ingredients. There is another carburet of iron, in
which the iron, though united only to an extremely small proportion of
carbon, acquires very remarkable properties; this is steel.
CAROLINE.
Really; and yet steel is much harder than iron?
MRS. B.
But carbon is not ductile like iron, and therefore may render the
steel more brittle, and prevent its bending so easily. Whether it is
that the carbon, by introducing itself into the pores of the iron, and,
by filling them, makes the metal both harder and heavier; or whether
this change depends upon some chemical cause, I cannot pretend to
decide. But there is a subsequent operation, by which the hardness of
steel is very much increased, which simply consists in heating the steel
till it is red-hot, and then plunging it into cold water.
Carbon, besides the combination just mentioned, enters into the
composition of a vast number of natural productions, such, for instance,
as all
306
the various kinds of oils, which result from the combination of carbon,
hydrogen, and caloric, in various proportions.
EMILY.
I thought that carbon, hydrogen, and caloric, formed carbonated
hydrogen gas?
MRS. B.
That is the case when a small portion of carbonic acid gas is held in
solution by hydrogen gas. Different proportions of the same principles,
together with the circumstances of their union, produce very different
combinations; of this you will see innumerable examples. Besides, we are
not now talking of gases, but of carbon and hydrogen, combined only with
a quantity of caloric sufficient to bring them to the consistency of oil
or fat.
CAROLINE.
But oil and fat are not of the same consistence?
MRS. B.
Fat is only congealed oil; or oil, melted fat. The one requires a
little more heat to maintain it in a fluid state than the other. Have
you never observed the fat of meat turned to oil by the caloric it has
imbibed from the fire?
307
EMILY.
Yet oils in general, as salad-oil, and lamp-oil, do not turn to fat
when cold?
MRS. B.
Not at the common temperature of the atmosphere, because they retain
too much caloric to congeal at that temperature; but if exposed to a
sufficient degree of cold, their latent heat is extricated, and they
become solid fat substances. Have you never seen salad oil frozen in
winter?
EMILY.
Yes; but it appears to me in that state very different from animal
fat.
MRS. B.
The essential constituent parts of either vegetable or animal oils
are the same, carbon and hydrogen; their variety arises from the
different proportions of these substances, and from other accessory
ingredients that may be mixed with them. The oil of a whale, and the oil
of roses, are, in their essential constituent parts, the same; but the
one is impregnated with the offensive particles of animal matter, the
other with the delicate perfume of a flower.
The difference of fixed oils, and volatile or
essential oils, consists also in the various proportions of
carbon and hydrogen. Fixed oils are those which
308
will not evaporate without being decomposed; this is the case with all
common oils, which contain a greater proportion of carbon than the
essential oils. The essential oils (which comprehend the whole class of
essences and perfumes) are lighter; they contain more equal proportions
of carbon and hydrogen, and are volatilized or evaporated without being
decomposed.
EMILY.
When you say that one kind of oil will evaporate, and the other be
decomposed, you mean, I suppose, by the application of heat?
MRS. B.
Not necessarily; for there are oils that will evaporate slowly at the
common temperature of the atmosphere; but for a more rapid
volatilization, or for their decomposition, the assistance of heat is
required.
CAROLINE.
I shall now remember, I think, that fat and oil are really the same
substances, both consisting of carbon and hydrogen; that in fixed oils
the carbon preponderates, and heat produces a decomposition; while, in
essential oils, the proportion of hydrogen is greater, and heat produces
a volatilization only.
EMILY.
I suppose the reason why oil burns so well in
309
lamps is because its two constituents are so combustible?
MRS. B.
Certainly; the combustion of oil is just the same as that of a
candle; if tallow, it is only oil in a concrete state; if wax, or
spermaceti, its chief chemical ingredients are still hydrogen and
carbon.
EMILY.
I wonder, then, there should be so great a difference between tallow
and wax?
MRS. B.
I must again repeat, that the same substances, in different
proportions, produce results that have sometimes scarcely any
resemblance to each other. But this is rather a general remark that I
wish to impress upon your minds, than one which is applicable to the
present case; for tallow and wax are far from being very dissimilar; the
chief difference consists in the wax being a purer compound of carbon
and hydrogen than the tallow, which retains more of the gross particles
of animal matter. The combustion of a candle, and that of a lamp, both
produce water and carbonic acid gas. Can you tell me how these are
formed?
EMILY.
Let me reflect . . . . Both the candle and lamp
310
burn by means of fixed oil—this is decomposed as the combustion
goes on; and the constituent parts of the oil being thus separated, the
carbon unites to a portion of oxygen from the atmosphere to form
carbonic acid gas, whilst the hydrogen combines with another portion of
oxygen, and forms with it water.—The products, therefore, of the
combustion of oils are water and carbonic acid gas.
CAROLINE.
But we see neither water nor carbonic acid produced by the combustion
of a candle.
MRS. B.
The carbonic acid gas, you know, is invisible, and the water being in
a state of vapour, is so likewise. Emily is perfectly correct in her
explanation, and I am very much pleased with it.
All the vegetable acids consist of various proportions of carbon and
hydrogen, acidified by oxygen. Gums, sugar, and starch, are likewise
composed of these ingredients; but, as the oxygen which they contain is
not sufficient to convert them into acids, they are classed with the
oxyds, and called vegetable oxyds.
CAROLINE.
I am very much delighted with all these new
311
ideas; but, at the same time, I cannot help being apprehensive that
I may forget many of them.
MRS. B.
I would advise you to take notes, or, what would answer better still,
to write down, after every lesson, as much of it as you can recollect.
And, in order to give you a little assistance, I shall lend you the
heads or index, which I occasionally consult for the sake of preserving
some method and arrangement in these conversations. Unless you follow
some such plan, you cannot expect to retain nearly all that you learn,
how great soever be the impression it may make on you at first.
EMILY.
I will certainly follow your advice.—Hitherto I have found that
I recollected pretty well what you have taught us; but the history of
carbon is a more extensive subject than any of the simple bodies we have
yet examined.
MRS. B.
I have little more to say on carbon at present; but hereafter you
will see that it performs a considerable part in most chemical
operations.
CAROLINE.
That is, I suppose, owing to its entering into
312
the composition of so great a variety of substances?
MRS. B.
Certainly; it is the basis, you have seen, of all vegetable matter;
and you will find that it is very essential to the process of
animalization. But in the mineral kingdom also, particularly in its form
of carbonic acid, we shall often discover it combined with a great
variety of substances.
In chemical operations, carbon is particularly useful, from its very
great attraction for oxygen, as it will absorb this substance from many
oxygenated or burnt bodies, and thus deoxygenate, or unburn them,
and restore them to their original combustible state.
CAROLINE.
I do not understand how a body can be unburnt, and restored to
its original state. This piece of tinder, for instance, that has been
burnt, if by any means the oxygen were extracted from it, would not be
restored to its former state of linen; for its texture is destroyed by
burning, and that must be the case with all organized or manufactured
substances, as you observed in a former conversation.
MRS. B.
A compound body is decomposed by combustion in a way which generally
precludes the possibility
313
of restoring it to its former state; the oxygen, for instance, does not
become fixed in the tinder, but it combines with its volatile parts, and
flies off in the shape of gas, or watery vapour. You see, therefore, how
vain it would be to attempt the recomposition of such bodies. But, with
regard to simple bodies, or at least bodies whose component parts are
not disturbed by the process of oxygenation or deoxygenation, it is
often possible to restore them, after combustion, to their original
state.—The metals, for instance, undergo no other alteration by
combustion than a combination with oxygen; therefore, when the oxygen is
taken from them, they return to their pure metallic state. But I shall
say nothing further of this at present, as the metals will furnish ample
subject for another morning; and they are the class of simple bodies
that come next under consideration.
314

MRS. B.
The METALS, which we are now to examine, are bodies of a
very different nature from those which we have hitherto considered. They
do not, like the bases of gases, elude the immediate observation of our
senses; for they are the most brilliant, the most ponderous, and the
most palpable substances in nature.
CAROLINE.
I doubt, however, whether the metals will appear to us so
interesting, and give us so much entertainment as those mysterious
elements which conceal themselves from our view. Besides, they cannot
afford so much novelty; they are bodies with which we are already so
well acquainted.
MRS. B.
You are not aware, my dear, of the interesting discoveries which were
a few years ago made by Sir H. Davy respecting this class of bodies. By
the aid of the Voltaic battery, he has obtained from
315
a variety of substances, metals before unknown, the properties of
which are equally new and curious. We shall begin, however, by noticing
those metals with which you profess to be so well acquainted. But the
acquaintance, you will soon perceive, is but very superficial; and I
trust that you will find both novelty and entertainment in considering
the metals in a chemical point of view. To treat of this subject fully,
would require a whole course of lectures; for metals form of themselves
a most important branch of practical chemistry. We must, therefore,
confine ourselves to a general view of them. These bodies are seldom
found naturally in their metallic form: they are generally more or less
oxygenated or combined with sulphur, earths, or acids, and are often
blended with each other. They are found buried in the bowels of the
earth in most parts of the world, but chiefly in mountainous districts,
where the surface of the globe has suffered from the earthquakes,
volcanos, and other convulsions of nature. They are spread in strata or
beds, called veins, and these veins are composed of a certain quantity
of metal, combined with various earthy substances, with which they form
minerals of different nature and appearance, which are called
ores.
CAROLINE.
I now feel quite at home, for my father has
316
a lead-mine in Yorkshire, and I have heard a great deal about veins
of ore, and of the roasting and smelting of the lead; but,
I confess, that I do not understand in what these operations
consist.
MRS. B.
Roasting is the process by which the volatile parts of the ore are
evaporated; smelting, that by which the pure metal is afterwards
separated from the earthy remains of the ore. This is done by throwing
the whole into a furnace, and mixing with it certain substances that
will combine with the earthy parts and other foreign ingredients of the
ore; the metal being the heaviest, falls to the bottom, and runs out by
proper openings in its pure metallic state.
EMILY.
You told us in a preceding lesson that metals had a great affinity
for oxygen. Do they not, therefore, combine with oxygen, when strongly
heated in the furnace, and run out in the state of oxyds?
MRS. B.
No; for the scoriæ, or oxyd, which soon forms on the surface of the
fused metal, when it is oxydable, prevents the air from having any
further influence on the mass; so that neither combustion nor
oxygenation can take place.
317
CAROLINE.
Are all the metals equally combustible?
MRS. B.
No; their attraction for oxygen varies extremely. There are some that
will combine with it only at a very high temperature, or by the
assistance of acids; whilst there are others that oxydate spontaneously
and with great rapidity, even at the lowest temperature; such is in
particular manganese, which scarcely ever exists in the metallic state,
as it immediately absorbs oxygen on being exposed to the air, and
crumbles to an oxyd in the course of a few hours.
EMILY.
Is not that the oxyd from which you extracted the oxygen gas?
MRS. B.
It is: so that, you see, this metal attracts oxygen at a low
temperature, and parts with it when strongly heated.
EMILY.
Is there any other metal that oxydates at the temperature of the
atmosphere?
MRS. B.
They all do, more or less, excepting gold, silver, and platina.
318
Copper, lead, and iron, oxydate slowly in the air, and cover
themselves with a sort of rust, a process which depends on the
gradual conversion of the surface into an oxyd. This rusty surface
preserves the interior metal from oxydation, as it prevents the air from
coming in contact with it. Strictly speaking, however, the word rust
applies only to the oxyd, which forms on the surface of iron, when
exposed to air and moisture, which oxyd appears to be united with a
small portion of carbonic acid.
EMILY.
When metals oxydate from the atmosphere without an elevation of
temperature, some light and heat, I suppose, must be disengaged,
though not in sufficient quantities to be sensible.
MRS. B.
Undoubtedly; and, indeed, it is not surprising that in this case the
light and heat should not be sensible, when you consider how extremely
slow, and, indeed, how imperfectly, most metals oxydate by mere exposure
to the atmosphere. For the quantity of oxygen with which metals are
capable of combining, generally depends upon their temperature; and the
absorption stops at various points of oxydation, according to the degree
to which their temperature is raised.
319
EMILY.
That seems very natural; for the greater the quantity of caloric
introduced into a metal, the more will its positive electricity be
exalted, and consequently the stronger will be its affinity for
oxygen.
MRS. B.
Certainly. When the metal oxygenates with sufficient rapidity for
light and heat to become sensible, combustion actually takes place. But
this happens only at very high temperatures, and the product is
nevertheless an oxyd; for though, as I have just said, metals will
combine with different proportions of oxygen, yet with the exception of
only five of them, they are not susceptible of acidification.
Metals change colour during the different degrees of oxydation which
they undergo. Lead, when heated in contact with the atmosphere, first
becomes grey; if its temperature be then raised, it turns yellow, and a
still stronger heat changes it to red. Iron becomes successively a
green, brown, and white oxyd. Copper changes from brown to blue, and
lastly green.
EMILY.
Pray, is the white lead with which houses are painted prepared by
oxydating lead?
320
MRS. B.
Not merely by oxydating, but by being also united with carbonic acid.
It is a carbonat of lead. The mere oxyd of lead is called red lead.
Litharge is another oxyd of lead, containing less oxygen. Almost all the
metallic oxyds are used as paints. The various sorts of ochres consist
chiefly of iron more or less oxydated. And it is a remarkable
circumstance, that if you burn metals rapidly, the light or flame they
emit during combustion partakes of the colours which the oxyd
successively assumes.
CAROLINE.
How is that accounted for, Mrs. B.? For light, you know, does not
proceed from the burning body, but from the decomposition of the oxygen
gas?
MRS. B.
The correspondence of the colour of the light with that of the oxyd
which emits it, is, in all probability, owing to some particles of the
metal which are volatilised and carried off by the caloric.
CAROLINE.
It is then a sort of metallic gas.
EMILY.
Why is it reckoned so unwholesome to breathe the air of a place in
which metals are melting?
321
MRS. B.
Perhaps the notion is too generally entertained. But it is true with
respect to lead, and some other noxious metals, because, unless care be
taken, the particles of the oxyd which are volatilised by the heat are
inhaled in with the breath, and may produce dangerous effects.
I must show you some instances of the combustion of metals; it would
require the heat of a furnace to make them burn in the common air, but
if we supply them with a stream of oxygen gas, we may easily
accomplish it.
CAROLINE.
But it will still, I suppose, be necessary in some degree to raise
their temperature?
MRS. B.
This, as you shall see, is very easily done, particularly if the
experiment be tried upon a small scale.—I begin by lighting
this piece of charcoal with the candle, and then increase the rapidity
of its
combustion by blowing upon it with a blow-pipe. (Plate XII. fig. 1.)
Plate XII.
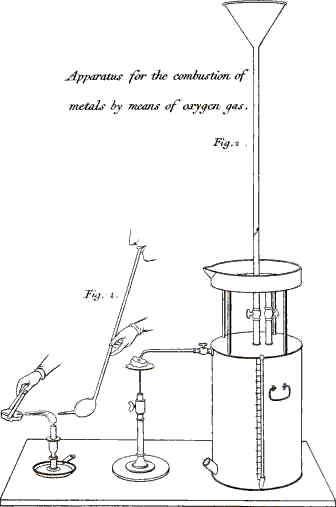
Fig. 1. Igniting charcoal with a taper & blow-pipe.
Fig. 2. Combustion of metals by means of a blow-pipe conveying a
stream of oxygen gas from a gas holder.
Larger
view
EMILY.
That I do not understand; for it is not every kind of air, but merely
oxygen gas, that produces combustion. Now you said that in breathing we
322
inspired, but did not expire oxygen gas. Why, therefore, should the air
which you breathe through the blow-pipe promote the combustion of the
charcoal?
MRS. B.
Because the air, which has but once passed through the lungs, is yet
but little altered, a small portion only of its oxygen being
destroyed; so that a great deal more is gained by increasing the
rapidity of the current, by means of the blow-pipe, than is lost in
consequence of the air passing once through the lungs, as you shall
see—
EMILY.
Yes, indeed, it makes the charcoal burn much brighter.
MRS. B.
Whilst it is red-hot, I shall drop some iron filings on it, and
supply them with a current of oxygen gas, by means of this apparatus,
(Plate XII. fig 2.) which
consists simply of a closed tin cylindrical vessel, full of oxygen gas,
with two apertures and stop-cocks, by one of which a stream of water is
thrown into the vessel through a long funnel, whilst by the other the
gas is forced out through a blow-pipe adapted to it, as the water gains
admittance.—Now that I pour water into the funnel, you may hear
the gas issuing from the
323
blow-pipe—I bring the charcoal close to the current, and drop the
filings upon it—
CAROLINE.
They emit much the same vivid light as the combustion of the iron
wire in oxygen gas.
MRS. B.
The process is, in fact, the same; there is only some difference in
the mode of conducting it. Let us burn some tin in the same
manner—you see that it is equally combustible.—Let us now
try some copper—
CAROLINE.
This burns with a greenish flame; it is, I suppose, owing to the
colour of the oxyd?
EMILY.
Pray, shall we not also burn some gold?
MRS. B.
That is not in our power, at least in this way. Gold, silver, and
platina, are incapable of being oxydated by the greatest heat that we
can produce by the common method. It is from this circumstance, that
they have been called perfect metals. Even these, however, have an
affinity for oxygen; but their oxydation or combustion can be performed
only by means of acids or by electricity.
324
The spark given out by the Voltaic battery produces at the point of
contact a greater degree of heat than any other process; and it is at
this very high temperature only that the affinity of these metals for
oxygen will enable them to act on each other.
I am sorry that I cannot show you the combustion of the perfect
metals by this process, but it requires a considerable Voltaic battery.
You will see these experiments performed in the most perfect manner,
when you attend the chemical lectures of the Royal Institution. But in
the mean time I can, without difficulty, show you an ingenious apparatus
lately contrived for the purpose of producing intense heats, the power
of which nearly equals that of the largest Voltaic batteries. It simply
consists, you see, in a strong box, made of iron or copper, (Plate X. fig. 2.) to which may be
adapted this air-syringe or condensing-pump, and a stop-cock terminating
in a small orifice similar to that of a blow-pipe. By working the
condensing syringe, up and down in this manner, a quantity of air
is accumulated in the vessel, which may be increased to almost any
extent; so that if we now turn the stop-cock, the condensed air will
rush out, forming a jet of considerable force; and if we place the flame
of a lamp in the current, you will see how violently the flame is driven
in that direction.
Plate X.
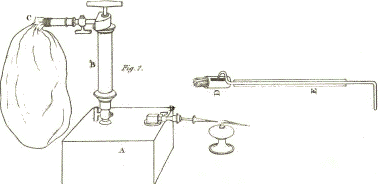
Fig. 2. A. the reservoir of condensed air.
B. the condensing Syringe.
C. the bladder for Oxygen.
D. the moveable jet.
Larger view
(complete Plate)
This part of the Plate was printed sideways to fit alongside the
illustration of the miner’s lamp, added for this edition.
325
CAROLINE.
It seems to be exactly the same effect as that of a blow-pipe worked
by the mouth, only much stronger.
EMILY.
Yes; and this new instrument has this additional advantage, that it
does not fatigue the mouth and lungs like the common blow-pipe, and
requires no art in blowing.
MRS. B.
Unquestionably; but yet this blow-pipe would be of very limited
utility, if its energy and power could not be greatly increased by some
other contrivance. Can you imagine any mode of producing such an
effect?
EMILY.
Could not the reservoir be charged with pure oxygen, instead of
common air, as in the case of the gas-holder?
MRS. B.
Undoubtedly; and this is precisely the contrivance I allude to. The
vessel need only be supplied with air from a bladder full of oxygen,
instead of the air of the room, and this, you see, may be easily done by
screwing the bladder on the upper part of the syringe, so that in
working the syringe the oxygen gas is forced from the bladder into the
condensing vessel.
326
CAROLINE.
With the aid of this small apparatus, therefore, we could obtain the
same effects as those we have just produced with the gas-holder, by
means of a column of water forcing the gas out of it?
MRS. B.
Yes; and much more conveniently so. But there is a mode of using this
apparatus by which more powerful effects still may be obtained. It
consists in condensing in the reservoir, not oxygen alone, but a mixture
of oxygen and hydrogen in the exact proportion in which they unite to
produce water; and then kindling the jet formed by the mixed gases. The
heat disengaged by this combustion, without the help of any lamp, is
probably the most intense known; and various effects are said to have
been obtained from it which exceed all expectation.
CAROLINE.
But why should we not try this experiment?
MRS. B.
Because it is not exempt from danger; the combustion (notwithstanding
various contrivances which have been resorted to with a view to prevent
accident) being apt to penetrate into the inside of the vessel, and to
produce a dangerous and violent
327
explosion.—We shall, therefore, now proceed in our subject.
CAROLINE.
I think you said the oxyds of metals could be restored to their
metallic state?
MRS. B.
Yes; this is called reviving a metal. Metals are in general
capable of being revived by charcoal, when heated red hot, charcoal
having a greater attraction for oxygen than the metals. You need only,
therefore, decompose, or unburn the oxyd, by depriving it of its oxygen,
and the metal will be restored to its pure state.
EMILY.
But will the carbon, by this operation, be burnt, and be converted
into carbonic acid?
MRS. B.
Certainly. There are other combustible substances to which metals at
a high temperature will part with their oxygen. They will also yield it
to each other, according to their several degrees of attraction for it;
and if the oxygen goes into a more dense state in the metal which it
enters, than it existed in that which it quits, a proportional
disengagement of caloric will take place.
328
CAROLINE.
And cannot the oxyds of gold, silver, and platina, which are formed
by means of acids or of the electric fluid, be restored to their
metallic state?
MRS. B.
Yes, they may; and the intervention of a combustible body is not
required; heat alone will take the oxygen from them, convert it into a
gas, and revive the metal.
EMILY.
You said that rust was an oxyd of iron; how is it, then, that water,
or merely dampness, produces it, which, you know, it very frequently
does on steel grates, or any iron instruments?
MRS. B.
In that case the metal decomposes the water, or dampness (which is
nothing but water in a state of vapour), and obtains the oxygen
from it.
CAROLINE.
I thought that it was necessary to bring metals to a very high
temperature to enable them to decompose water.
MRS. B.
It is so, if it is required that the process should be performed
rapidly, and if any considerable quantity is to be decomposed. Rust, you
knew,
329
is sometimes months in forming, and then it is only the surface of the
metal that is oxydated.
EMILY.
Metals, then, that do not rust, are incapable of spontaneous
oxydation, either by air or water?
MRS. B.
Yes; and this is the case with the perfect metals, which, on that
account, preserve their metallic lustre so well.
EMILY.
Are all metals capable of decomposing water, provided their
temperature be sufficiently raised?
MRS. B.
No; a certain degree of attraction is requisite, besides the
assistance of heat. Water, you recollect, is composed of oxygen and
hydrogen; and, unless the affinity of the metal for oxygen be stronger
than that of hydrogen, it is in vain that we raise its temperature, for
it cannot take the oxygen from the hydrogen. Iron, zinc, tin, and
antimony, have a stronger affinity for oxygen than hydrogen has,
therefore these four metals are capable of decomposing water. But
hydrogen having an advantage over all the other metals with respect to
its affinity for oxygen, it not only withholds its oxygen from them, but
is even capable,
330
under certain circumstances, of taking the oxygen from the oxyds of
these metals.
EMILY.
I confess that I do not quite understand why hydrogen can take oxygen
from those metals that do not decompose water.
CAROLINE.
Now I think I do perfectly. Lead, for instance, will not decompose
water, because it has not so strong an attraction for oxygen as hydrogen
has. Well, then, suppose the lead to be in a state of oxyd; hydrogen
will take the oxygen from the lead, and unite with it to form water,
because hydrogen has a stronger attraction for oxygen, than oxygen has
for lead; and it is the same with all the other metals which do not
decompose water.
EMILY.
I understand your explanation, Caroline, very well; and I imagine
that it is because lead cannot decompose water that it is so much
employed for pipes for conveying that fluid.
MRS. B.
Certainly; lead is, on that account, particularly appropriate to such
purposes; whilst, on the contrary, this metal, if it was oxydable by
water,
331
would impart to it very noxious qualities, as all oxyds of lead are more
or less pernicious.
But, with regard to the oxydation of metals, the most powerful mode
of effecting it is by means of acids. These, you know, contain a much
greater proportion of oxygen than either air or water; and will, most of
them, easily yield it to metals. Thus, you recollect, the zinc plates of
the Voltaic battery are oxydated by the acid and water, much more
effectually than by water alone.
CAROLINE.
And I have often observed that if I drop vinegar, lemon, or any acid
on the blade of a knife, or on a pair of scissars, it will immediately produce a spot of
rust.
EMILY.
Metals have, then, three ways of obtaining oxygen; from the
atmosphere, from water, and from acids.
MRS. B.
The two first you have already witnessed, and I shall now show you
how metals take the oxygen from an acid. This bottle contains nitric
acid; I shall pour some of it over this piece of copper-leaf
. . . . . . .
CAROLINE.
Oh, what a disagreeable smell!
332
EMILY.
And what is it that produces the effervescency and that thick yellow
vapour?
MRS. B.
It is the acid, which being abandoned by the greatest part of its
oxygen, is converted into a weaker acid, which escapes in the form of
gas.
CAROLINE.
And whence proceeds this heat?
MRS. B.
Indeed, Caroline, I think you might now be able to answer that
question yourself.
CAROLINE.
Perhaps it is that the oxygen enters into the metal in a more solid
state than it existed in the acid, in consequence of which caloric is
disengaged.
MRS. B.
If the combination of the oxygen and the metal results from the union
of their opposite electricities, of course caloric must be given
out.
EMILY.
The effervescence is over; therefore I suppose that the metal is now
oxydated.
333
MRS. B.
Yes. But there is another important connection between metals and
acids, with which I must now make you acquainted. Metals, when in the
state of oxyds, are capable of being dissolved by acids. In this
operation they enter into a chemical combination with the acid, and form
an entirely new compound.
CAROLINE.
But what difference is there between the oxydation and the
dissolution of the metal by an acid?
MRS. B.
In the first case, the metal merely combines with a portion of oxygen
taken from the acid, which is thus partly deoxygenated, as in the
instance you have just seen; in the second case, the metal, after being
previously oxydated, is actually dissolved in the acid, and enters into
a chemical combination with it, without producing any further
decomposition or effervescence.—This complete combination of an
oxyd and an acid forms a peculiar and important class of compound
salts.
EMILY.
The difference between an oxyd and a compound salt, therefore, is
very obvious: the one consists of a metal and oxygen; the other of an
oxyd and an acid.
334
MRS. B.
Very well: and you will be careful to remember that the metals are
incapable of entering into this combination with acids, unless they are
previously oxydated; therefore, whenever you bring a metal in contact
with an acid, it will be first oxydated and afterwards dissolved,
provided that there be a sufficient quantity of acid for both
operations.
There are some metals, however, whose solution is more easily
accomplished, by diluting the acid in water; and the metal will, in this
case, be oxydated, not by the acid, but by the water, which it will
decompose. But in proportion as the oxygen of the water oxydates the
surface of the metal, the acid combines with it, washes it off, and
leaves a fresh surface for the oxygen to act upon: then other coats of
oxyd are successively formed, and rapidly dissolved by the acid, which
continues combining with the new-formed surfaces of oxyd till the whole
of the metal is dissolved. During this process the hydrogen gas of the
water is disengaged, and flies off with effervescence.
EMILY.
Was not this the manner in which the sulphuric acid assisted the iron
filings in decomposing water?
335
MRS. B.
Exactly; and it is thus that several metals, which are incapable
alone of decomposing water, are enabled to do it by the assistance of an
acid, which, by continually washing off the covering of oxyd, as it is
formed, prepares a fresh surface of metal to act upon the water.
CAROLINE.
The acid here seems to act a part not very different from that of a
scrubbing-brush.—But pray would not this be a good method of
cleaning metallic utensils?
MRS. B.
Yes; on some occasions a weak acid, as vinegar, is used for cleaning
copper. Iron plates, too, are freed from the rust on their surface by
diluted muriatic acid, previous to their being covered with tin. You
must remember, however, that in this mode of cleaning metals the acid
should be quickly afterwards wiped off, otherwise it would produce fresh
oxyd.
CAROLINE.
Let us watch the dissolution of the copper in the nitric acid; for I
am very impatient to see the salt that is to result from it. The mixture
is now of a beautiful blue colour; but there is no appearance
336
of the formation of a salt; it seems to be a tedious operation.
MRS. B.
The crystallisation of the salt requires some length of time to be
completed; if, however, you are so impatient, I can easily show you
a metallic salt already formed.
CAROLINE.
But that would not satisfy my curiosity half so well as one of our
own manufacturing.
MRS. B.
It is one of our own preparing that I mean to show you. When we
decomposed water a few days since, by the oxydation of iron filings
through the assistance of sulphuric acid, in what did the process
consist?
CAROLINE.
In proportion as the water yielded its oxygen to the iron, the acid
combined with the new-formed oxyd, and the hydrogen escaped alone.
MRS. B.
Very well; the result, therefore, was a compound salt, formed by the
combination of sulphuric acid with oxyd of iron. It still remains in
337
the vessel in which the experiment was performed. Fetch it, and we shall
examine it.
EMILY.
What a variety of processes the decomposition of water, by a metal
and an acid, implies; 1st, the decomposition of the water; 2dly, the
oxydation of the metal; and 3dly, the formation of a compound salt.
CAROLINE.
Here it is, Mrs. B.—What beautiful green crystals! But we do
not perceive any crystals in the solution of copper in nitrous acid?
MRS. B.
Because the salt is now suspended in the water which the nitrous acid
contains, and will remain so till it is deposited in consequence of rest
and cooling.
EMILY.
I am surprised that a body so opake as iron can be converted into
such transparent crystals.
MRS. B.
It is the union with the acid that produces the transparency; for if
the pure metal were melted, and afterwards permitted to cool and
crystallise, it would be found just as opake as before.
338
EMILY.
I do not understand the exact meaning of crystallisation?
MRS. B.
You recollect that when a solid body is dissolved either by water or
caloric it is not decomposed; but that its integrant parts are only
suspended in the solvent. When the solution is made in water, the
integrant particles of the body will, on the water being evaporated,
again unite into a solid mass by the force of their mutual attraction.
But when the body is dissolved by caloric alone, nothing more is
necessary, in order to make its particles reunite, than to reduce its
temperature. And, in general, if the solvent, whether water or caloric,
be slowly separated by evaporation or by cooling, and care taken that
the particles be not agitated during their reunion, they will arrange
themselves in regular masses, each individual substance assuming a
peculiar form or arrangement; and this is what is called
crystallisation.
EMILY.
Crystallisation, therefore, is simply the reunion of the particles of
a solid body that has been dissolved in a fluid.
339
MRS. B.
That is a very good definition of it. But I must not forget to
observe, that heat and water may unite their solvent
powers; and, in this case, crystallisation may be hastened by cooling,
as well as by evaporating the liquid.
CAROLINE.
But if the body dissolved is of a volatile nature, will it not
evaporate with the fluid?
MRS. B.
A crystallised body held in solution only by water is scarcely ever
so volatile as the fluid itself, and care must be taken to manage the
heat so that it may be sufficient to evaporate the water only.
I should not omit also to mention that bodies, in crystallising from
their watery solution, always retain a small portion of water, which
remains confined in the crystal in a solid form, and does not reappear
unless the body loses its crystalline state. This is called the water
of crystallisation. But you must observe, that whilst a body may be
separated from its solution in water or caloric simply by cooling or by
evaporation, an acid can be taken from a metal with which it is combined
only by stronger affinities, which produce a decomposition.
340
EMILY.
Are the perfect metals susceptible of being dissolved and converted
into compound salts by acids?
MRS. B.
Gold is acted upon by only one acid, the oxygenated muriatic,
a very remarkable acid, which, when in its most concentrated state,
dissolves gold or any other metal, by burning them rapidly.
Gold can, it is true, be dissolved likewise by a mixture of two
acids, commonly called aqua regia; but this mixed solvent derives
that property from containing the peculiar acid which I have just
mentioned. Platina is also acted upon by this acid only; silver is
dissolved by nitric acid.
CAROLINE.
I think you said that some of the metals might be so strongly
oxydated as to become acid?
MRS. B.
There are five metals, arsenic, molybdena, chrome, tungsten, and
columbium, which are susceptible of combining with a sufficient quantity
of oxygen to be converted into acids.
CAROLINE.
Acids are connected with metals in such a variety of ways, that I am
afraid of some confusion in remembering
341
them.—In the first place, acids will yield their oxygen to metals.
Secondly, they will combine with them in their state of oxyds, to form
compound salts; and lastly, several of the metals are themselves
susceptible of acidification.
MRS. B.
Very well; but though metals have so great an affinity for acids, it
is not with that class of bodies alone that they will combine. They are
most of them, in their simple state, capable of uniting with sulphur,
with phosphorus, with carbon, and with each other; these combinations,
according to the nomenclature which was explained to you on a former
occasion, are called sulphurets, phosphorets,
carburets, &c.
The metallic phosphorets offer nothing very remarkable. The
sulphurets form the peculiar kind of mineral called pyrites, from
which certain kinds of mineral waters, as those of Harrogate, derive
their chief chemical properties. In this combination, the sulphur,
together with the iron, have so strong an attraction for oxygen, that
they obtain it both from the air and from water, and by condensing it in
a solid form, produce the heat which raises the temperature of the water
in such a remarkable degree.
EMILY.
But if pyrites obtain oxygen from water, that
342
water must suffer a decomposition, and hydrogen gas be evolved.
MRS. B.
That is actually the case in the hot springs alluded to, which give
out an extremely fetid gas, composed of hydrogen impregnated with
sulphur.
CAROLINE.
If I recollect right, steel and plumbago, which you mentioned in the
last lesson, are both carburets of iron?
MRS. B.
Yes; and they are the only carburets of much consequence.
A curious combination of metals has lately very much attracted the
attention of the scientific world: I mean the meteoric stones that
fall from the atmosphere. They consist principally of native or pure
iron, which is never found in that state in the bowels of the earth; and
contain also a small quantity of nickel and chrome, a combination
likewise new in the mineral kingdom.
These circumstances have led many scientific persons to believe that
those substances have fallen from the moon, or some other planet, while
others are of opinion either that they are formed in the atmosphere, or
are projected into it by some unknown volcano on the surface of our
globe.
343
CAROLINE.
I have heard much of these stones, but I believe many people are of
opinion that they are formed on the surface of the earth, and laugh at
their pretended celestial origin.
MRS. B.
The fact of their falling is so well ascertained, that I think no
person who has at all investigated the subject, can now entertain any
doubt of it. Specimens of these stones have been discovered in all parts
of the world, and to each of them some tradition or story of its fall
has been found connected. And as the analysis of all those specimens
affords precisely the same results, there is strong reason to conjecture
that they all proceed from the same source. It is to Mr. Howard that
philosophers are indebted for having first analysed these stones, and
directed their attention to this interesting subject.
CAROLINE.
But pray, Mrs. B., how can solid masses of iron and nickel be formed
from the atmosphere, which consists of the two airs, nitrogen and
oxygen?
MRS. B.
I really do not see how they could, and think it much more probable
that they fall from the moon.—But we must not suffer this
digression to take up too much of our time.
344
The combinations of metals with each other are called alloys; thus
brass is an alloy of copper and zinc; bronze, of copper and
tin, &c.
EMILY.
And is not pewter also a combination of metal?
MRS. B.
It is. The pewter made in this country is mostly composed of tin,
with a very small proportion of zinc and lead.
CAROLINE.
Block-tin is a kind of pewter, I believe?
MRS. B.
Properly speaking, block-tin means tin in blocks, or square massive
ingots; but in the sense in which it is used by ignorant workmen, it is
iron plated with tin, which renders it more durable, as tin will not so
easily rust. Tin alone, however, would be too soft a metal to be worked
for common use, and all tin-vessels and utensils are in fact made of
plates of iron, thinly coated with tin, which prevents the iron from
rusting.
CAROLINE.
Say rather oxydating, Mrs. B.—Rust is a word that should
be exploded in chemistry.
345
MRS. B.
Take care, however, not to introduce the word oxydate, instead of
rust, in general conversation; for you would probably not be understood,
and you might be suspected of affectation.
Metals differ very much in their affinity for each other; some will
not unite at all, others readily combine together, and on this property
of metals the art of soldering depends.
EMILY.
What is soldering?
MRS. B.
It is joining two pieces of metal together, by a more fusible metal
interposed between them. Thus tin is a solder for lead; brass, gold, or
silver, are solder for iron, &c.
CAROLINE.
And is not plating metals something of the same nature?
MRS. B.
In the operation of plating, two metals are united, one being covered
with the other, but without the intervention of a third; iron or copper
may thus be covered with gold or silver.
346
EMILY.
Mercury appears to me of a very different nature from the other
metals.
MRS. B.
One of its greatest peculiarities is, that it retains a fluid state
at the temperature of the atmosphere. All metals are fusible at
different degrees of heat, and they have likewise each the property of
freezing or becoming solid at a certain fixed temperature. Mercury
congeals only at seventy-two degrees below the freezing point.
EMILY.
That is to say, that in order to freeze, it requires a temperature of
seventy-two degrees colder than that at which water freezes.
MRS. B.
Exactly so.
CAROLINE.
But is the temperature of the atmosphere ever so low as that?
MRS. B.
Yes, often in Siberia; but happily never in this part of the globe.
Here, however, mercury may be congealed by artificial cold; I mean
such intense cold as can be produced by some chemical
347
mixtures, or by the rapid evaporation of ether under the air-pump.*
CAROLINE.
And can mercury be made to boil and evaporate?
MRS. B.
Yes, like any other liquid; only it requires a much greater degree of
heat. At the temperature of six hundred degrees, it begins to boil and
evaporate like water.
Mercury combines with gold, silver, tin, and with several other
metals; and, if mixed with any of them in a sufficient proportion, it
penetrates the solid metal, softens it, loses its own fluidity, and
forms an amalgam, which is the name given to the combination of
any metal with mercury, forming a substance more or less solid,
according as the mercury or the other metal predominates.
EMILY.
In the list of metals there are some whose names I have never before
heard mentioned.
MRS. B.
Besides those which Sir H. Davy has obtained, there are several that
have been recently discovered,
348
whose properties are yet but little known, as for instance, titanium,
which was discovered by the Rev. Mr. Gregor, in the tin-mines of
Cornwall; columbium or tantalium, which has lately been discovered by
Mr. Hatchett; and osmium, iridium, palladium, and rhodium, all of which
Dr. Wollaston and Mr. Tennant found mixed in minute quantities with
crude platina, and the distinct existence of which they proved by
curious and delicate experiments.
CAROLINE.
Arsenic has been mentioned amongst the metals. I had no notion that it belonged to
that class of bodies, for I had never seen it but as a powder, and never
thought of it but as a most deadly poison.
MRS. B.
In its pure metallic state, I believe, it is not so poisonous; but it
has such a great affinity for oxygen, that it absorbs it from the
atmosphere at its natural temperature: you have seen it, therefore, only
in its state of oxyd, when, from its combination with oxygen, it has
acquired its very poisonous properties.
CAROLINE.
Is it possible that oxygen can impart poisonous qualities? That
valuable substance which produces
349
light and fire, and which all bodies in nature are so eager to
obtain?
MRS. B.
Most of the metallic oxyds are poisonous, and derive this property
from their union with oxygen. The white lead, so much used in paint,
owes its pernicious effects to oxygen. In general, oxygen, in a concrete
state, appears to be particularly destructive in its effects on flesh or
any animal matter; and those oxyds are most caustic that have an acrid
burning taste, which proceeds from the metal having but a slight
affinity for oxygen, and therefore easily yielding it to the flesh,
which it corrodes and destroys.
EMILY.
What is the meaning of the word caustic, which you have just
used?
MRS. B.
It expresses that property which some bodies possess, of
disorganizing and destroying animal matter, by operating a kind of
combustion, or at least a chemical decomposition. You must often have
heard of caustic used to burn warts, or other animal excrescences; most
of these bodies owe their destructive power to the oxygen with which
they are combined. The common caustic, called
350
lunar caustic, is a compound formed by the union of nitric acid
and silver; and it is supposed to owe its caustic qualities to the
oxygen contained in the nitric acid.
CAROLINE.
But, pray, are not acids still more caustic than oxyds, as they
contain a greater proportion of oxygen?
MRS. B.
Some of the acids are; but the caustic property of a body depends not
only upon the quantity of oxygen which it contains, but also upon its
slight affinity for that principle, and the consequent facility with
which it yields it.
EMILY.
Is not this destructive property of oxygen accounted for?
MRS. B.
It proceeds probably from the strong attraction of oxygen for
hydrogen; for if the one rapidly absorb the other from the animal fibre,
a disorganisation of the substance must ensue.
EMILY.
Caustics are, then, very properly said to burn the flesh, since the
combination of oxygen and hydrogen is an actual combustion.
351
CAROLINE.
Now, I think, this effect would be more properly termed an oxydation,
as there is no disengagement of light and heat.
MRS. B.
But there really is a sensation of heat produced by the action of
caustics.
EMILY.
If oxygen is so caustic, why does not that which is contained in the
atmosphere burn us?
MRS. B.
Because it is in a gaseous state, and has a greater attraction for
its electricity than for the hydrogen of our bodies. Besides, should the
air be slightly caustic, we are in a great measure sheltered from its
effects by the skin; you know how much a wound, however trifling, smarts
on being exposed to it.
CAROLINE.
It is a curious idea, however, that we should live in a slow fire.
But, if the air was caustic, would it not have an acrid taste?
MRS. B.
It possibly may have such a taste; though in so
352
slight a degree, that custom has rendered it insensible.
CAROLINE.
And why is not water caustic? When I dip my hand into water, though
cold, it ought to burn me from the caustic nature of its oxygen.
MRS. B.
Your hand does not decompose the water; the oxygen in that state is
much better supplied with hydrogen than it would be by animal matter,
and if its causticity depend on its affinity for that principle, it will
be very far from quitting its state of water to act upon your hand. You
must not forget that oxyds are caustic in proportion as the oxygen
adheres slightly to them.
EMILY.
Since the oxyd of arsenic is poisonous, its acid, I suppose, is
fully as much so?
MRS. B.
Yes; it is one of the strongest poisons in nature.
EMILY.
There is a poison called verdigris, which forms on brass and
copper when not kept very clean; and this, I have heard, is an
objection to these
353
metals being made into kitchen utensils. Is this poison likewise
occasioned by oxygen?
MRS. B.
It is produced by the intervention of oxygen; for verdigris is a
compound salt formed by the union of vinegar and copper; it is of a
beautiful green colour, and much used in painting.
EMILY.
But, I believe, verdigris is often formed on copper when no vinegar
has been in contact with it.
MRS. B.
Not real verdigris, but compound salts, somewhat resembling it, may
be produced by the action of any acid on copper.
The solution of copper in nitric acid, if evaporated, affords a salt
which produces an effect on tin that will surprise you, and I have
prepared some from the solution we made before, that I might show it to
you. I shall first sprinkle some water on this piece of tin-foil,
and then some of the salt.—Now observe that I fold it up suddenly,
and press it into one lump.
CAROLINE.
What a prodigious vapour issues from it—and sparks of fire I
declare!
354
MRS. B.
I thought it would surprise you. The effect, however, I dare say
you could account for, since it is merely the consequence of the oxygen
of the salt rapidly entering into a closer combination with the tin.
There is also a beautiful green salt too curious to be omitted; it is
produced by the combination of cobalt with muriatic acid, which has the
singular property of forming what is called sympathetic ink.
Characters written with this solution are invisible when cold, but when
a gentle heat is applied, they assume a fine bluish green colour.
CAROLINE.
I think one might draw very curious landscapes with the assistance of
this ink; I would first make a water-colour drawing of a
winter-scene, in which the trees should be leafless, and the grass
scarcely green: I would then trace all the verdure with the
invisible ink, and whenever I chose to create spring, I should hold
it before the fire, and its warmth would cover the landscape with a rich
verdure.
MRS. B.
That will be a very amusing experiment, and I advise you by all means
to try it.
Several cobalt compounds, including the cobalt chloride described here,
are still in use as invisible (“sympathetic”) inks. They are safe if
used appropriately.
Before we part, I must introduce to your acquaintance the curious
metals which Sir H. Davy
355
has recently discovered. The history of these extraordinary bodies is
yet so much in its infancy, that I shall confine myself to a very short
account of them; it is more important to point out to you the vast, and
apparently inexhaustible, field of research which has been thrown open
to our view by Sir H. Davy’s memorable discoveries, than to enter into a
minute account of particular bodies or experiments.
CAROLINE.
But I have heard that these discoveries, however splendid and
extraordinary, are not very likely to prove of any great benefit to the
world, as they are rather objects of curiosity than of use.
MRS. B.
Such may be the illiberal conclusions of the ignorant and
narrow-minded; but those who can duly estimate the advantages of
enlarging the sphere of science, must be convinced that the acquisition
of every new fact, however unconnected it may at first appear with
practical utility, must ultimately prove beneficial to mankind. But
these remarks are scarcely applicable to the present subject; for some
of the new metals have already proved eminently useful as chemical
agents, and are likely soon to be employed in the arts. For the
enumeration of these metals, I must refer you to our list of simple
bodies; they are derived from the alkalies, the
356
earths, and three of the acids, all of which had been hitherto
considered as undecompoundable or simple bodies.
When Sir H. Davy first turned his attention to the effects of the
Voltaic battery, he tried its power on a variety of compound bodies, and
gradually brought to light a number of new and interesting facts, which
led the way to more important discoveries. It would be highly
interesting to trace his steps in this new department of science, but it
would lead us too far from our principal object. A general view of
his most remarkable discoveries is all that I can aim at, or that you
could, at present, understand.
The facility with which compound bodies yielded to the Voltaic
electricity, induced him to make trial of its effects on substances
hitherto considered as simple, but which he suspected of being compound,
and his researches were soon crowned with the most complete success.
The body which he first submitted to the Voltaic battery, and which
had never yet been decomposed, was one of the fixed alkalies, called
potash. This substance gave out an elastic fluid at the positive wire,
which was ascertained to be oxygen, and at the negative wire, small
globules of a very high metallic lustre, very similar in appearance to
mercury; thus proving that potash, which had hitherto been considered as
a simple incombustible body,
357
was in fact a metallic oxyd; and that its incombustibility proceeded
from its being already combined with oxygen.
EMILY.
I suppose the wires used in this experiment were of platina, as they
were when you decomposed water; for if of iron, the oxygen would have
combined with the wire, instead of appearing in the form of gas.
MRS. B.
Certainly: the metal, however, would equally have been disengaged.
Sir H. Davy has distinguished this new substance by the name of POTASSIUM, which is derived from that of the
alkali, from which it is procured. I have some small pieces of it
in this phial, but you have already seen it, as it is the metal which we
burnt in contact with sulphur.
EMILY.
What is the liquid in which you keep it?
MRS. B.
It is naptha, a bituminous liquid, with which I shall hereafter make
you acquainted. It is almost the only fluid in which potassium can be
preserved, as it contains no oxygen, and this metal has so powerful an
attraction for oxygen, that it will not only absorb it from the air, but
likewise from water, or any body whatever that contains it.
358
EMILY.
This, then, is one of the bodies that oxydates spontaneously without
the application of heat?
MRS. B.
Yes; and it has this remarkable peculiarity that it attracts oxygen
much more rapidly from water than from air; so that when thrown into
water, however cold, it actually bursts into flame. I shall now
throw a small piece, about the size of a pin’s head, on this drop of
water.
CAROLINE.
It instantaneously exploded, producing a little flash of light! this
is, indeed, a most curious substance!
MRS. B.
By its combustion it is reconverted into potash; and as potash is now
decidedly a compound body, I shall not enter into any of its
properties till we have completed our review of the simple bodies; but
we may here make a few observations on its basis, potassium. If this
substance is left in contact with air, it rapidly returns to the state
of potash, with a disengagement of heat, but without any flash of
light.
EMILY.
But is it not very singular
that it should burn better in water than in air?
359
CAROLINE.
I do not think so: for if the attraction of potassium for oxygen is
so strong that it finds no more difficulty in separating it from the
hydrogen in water, than in absorbing it from the air, it will no doubt
be more amply and rapidly supplied by water than by air.
MRS. B.
That cannot, however, be precisely the reason, for when potassium is
introduced under water, without contact of air, the combustion is not so
rapid, and indeed, in that case, there is no luminous appearance; but a
violent action takes place, much heat is excited, the potash is
regenerated, and hydrogen gas is evolved.
Potassium is so eminently combustible, that instead of requiring,
like other metals, an elevation of temperature, it will burn rapidly in
contact with water, even below the freezing point. This you may witness
by throwing a piece on this lump of ice.
CAROLINE.
It again exploded with flame, and has made a deep hole in the
ice.
MRS. B.
This hole contains a solution of potash; for the alkali being
extremely soluble, disappears in the
360
water at the instant it is produced. Its presence, however, may be
easily ascertained, alkalies having the property of changing paper,
stained with turmeric, to a red colour; if you dip one end of this slip
of paper into the hole in the ice you will see it change colour, and the
same, if you wet it with the drop of water in which the first piece of
potassium was burnt.
CAROLINE.
It has indeed changed the paper from yellow to red.
MRS. B.
This metal will burn likewise in carbonic acid gas, a gas that
had always been supposed incapable of supporting combustion, as we were
unacquainted with any substance that had a greater attraction for oxygen
than carbon. Potassium, however, readily decomposes this gas, by
absorbing its oxygen, as I shall show you. This retort is filled with
carbonic acid gas.—I will put a small piece of potassium in
it; but for this combustion a slight elevation of temperature is
required, for which purpose I shall hold the retort over the lamp.
CAROLINE.
Now it has taken fire, and burns with violence! It has burst the
retort.
361
MRS. B.
Here is the piece of regenerated potash; can you tell me why it is
become so black?
EMILY.
No doubt it is blackened by the carbon, which, when its oxygen
entered into combination with the potassium, was deposited on its
surface.
MRS. B.
You are right. This metal is perfectly fluid at the temperature of
one hundred degrees; at fifty degrees it is solid, but soft and
malleable; at thirty-two degrees it is hard and brittle, and its
fracture exhibits an appearance of confused crystallization. It is
scarcely more than half as heavy as water; its specific gravity being
about six when water is reckoned at ten; so that this metal is actually
lighter than any known fluid, even than ether.
Potassium combines with sulphur and phosphorus, forming sulphurets
and phosphurets; it likewise forms alloys with several metals, and
amalgamates with mercury.
EMILY.
But can a sufficient quantity of potassium be obtained, by means of
the Voltaic battery, to admit of all its properties and relations to
other bodies being satisfactorily ascertained?
362
MRS. B.
Not easily; but I must not neglect to inform you that a method of
obtaining this metal in considerable quantities has since been
discovered. Two eminent French chemists, Thenard and Gay
Lussac, stimulated by the triumph which Sir H. Davy had obtained,
attempted to separate potassium from its combination with oxygen, by
common chemical means, and without the aid of electricity. They caused
red hot potash in a state of fusion to filter through iron turnings in
an iron tube, heated to whiteness. Their experiment was crowned with the
most complete success; more potassium was obtained by this single
operation, that could have been collected in many weeks by the most
diligent use of the Voltaic battery.
EMILY.
In this experiment, I suppose, the oxygen quitted its combination
with the potassium to unite with the iron turnings?
MRS. B.
Exactly so; and the potassium was thus obtained in its simple state.
From that time it has become a most convenient and powerful instrument
of deoxygenation in chemical experiments. This important improvement,
engrafted on Sir H. Davy’s previous discoveries, served but to add to
his glory, since the facts which he had established,
363
when possessed of only a few atoms of this curious substance, and the
accuracy of his analytical statements, were all confirmed when an
opportunity occurred of repeating his experiments upon this substance,
which can now be obtained in unlimited quantities.
CAROLINE.
What a satisfaction Sir H. Davy must have felt, when by an effort of
genius he succeeded in bringing to light and actually giving existence,
to these curious bodies, which without him might perhaps have ever
remained concealed from our view!
MRS. B.
The next substance which Sir H. Davy submitted to the influence of
the Voltaic battery was Soda, the other fixed alkali, which
yielded to the same powers of decomposition; from this alkali too,
a metallic substance was obtained, very analogous in its properties
to that which had been discovered in potash; Sir H. Davy has called it
SODIUM. It is rather heavier than
potassium, though considerably lighter than water; it is not so easily
fusible as potassium.
Encouraged by these extraordinary results, Sir H. Davy next performed
a series of beautiful experiments on Ammonia, or the volatile
alkali, which, from analogy, he was led to suspect might also contain
oxygen. This he soon ascertained to be the
364
fact, but he has not yet succeeded in obtaining the basis of ammonia in
a separate state; it is from analogy, and from the power which the
volatile alkali has, in its gaseous form, to oxydate iron, and also from
the amalgams which can be obtained from ammonia by various processes,
that the proofs of that alkali being also a metallic oxyd are
deduced.
Thus, then, the three alkalies, two of which had always been
considered as simple bodies, have now lost all claim to that title, and
I have accordingly classed the alkalies amongst the compounds, whose
properties we shall treat of in a future conversation.
EMILY.
What are the other newly discovered metals which you have alluded to
in your list of simple bodies?
MRS. B.
They are the metals of the earths which became next the object of Sir
H. Davy’s researches; these bodies had never yet been decomposed, though
they were strongly suspected not only of being compounds, but of being
metallic oxyds. From the circumstance of their incombustibility it was
conjectured, with some plausibility, that they might possibly be bodies
that had been already burnt.
CAROLINE.
And metals, when oxydated, become, to all appearance, a kind of
earthy substance.
365
MRS. B.
They have, besides, several features of resemblance with metallic
oxyds; Sir H. Davy had therefore great reason to be sanguine in his
expectations of decomposing them, and he was not disappointed. He could
not, however, succeed in obtaining the basis of the earths in a pure
separate state; but metallic alloys were formed with other metals, which
sufficiently proved the existence of the metallic basis of the
earths.
The last class of new metallic bodies which Sir H. Davy discovered
was obtained from the three undecompounded acids, the boracic, the
fluoric, and the muriatic acids; but as you are entirely unacquainted
with these bodies, I shall reserve the account of their
decomposition till we come to treat of their properties as acids.
Thus in the course of two years, by the unparalleled exertions of a
single individual, chemical science has assumed a new aspect. Bodies
have been brought to light which the human eye never before beheld, and
which might have remained eternally concealed under their impenetrable
disguise.
It is impossible at the present period to appreciate to their full
extent the consequences which science or the arts may derive from these
discoveries; we may, however, anticipate the most important results.
366
In chemical analysis we are now in possession of more energetic
agents of decomposition than were ever before known.
In geology new views are opened, which will probably operate a
revolution in that obscure and difficult science. It is already proved
that all the earths, and, in fact, the solid surface of this globe, are
metallic bodies mineralized by oxygen, and as our planet has been
calculated to be considerably more dense upon the whole than on the
surface, it is reasonable to suppose that the interior part is composed
of a metallic mass, the surface of which only has been mineralized by
the atmosphere.
The eruptions of volcanos, those stupendous problems of nature, admit
now of an easy explanation. For if the bowels of the earth are the grand
recess of these newly discovered inflammable bodies, whenever water
penetrates into them, combustions and explosions must take place; and it
is remarkable that the lava which is thrown out, is the very kind of
substance which might be expected to result from these combustions.
I must now take my leave of you; we have had a very long conversation
to-day, and I hope you will be able to recollect what you have learnt.
At our next interview we shall enter on a new subject.
END OF THE FIRST VOLUME.
Printed by A. Strahan,
Printers-Street, London.
CONVERSATIONS
ON
CHEMISTRY;
IN WHICH
THE ELEMENTS OF THAT SCIENCE
ARE
FAMILIARLY EXPLAINED
AND
ILLUSTRATED BY EXPERIMENTS.
IN TWO VOLUMES.
The Fifth Edition, revised, corrected, and
considerably enlarged.
VOL. II.
ON COMPOUND BODIES.

LONDON:
PRINTED FOR LONGMAN, HURST, REES, ORME, AND BROWN,
PATERNOSTER-ROW.
1817.
v
OF
THE SECOND VOLUME.
ON COMPOUND BODIES.
|
CONVERSATION XIII. |
|
| ON THE ATTRACTION OF COMPOSITION. |
Page
1 |
Of the laws which regulate the Phenomena of
the Attraction of Composition.—1. It takes place only between
Bodies of a different Nature.—2. Between the most minute
Particles only.—3. Between 2, 3, 4, or more Bodies.—Of
Compound or Neutral Salts.—4. Produces a Change of
Temperature.—5. The Properties which characterise Bodies in
their separate State, destroyed by Combination.—6. The Force
of Attraction estimated by that which is required by the Separation of
the Constituents.—7. Bodies have amongst themselves different
Degrees of Attraction.—Of simple elective and double elective
Attractions.—Of quiescent and divellent Forces.—Law of
definite Proportions.—Decomposition of Salts by Voltaic
Electricity. |
|
|
vi
CONVERSATION XIV. |
|
| ON ALKALIES. |
19 |
Of the Composition and general Properties of the
Alkalies.—Of Potash.—Manner of preparing
it.—Pearlash.—Soap.—Carbonat of Potash.—Chemical
Nomenclature.—Solution of Potash.—Of Glass.—Of Nitrat
of Potash or Saltpetre.—Effect of Alkalies on Vegetable
Colours.—Of Soda.—Of Ammonia or Volatile
Alkali.—Muriat of Ammonia.—Ammoniacal Gas.—Composition
of Ammonia.—Hartshorn and Sal Volatile.—Combustion of Ammoniacal
Gas. |
|
|
CONVERSATION XV. |
|
| ON EARTHS. |
44 |
Composition of the Earths.—Of their
Incombustibility.—Form the Basis of all Minerals.—Their
Alkaline Properties.—Silex; its Properties and Uses in the
Arts.—Alumine; its Uses in Pottery, &c.—Alkaline
Earths.—Barytes.—Lime; its extensive chemical Properties and
Uses in the Arts.—Magnesia.—Strontian. |
|
|
CONVERSATION XVI. |
|
| ON ACIDS. |
69 |
Nomenclature of the Acids.—Of the Classification of
Acids.—1st Class—Acids of simple and known Radicals, or
Mineral Acids.—2d Class—Acids of double Radicals, or
Vegetable Acids.—3d Class—Acids of triple Radicals or
Animal Acids.—Of the Decomposition of Acids of the 1st Class by
Combustible bodies. |
|
|
vii
CONVERSATION XVII. |
|
OF THE SULPHURIC AND PHOSPHORIC ACIDS:
OR, THE COMBINATIONS OF OXYGEN WITH SULPHUR AND WITH PHOSPHORUS; AND OF
THE SULPHATS AND PHOSPHATS. |
80 |
Of the Sulphuric Acid.—Combustion of Animal or Vegetable
Bodies by this Acid.—Method of preparing it.—The Sulphurous
Acid obtained in the Form of Gas.—May be obtained from Sulphuric
Acid.—May be reduced to Sulphur.—Is absorbable by
Water.—Destroys Vegetable Colours.—Oxyd of Sulphur.—Of
Salts in general.—Sulphats.—Sulphat of Potash, or Sal
Polychrest.—Cold produced by the melting of Salts.—Sulphat
of Soda, or Glauber’s Salt.—Heat evolved during the Formation of
Salts.—Crystallisation of Salts.—Water of
Crystallisation.—Efflorescence and Deliquescence of
Salts.—Sulphat of Lime, Gypsum or Plaister of Paris.—Sulphat
of Magnesia.—Sulphat of Alumine, or Alum.—Sulphat of
Iron.—Of Ink.—Of the Phosphoric and Phosphorous
Acids.—Phosphorus obtained from Bones.—Phosphat of
Lime. |
|
|
CONVERSATION XVIII. |
|
OF THE NITRIC AND CARBONIC ACIDS: OR THE
COMBINATION OF OXYGEN WITH NITROGEN AND WITH CARBON; AND OF THE NITRATS
AND CARBONATS. |
100 |
Nitrogen susceptible of various Degrees of
Acidification.—Of the Nitric Acid.—Its Nature and
Composition discovered by Mr. Cavendish.—Obtained from Nitrat
of Potash.—Aqua Fortis.—Nitric Acid may be converted
viii
into Nitrous Acid.—Nitric Oxyd Gas.—Its Conversion into
Nitrous Acid Gas.—Used as an Eudiometrical Test.—Gaseous
Oxyd of Nitrogen, or exhilarating Gas, obtained from Nitrat of
Ammonia.—Its singular Effects on being
respired.—Nitrats.—Of Nitrat of Potash, Nitre or
Saltpetre.—Of Gunpowder.—Causes of
Detonation.—Decomposition of
Nitre.—Deflagration.—Nitrat of Ammonia.—Nitrat of
Silver.—Of the Carbonic Acid.—Formed by the Combustion of
Carbon.—Constitutes a component Part of the
Atmosphere.—Exhaled in some Caverns.—Grotto del
Cane.—Great Weight of this Gas.—Produced from calcareous
Stones by Sulphuric Acid.—Deleterious Effects of this Gas when
respired.—Sources which keep up a Supply of this Gas in the
Atmosphere.—Its Effects on Vegetation.—Of the Carbonats of
Lime; Marble, Chalk, Shells, Spars, and calcareous Stones. |
|
|
CONVERSATION XIX. |
|
| ON THE BORACIC, FLUORIC, MURIATIC, AND OXYGENATED
MURIATIC ACIDS; AND ON MURIATS. |
131 |
On the Boracic Acid.—Its Decomposition by Sir H.
Davy.—Its Basis Boracium.—Its Recomposition.—Its Uses
in the Arts.—Borax or Borat of Soda.—Of the Fluoric
Acid.—Obtained from Fluor; corrodes Siliceous Earth; its supposed
Composition.—Fluorine; its supposed Basis.—Of the Muriatic
Acid.—Obtained from Muriats.—Its gaseous Form.—Is
absorbable by Water.—Its Decomposition.—Is susceptible of a
stronger Degree of Oxygenation.—Oxygenated Muriatic
Acid.—Its gaseous Form and other Properties.—Combustion of
Bodies in this Gas.—It dissolves Gold.—Composition
ix
of Aqua Regia.—Oxygenated Muriatic Acid destroys all
Colours.—Sir H. Davy’s Theory of the Nature of Muriatic and
Oxymuriatic Acid.—Chlorine.—Used for Bleaching and for
Fumigations.—Its offensive Smell,
&c.—Muriats.—Muriat of Soda, or common
Salt.—Muriat of Ammonia.—Oxygenated Muriat of
Potash.—Detonates with Sulphur, Phosphorus,
&c.—Experiment of burning Phosphorus under Water by means of
this Salt and of Sulphuric Acid. |
|
|
CONVERSATION XX. |
|
| ON THE NATURE AND COMPOSITION OF VEGETABLES. |
162 |
Of organised Bodies.—Of the Functions of
Vegetables.—Of the Elements of Vegetables.—Of the Materials
of Vegetables.—Analysis of Vegetables.—Of
Sap.—Mucilage, or Gum.—Sugar.—Manna, and
Honey.—Gluten.—Vegetable Oils.—Fixed Oils, Linseed,
Nut, and Olive Oils.—Volatile Oils, forming Essences and
Perfumes.—Camphor.—Resins and Varnishes.—Pitch, Tar,
Copal, Mastic, &c.—Gum Resins.—Myrrh, Assafœtida,
&c.—Caoutchouc, or Gum Elastic.—Extractive colouring
Matter; its Use in the Arts of Dyeing and Painting.—Tannin; its
Use in the Art of preparing Leather.—Woody Fibre.—Vegetable
Acids.—The Alkalies and Salts contained in Vegetables. |
|
|
CONVERSATION XXI. |
|
| ON THE DECOMPOSITION OF VEGETABLES. |
202 |
Of Fermentation in general.—Of the Saccharine Fermentation,
the Product of which is Sugar.—Of the Vinous Fermentation, the
Product of which is Wine.
x
—Alcohol, or Spirit of Wine.—Analysis of Wine by
Distillation.—Of Brandy, Rum, Arrack, Gin, &c.—Tartrit
of Potash, or Cream of Tartar.—Liqueurs.—Chemical Properties
of Alcohol.—Its Combustion.—Of Ether.—Of the Acetous
Fermentation, the Product of which is Vinegar.—Fermentation of
Bread.—Of the Putrid Fermentation, which reduces Vegetables to
their Elements.—Spontaneous Succession of these
Fermentations.—Of Vegetables said to be petrified.—Of
Bitumens: Naphtha, Asphaltum, Jet, Coal, Succin, or Yellow
Amber.—Of Fossil Wood, Peat, and Turf. |
|
|
CONVERSATION XXII. |
|
| HISTORY OF VEGETATION. |
243 |
Connexion between the Vegetable and Animal Kingdoms.—Of
Manures.—Of Agriculture.—Inexhaustible Sources of Materials
for the Purposes of Agriculture.—Of sowing Seed.—Germination
of the Seed.—Function of the Leaves of Plants.—Effects of
Light and Air on Vegetation.—Effects of Water on
Vegetation.—Effects of Vegetation on the
Atmosphere.—Formation of Vegetable Materials by the Organs of
Plants.—Vegetable Heat.—Of the Organs of Plants.—Of
the Bark, consisting of Epidermis, Parenchyma, and Cortical
Layers.—Of Alburnum, or Wood.—Leaves, Flowers, and
Seeds.—Effects of the Season on Vegetation.—Vegetation of
Evergreens in Winter. |
|
|
CONVERSATION XXIII. |
|
| ON THE COMPOSITION OF ANIMALS. |
276 |
Elements of Animals.—Of the principal Materials of Animals,
viz.—Gelatine, Albumen, Fibrine, Mucus.
xi
—Of Animal Acids.—Of Animal Colours, Prussian Blue, Carmine,
and Ivory Black. |
|
|
CONVERSATION XXIV. |
|
| ON THE ANIMAL ECONOMY. |
297 |
Of the principal Animal Organs.—Of Bones, Teeth, Horns,
Ligaments, and Cartilage.—Of the Muscles, constituting the Organs
of Motion.—Of the Vascular System, for the Conveyance of
Fluids.—Of the Glands, for the Secretion of Fluids.—Of the
Nerves, constituting the Organs of Sensation.—Of the Cellular
Substance which connects the several Organs.—Of the Skin. |
|
|
CONVERSATION XXV. |
|
| ON ANIMALISATION, NUTRITION, AND
RESPIRATION. |
314 |
Digestion.—Solvent Power of the Gastric
Juice.—Formation of a Chyle.—Its Assimilation, or Conversion
into Blood.—Of Respiration.—Mechanical Process of
Respiration.—Chemical Process of Respiration.—Of the
Circulation of the Blood.—Of the Functions of the Arteries, the
Veins, and the Heart.—Of the Lungs.—Effects of Respiration
on the Blood. |
|
|
CONVERSATION XXVI. |
|
| ON ANIMAL HEAT; AND OF VARIOUS ANIMAL
PRODUCTS. |
336 |
Of the Analogy of Combustion and Respiration.—Animal Heat
evolved in the Lungs.—Animal Heat evolved in the
Circulation.—Heat produced by Fever.—Perspiration.
xii
—Heat produced by Exercise.—Equal Temperature of Animals at
all Seasons.—Power of the Animal Body to resist the Effects of
Heat.—Cold produced by Perspiration.—Respiration of Fish and
of Birds.—Effects of Respiration on Muscular Strength.—Of
several Animal Products, viz. Milk, Butter, and Cheese; Spermaceti;
Ambergris; Wax; Lac; Silk; Musk; Civet; Castor.—Of the putrid
Fermentation.—Conclusion. |
|
CONVERSATIONS
ON
CHEMISTRY.

MRS. B.
Having completed our examination of the
simple or elementary bodies, we are now to proceed to those of a
compound nature; but before we enter on this extensive subject, it will
be necessary to make you acquainted with the principal laws by which
chemical combinations are governed.
You recollect, I hope, what we formerly said of the nature of the
attraction of composition, or chemical attraction, or affinity, as it is
also called?
EMILY.
Yes, I think perfectly; it is the attraction that
2
subsists between bodies of a different nature, which occasions them to
combine and form a compound, when they come in contact, and, according
to Sir H. Davy’s opinion, this effect is produced by the attraction of
the opposite electricities, which prevail in bodies of different
kinds.
MRS. B.
Very well; your definition comprehends the first law of chemical
attraction, which is, that it takes place only between bodies of a
different nature; as, for instance, between an acid and an alkali;
between oxygen and a metal, &c.
CAROLINE.
That we understand of course; for the attraction between particles of
a similar nature is that of aggregation, or cohesion, which is
independent of any chemical power.
MRS. B.
The 2d law of chemical attraction is, that it takes place only
between the most minute particles of bodies; therefore, the more you
divide the particles of the bodies to be combined, the more readily they
act upon each other.
CAROLINE.
That is again a circumstance which we might
3
have supposed, for the finer the particles of the two substances are,
the more easily and perfectly they will come in contact with each other,
which must greatly facilitate their union. It was for this purpose, you
said, that you used iron filings, in preference to wires or pieces of
iron, for the decomposition of water.
MRS. B.
It was once supposed that no mechanical power could divide bodies
into particles sufficiently minute for them to act on each other; and
that, in order to produce the extreme division requisite for a chemical
action, one, if not both of the bodies, should be in a fluid state.
There are, however, a few instances in which two solid bodies, very
finely pulverized, exert a chemical action on one another; but such
exceptions to the general rule are very rare indeed.
EMILY.
In all the combinations that we have hitherto seen, one of the
constituents has, I believe, been either liquid or aëriform. In
combustions, for instance, the oxygen is taken from the atmosphere, in
which it existed in the state of gas; and whenever we have seen acids
combine with metals or with alkalies, they were either in a liquid or an
aëriform state.
4
MRS. B.
The 3d law of chemical attraction is, that it can take place
between two, three, four, or even a greater number of bodies.
CAROLINE.
Oxyds and acids are bodies composed of two constituents; but I
recollect no instance of the combination of a greater number of
principles.
MRS. B.
The compound salts, formed by the union of the metals with acids, are
composed of three principles. And there are salts formed by the
combination of the alkalies with the earths which are of a similar
description.
CAROLINE.
Are they of the same kind as the metallic salts?
MRS. B.
Yes; they are very analogous in their nature, although different in
many of their properties.
A methodical nomenclature, similar to that of the acids, has been
adopted for the compound salts. Each individual salt derives its name
from its constituent parts, so that every name implies a knowledge of
the composition of the salt.
The three alkalies, the alkaline earths, and the
5
metals, are called salifiable bases or radicals; and the
acids, salifying principles. The name of each salt is composed
both of that of the acid and the salifiable base; and it terminates in
at or it, according to the degree of the oxygenation of
the acid. Thus, for instance, all those salts which are formed by the
combination of the sulphuric acid with any of the salifiable bases are
called sulphats, and the name of the radical is added for the
specific distinction of the salt; if it be potash, it will compose a
sulphat of potash; if ammonia, sulphat of
ammonia, &c.
EMILY.
The crystals which we obtained from the combination of iron and
sulphuric acid were therefore sulphat of iron?
MRS. B.
Precisely; and those which we prepared by dissolving copper in nitric
acid, nitrat of copper, and so on.—But this is not all; if
the salt be formed by that class of acids which ends in ous,
(which you know indicates a less degree of oxygenation,) the termination
of the name of the salt will be in it, as sulphit of
potash, sulphit of ammonia, &c.
EMILY.
There must be an immense number of compound
6
salts, since there is so great a variety of salifiable radicals, as well
as of salifying principles.
MRS. B.
Their real number cannot be ascertained, since it increases every
day. But we must not proceed further in the investigation of the
compound salts, until we have completed the examination of the nature of
the ingredients of which they are composed.
The 4th law of chemical attraction is, that a change of
temperature always takes place at the moment of combination. This
arises from the extrication of the two electricities in the form of
caloric, which takes place when bodies unite; and also sometimes in part
from a change of capacity of the bodies for heat, which always takes
place when the combination is attended with an increase of density, but
more especially when the compound passes from the liquid to the solid
form. I shall now show you a striking instance of a change of
temperature from chemical union, merely by pouring some nitrous acid on
this small quantity of oil of turpentine—the oil will instantly
combine with the oxygen of the acid, and produce a considerable change
of temperature.
CAROLINE.
What a blaze! The temperature of the oil and
7
the acid must be greatly raised, indeed, to produce such a violent
combustion.
MRS. B.
There is, however, a peculiarity in this combustion, which is, that
the oxygen, instead of being derived from the atmosphere alone, is
principally supplied by the acid itself.
EMILY.
And are not all combustions instances of the change of temperature
produced by the chemical combination of two bodies?
MRS. B.
Undoubtedly; when oxygen loses its gaseous form, in order to combine
with a solid body, it becomes condensed, and the caloric evolved
produces the elevation of temperature. The specific gravity of bodies is
at the same time altered by chemical combination; for in consequence of
a change of capacity for heat, a change of density must be
produced.
CAROLINE.
That was the case with the sulphuric acid and water, which, by being
mixed together, gave out a great deal of heat, and increased in
density.
8
MRS. B.
The 5th law of chemical attraction is, that the properties which
characterise bodies, when separate, are altered or destroyed by their
combination.
CAROLINE.
Certainly; what, for instance, can be so different from water as the
hydrogen and oxygen gases?
EMILY.
Or what more unlike sulphat of iron than iron or sulphuric acid?
MRS. B.
Every chemical combination is an illustration of this rule. But let
us proceed—
The 6th law is, that the force of chemical affinity between the
constituents of a body is estimated by that which is required for their
separation. This force is not always proportional to the facility
with which bodies unite; for manganese, for instance, which, you know,
is so much disposed to unite with oxygen that it is never found in a
metallic state, yields it more easily than any other metal.
EMILY.
But, Mrs. B., you speak of estimating the force of attraction between
bodies, by the force required
9
to separate them; how can you measure these forces?
MRS. B.
They cannot be precisely measured, but they are comparatively
ascertained by experiment, and can be represented by numbers which
express the relative degrees of attraction.
The 7th law is, that bodies have amongst themselves different
degrees of attraction. Upon this law, (which you may have discovered
yourselves long since,) the whole science of chemistry depends; for it
is by means of the various degrees of affinity which bodies have for
each other, that all the chemical compositions and decompositions are
effected. Every chemical fact or experiment is an instance of the same
kind; and whenever the decomposition of a body is performed by the
addition of any single new substance, it is said to be effected by
simple elective attractions. But it often happens that no simple
substance will decompose a body, and that, in order to effect this, you
must offer to the compound a body which is itself composed of two, or
sometimes three principles, which would not, each separately, perform
the decomposition. In this case there are two new compounds formed in
consequence of a reciprocal decomposition and recomposition. All
instances of this kind are called double elective
attractions.
10
CAROLINE.
I confess I do not understand this clearly.
MRS. B.
You will easily comprehend it by the assistance of this diagram, in
which the reciprocal forces of attraction are represented by
numbers:
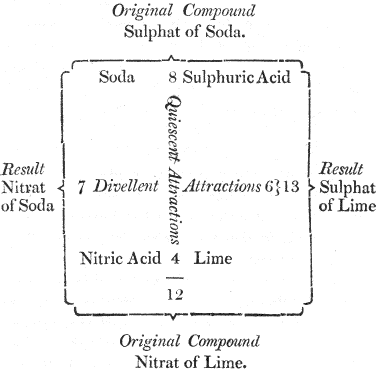
We here suppose that we are to decompose sulphat of soda; that is, to
separate the acid from
11
the alkali; if, for this purpose, we add some lime, in order to make it
combine with the acid, we shall fail in our attempt, because the soda
and the sulphuric acid attract each other by a force which is superior,
and (by way of supposition) is represented by the number 8; while
the lime tends to unite with this acid by an affinity equal only to the
number 6. It is plain, therefore, that the sulphat of soda will not
be decomposed, since a force equal to 8 cannot be overcome by a force
equal only to 6.
CAROLINE.
So far, this appears very clear.
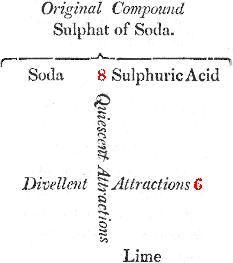
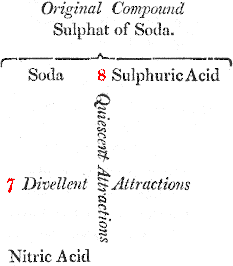
MRS. B.
If, on the other hand, we endeavour to decompose this salt by nitric
acid, which tends to combine with soda, we shall be equally
unsuccessful, as nitric acid tends to unite with the alkali by a force
equal only to 7.
In neither of these cases of simple elective attraction, therefore,
can we accomplish our purpose. But let us previously combine together
the lime and nitric acid, so as to form a nitrat of lime,
a compound salt, the constituents of which are united by a power
equal to 4. If then we present this compound to the sulphat of soda,
a decomposition will ensue, because the sum of the forces
12
which tend to preserve the two salts in their actual state is not equal
to that of the forces which tend to decompose them, and to form new
combinations. The nitric acid, therefore, will combine with the soda,
and the sulphuric acid with the lime.
CAROLINE.
I understand you now very well. This double effect takes place
because the numbers 8 and 4, which represent the degrees of attraction
of the constituents of the two original salts, make a sum less than the
numbers 7 and 6, which represent the degrees of attraction of the two
new compounds that will in consequence be formed.
MRS. B.
Precisely so.
CAROLINE.
But what is the meaning of quiescent and divellent
forces, which are written in the diagram?
MRS. B.
Quiescent forces are those which tend to preserve compounds in a
state of rest, or such as they actually are: divellent forces, those
which tend to destroy that state of combination, and to form new
compounds.
These are the principal circumstances relative to the doctrine of
chemical attractions, which
13
have been laid down as rules by modern chemists; a few others might
be mentioned respecting the same theory, but of less importance, and
such as would take us too far from our plan. I should, however, not
omit to mention that Mr. Berthollet, a celebrated French chemist,
has questioned the uniform operation of elective attraction, and has
advanced the opinion, that, in chemical combinations, the changes which
take place depend not only upon the affinities, but also, in some
degree, on the respective quantities of the substances concerned, on the
heat applied during the process, and some other circumstances.
CAROLINE.
In that case, I suppose, there would hardly be two compounds exactly
similar, though composed of the same materials?
MRS. B.
On the contrary, it is found that a remarkable uniformity prevails,
as to proportions, between the ingredients of bodies of similar
composition. Thus water, as you may recollect to have seen in a former
conversation, is composed of two volumes of hydrogen gas to one of
oxygen, and this is always found to be precisely the proportion of its
constituents, from whatever source the water be derived. The same
uniformity prevails with regard to the various salts; the acid and
alkali, in
14
each kind of salt, being always found to combine in the same
proportions. Sometimes, it is true, the same acid, and the same alkali,
are capable of making two distinct kinds of salts; but in all these
cases it is found that one of the salts contains just twice, or in some
instances, thrice as much acid, or alkali, as the other.
EMILY.
If the proportions in which bodies combine are so constant and so
well defined, how can Mr. Berthollet’s remark be reconciled with this
uniform system of combination?
MRS. B.
Great as that philosopher’s authority is in chemistry, it is now
generally supposed that his doubts on this subject were in a great
degree groundless, and that the exceptions he has observed in the laws
of definite proportions, have been only apparent, and may be accounted
for consistently with those laws.
CAROLINE.
Pray, Mrs. B., can you decompose a salt by means of electricity, in
the same way as we decompose water?
MRS. B.
Undoubtedly; and I am glad this question occurred to you, because it
gives me an opportunity
15
of showing you some very interesting experiments on the subject.
If we dissolve a quantity, however small, of any salt in a glass of
water, and if we plunge into it the extremities of the wires which
proceed from the two ends of the Voltaic battery, the salt will be
gradually decomposed, the acid being attracted by the positive, and the
alkali by the negative wire.
EMILY.
But how can you render that decomposition perceptible?
MRS. B.
By placing in contact with the extremities of each wire, in the
solution, pieces of paper stained with certain vegetable colours, which
are altered by the contact of an acid or an alkali. Thus this blue
vegetable preparation called litmus becomes red when touched by an acid;
and the juice of violets becomes green by the contact of an alkali.
But the experiment can be made in a much more distinct manner, by
receiving the extremities of the wires into two different vessels, so
that the alkali shall appear in one vessel and the acid in the
other.
CAROLINE.
But then the Voltaic circle will not be completed; how can any effect
be produced?
16
MRS. B.
You are right; I ought to have added that the two vessels must be
connected together by some interposed substance capable of conducting
electricity. A piece of moistened cotton-wick answers this purpose
very well. You see that the cotton (Plate XIII. fig. 2. c.) has one end immersed
in one glass and the other end in the other, so as to establish a
communication between any fluids contained in them. We shall now put
into each of the glasses a little glauber salt, or sulphat of soda,
(which consists of an acid and an alkali,) and then we shall fill the
glasses with water, which will dissolve the salt. Let us now connect the
glasses by means of the wires (e, d,) with the two ends of the
battery, thus . . . .
Plate XIII.
Vol. II. page 16.
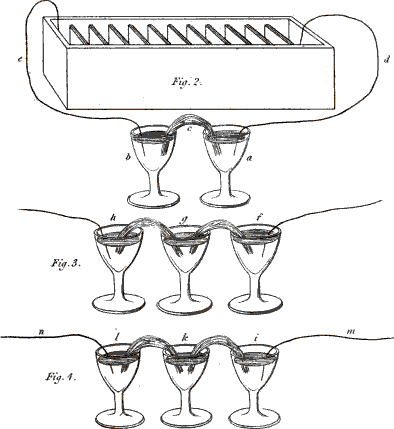
Fig. 2. 3 & 4. Instances of Chemical decomposition by the
Voltaic Battery.
Larger view
(complete Plate)
CAROLINE.
The wires are already giving out small bubbles; is this owing to the
decomposition of the salt?
MRS. B.
No; these are bubbles produced by the decomposition of the water, as
you saw in a former experiment. In order to render the separation of the
acid from the alkali visible, I pour into the glass (a), which
is connected with the positive wire, a few drops of a solution of
litmus,
17
which the least quantity of acid turns red; and into the other
glass (b), which is connected with the negative wire, I pour a
few drops of the juice of violets . . . .
EMILY.
The blue solution is already turning red all round the wire.
CAROLINE.
And the violet solution is beginning to turn green. This is indeed
very singular!
MRS. B.
You will be still more astonished when we vary the experiment in this
manner:—These three glasses (fig. 3. f, g, h,) are, as
in the former instance, connected together by wetted cotton, but the
middle one alone contains a saline solution, the two others containing
only distilled water, coloured as before by vegetable infusions. Yet, on
making the connection with the battery, the alkali will appear in the
negative glass (h), and the acid in the positive glass (f),
though neither of them contained any saline matter.
EMILY.
So that the acid and alkali must be conveyed right and left from the
central glass, into the
18
other glasses, by means of the connecting moistened cotton?
MRS. B.
Exactly so; and you may render the experiment still more striking, by
putting into the central glass (k, fig. 3.) an alkaline
solution, the glauber salt being placed into the negative
glass (l), and the positive glass (i) containing only water. The
acid will be attracted by the positive wire (m), and will actually
appear in the vessel (i), after passing through the alkaline
solution (k), without combining with it, although, you know, acids
and alkalies are so much disposed to combine.—But this
conversation has already much exceeded our usual limits, and we cannot
enlarge more upon this interesting subject at present.
19

MRS. B.
Having now given you some idea of the laws
by which chemical attractions are governed, we may proceed to the
examination of bodies which are formed in consequence of these
attractions.
The first class of compounds that present themselves to our notice,
in our gradual ascent to the most complicated combinations, are bodies
composed of only two principles. The sulphurets, phosphurets, carburets,
&c. are of this description; but the most numerous and important of
these compounds are the combinations of oxygen with the various simple
substances with which it has a tendency to unite. Of these you have
already acquired some knowledge, but it will be necessary to enter into
further particulars respecting the nature and properties of those most
deserving our notice. Of this class are the ALKALIES
20
and the EARTHS, which we shall
successively examine.
We shall first take a view of the alkalies, of which there are three,
viz. POTASH, SODA, and AMMONIA.
The two first are called fixed alkalies, because they exist in a
solid form at the temperature of the atmosphere, and require a great
heat to be volatilised. They consist, as you already know, of metallic
bases combined with oxygen. In potash, the proportions are about
eighty-six parts of potassium to fourteen of oxygen; and in soda,
seventy-seven parts of sodium to twenty-three of oxygen. The third
alkali, ammonia, has been distinguished by the name of volatile
alkali, because its natural form is that of gas. Its composition is
of a more complicated nature, of which we shall speak hereafter.
Some of the earths bear so strong a resemblance in their properties
to the alkalies, that it is difficult to know under which head to place
them. The celebrated French chemist, Fourcroy, has classed two of them
(barytes and strontites) with the alkalies; but as lime and magnesia
have almost an equal title to that rank, I think it better not to
separate them, and therefore have adopted the common method of classing
them with the earths, and of distinguishing them by the name of
alkaline earths.
The general properties of alkalies are, an acrid
21
burning taste, a pungent smell, and a caustic action on the skin
and flesh.
CAROLINE.
I wonder they should be caustic, Mrs. B., since they contain so
little oxygen.
MRS. B.
Whatever substance has an affinity for any one of the constituents of
animal matter, sufficiently powerful to decompose it, is entitled to the
appellation of caustic. The alkalies, in their pure state, have a very
strong attraction for water, for hydrogen, and for carbon, which, you
know, are the constituent principles of oil, and it is chiefly by
absorbing these substances from animal matter that they effect its
decomposition; for, when diluted with a sufficient quantity of water, or
combined with any oily substance, they lose their causticity.
But, to return to the general properties of alkalies—they
change, as we have already seen, the colour of syrup of violets, and
other blue vegetable infusions, to green; and have, in general,
a very great tendency to unite with acids, although the respective
qualities of these two classes of bodies form a remarkable contrast.
We shall examine the result of the combination of acids and alkalies
more particularly hereafter.
22
It will be sufficient at present to inform you, that whenever acids are
brought in contact with alkalies, or alkaline earths, they unite with a
remarkable eagerness, and form compounds perfectly different from either
of their constituents; these bodies are called neutral or
compound salts.
The dry white powder which you see in this phial is pure caustic
POTASH; it is very difficult to
preserve it in this state, as it attracts, with extreme avidity, the
moisture from the atmosphere, and if the air were not perfectly
excluded, it would, in a very short time, be actually melted.
EMILY.
It is then, I suppose, always found in a liquid state?
MRS. B.
No; it exists in nature in a great variety of forms and combinations,
but is never found in its pure separate state; it is combined with
carbonic acid, with which it exists in every part of the vegetable
kingdom, and is most commonly obtained from the ashes of vegetables,
which are the residue that remains after all the other parts have been
volatilised by combustion.
CAROLINE.
But you once said, that after all the volatile
23
parts of a vegetable were evaporated, the substance that remained was
charcoal?
MRS. B.
I am surprised that you should still confound the processes of
volatilisation and combustion. In order to procure charcoal, we
evaporate such parts as can be reduced to vapour by the operation of
heat alone; but when we burn the vegetable, we burn the carbon
also, and convert it into carbonic acid gas.
CAROLINE.
That is true; I hope I shall make no more mistakes in my favourite
theory of combustion.
MRS. B.
Potash derives its name from the pots in which the vegetables,
from which it was obtained, used formerly to be burnt; the alkali
remained mixed with the ashes at the bottom, and was thence called
potash.
EMILY.
The ashes of a wood-fire, then, are potash, since they are vegetable
ashes?
MRS. B.
They always contain more or less potash, but are very far from
consisting of that substance alone, as they are a mixture of various
earths and salts
24
which remain after the combustion of vegetables, and from which it is
not easy to separate the alkali in its pure form. The process by which
potash is obtained, even in the imperfect state in which it is used in
the arts, is much more complicated than simple combustion. It was once
deemed impossible to separate it entirely from all foreign substances,
and it is only in chemical laboratories that it is to be met with in the
state of purity in which you find it in this phial. Wood-ashes are,
however, valuable for the alkali which they contain, and are used for
some purposes without any further preparation. Purified in a certain
degree, they make what is commonly called pearlash, which is of
great efficacy in taking out grease, in washing linen, &c.; for
potash combines readily with oil or fat, with which it forms a compound
well known to you under the name of soap.
CAROLINE.
Really! Then I should think it would be better to wash all linen with
pearlash than with soap, as, in the latter case, the alkali being
already combined with oil, must be less efficacious in extracting
grease.
MRS. B.
Its effect would be too powerful on fine linen, and would injure its
texture; pearlash is therefore only used for that which is of a strong
coarse
25
kind. For the same reason you cannot wash your hands with plain potash;
but, when mixed with oil in the form of soap, it is soft as well as
cleansing, and is therefore much better adapted to the purpose.
Caustic potash, as we already observed, acts on the skin, and animal
fibre, in virtue of its attraction for water and oil, and converts all
animal matter into a kind of saponaceous jelly.
EMILY.
Are vegetables the only source from which potash can be derived?
MRS. B.
No: for though far most abundant in vegetables, it is by no means
confined to that class of bodies, being found also on the surface of the
earth, mixed with various minerals, especially with earths and stones,
whence it is supposed to be conveyed into vegetables by the roots of the
plant. It is also met with, though in very small quantities, in some
animal substances. The most common state of potash is that of
carbonat; I suppose you understand what that is?
EMILY.
I believe so; though I do not recollect that you ever mentioned the
word before. If I am not mistaken,
26
it must be a compound salt, formed by the union of carbonic acid with
potash.
MRS. B.
Very true; you see how admirably the nomenclature of modern chemistry
is adapted to assist the memory; when you hear the name of a compound,
you necessarily learn what are its constituent parts; and when you are
acquainted with these constituents, you can immediately name the
compound which they form.
CAROLINE.
Pray, how were bodies arranged and distinguished before this
nomenclature was introduced?
MRS. B.
Chemistry was then a much more difficult study; for every substance
had an arbitrary name, which it derived either from the person who
discovered it, as Glauber’s salts for instance; or from some
other circumstance relative to it, though quite unconnected with its
real nature, as potash.
These names have been retained for some of the simple bodies; for as
this class is not numerous, and therefore can easily be remembered, it
has not been thought necessary to change them.
EMILY.
Yet I think it would have rendered the new nomenclature more complete
to have methodised
27
the names of the elementary, as well as of the compound bodies, though
it could not have been done in the same manner. But the names of the
simple substances might have indicated their nature, or, at least, some
of their principal properties; and if, like the acids and compound
salts, all the simple bodies had a similar termination, they would have
been immediately known as such. So complete and regular a nomenclature
would, I think, have given a clearer and more comprehensive view of
chemistry than the present, which is a medley of the old and new
terms.
MRS. B.
But you are not aware of the difficulty of introducing into science
an entire set of new terms; it obliges all the teachers and professors
to go to school again, and if some of the old names, that are least
exceptionable, were not left as an introduction to the new ones, few
people would have had industry and perseverance enough to submit to the
study of a completely new language; and the inferior classes of artists,
who can only act from habit and routine, would, at least for a time,
have felt material inconvenience from a total change of their habitual
terms. From these considerations, Lavoisier and his colleagues, who
invented the new nomenclature, thought it most prudent to leave a few
links of the old chain, in
28
order to connect it with the new one. Besides, you may easily conceive
the inconvenience which might arise from giving a regular nomenclature
to substances, the simple nature of which is always uncertain; for the
new names might, perhaps, have proved to have been founded in error.
And, indeed, cautious as the inventors of the modern chemical language
have been, it has already been found necessary to modify it in many
respects. In those few cases, however, in which new terms have been
adopted to designate simple bodies, these names have been so contrived
as to indicate one of the chief properties of the body in question; this
is the case with oxygen, which, as I explained to you, signifies
generator of acids; and hydrogen generator of water. If all the
elementary bodies had a similar termination, as you propose, it would be
necessary to change the name of any that might hereafter be found of a
compound nature, which would be very inconvenient in this age of
discovery.
But to return to the alkalies.—We shall now try to melt some of
this caustic potash in a little water, as a circumstance occurs during
its solution very worthy of observation.—Do you feel the heat that
is produced?
CAROLINE.
Yes, I do; but is not this directly contrary to
29
our theory of latent heat, according to which heat is disengaged when fluids become
solid, and cold produced when solids are melted?
MRS. B.
The latter is really the case in all solutions; and if the solution
of caustic alkalies seems to make an exception to the rule, it does not,
I believe, form any solid objection to the theory. The matter may
be explained thus: When water first comes in contact with the potash, it
produces an effect similar to the slaking of lime, that is, the water is
solidified in combining with the potash, and thus loses its latent heat;
this is the heat that you now feel, and which is, therefore, produced
not by the melting of the solid, but by the solidification of the fluid.
But when there is more water than the potash can absorb and solidify,
the latter then yields to the solvent power of the water; and if we do
not perceive the cold produced by its melting, it is because it is
counterbalanced by the heat previously disengaged.*
A very remarkable property of potash is the
30
formation of glass by its fusion with siliceous earth. You are not yet
acquainted with this last substance, further than its being in the list
of simple bodies. It is sufficient, for the present, that you should
know that sand and flint are chiefly composed of it; alone, it is
infusible, but mixed with potash, it melts when exposed to the heat of a
furnace, combines with the alkali, and runs into glass.
CAROLINE.
Who would ever have supposed that the same substance which converts
transparent oil into such an opake body as soap, should transform that
opake substance, sand, into transparent glass!
MRS. B.
The transparency, or opacity of bodies, does not, I conceive,
depend so much upon their intimate nature, as upon the arrangement of
their particles: we cannot have a more striking instance of this, than
is afforded by the different states of carbon, which, though it commonly
appears in the form of a black opake body, sometimes assumes the most
dazzling transparent form in nature, that of diamond, which, you
recollect, is carbon, and which, in all probability, derives its
beautiful transparency from the peculiar arrangement of its particles
during their crystallisation.
31
EMILY.
I never should have supposed that the formation of glass was so
simple a process as you describe it.
MRS. B.
It is by no means an easy operation to make perfect glass; for if the
sand, or flint, from which the siliceous earth is obtained, be mixed
with any metallic particles, or other substance, which cannot be
vitrified, the glass will be discoloured, or defaced, by opake
specks.
CAROLINE.
That, I suppose, is the reason why objects so often appear irregular
and shapeless through a common glass-window.
MRS. B.
This species of imperfection proceeds, I believe, from another cause.
It is extremely difficult to prevent the lower part of the vessels, in
which the materials of glass are fused, from containing a more dense
vitreous matter than the upper, on account of the heavier ingredients
falling to the bottom. When this happens, it occasions the appearance of
veins or waves in the glass, from the difference of density in its
several parts, which produces an irregular refraction of the rays of
light that pass through it.
32
Another species of imperfection sometimes arises from the fusion not
being continued for a length of time sufficient to combine the two
ingredients completely, or from the due proportion of potash and silex
(which are as two to one) not being carefully observed; the glass, in
those cases, will be liable to alteration from the action of the air, of
salts, and especially of acids, which will effect its decomposition by
combining with the potash, and forming compound salts.
EMILY.
What an extremely useful substance potash is!
MRS. B.
Besides the great importance of potash in the manufactures of glass
and soap, it is of very considerable utility in many of the other arts,
and in its combinations with several acids, particularly the nitric,
with which it forms saltpetre.
CAROLINE.
Then saltpetre must be a nitrat of potash? But we are not yet
acquainted with the nitric acid?
MRS. B.
We shall therefore defer entering into the particulars of these
combinations till we come to a general review of the compound salts. In
order
33
to avoid confusion, it will be better at present to confine ourselves to
the alkalies.
EMILY.
Cannot you show us the change of colour which you said the alkalies
produced on blue vegetable infusions?
MRS. B.
Yes; very easily. I shall dip a piece of white paper into this syrup
of violets, which, you see, is of a deep blue, and dyes the paper of the
same colour.—As soon as it is dry, we shall dip it into a solution
of potash, which, though itself colourless, will turn the paper
green—
CAROLINE.
So it has, indeed! And do the other alkalies produce a similar
effect?
MRS. B.
Exactly the same.—We may now proceed to SODA, which, however important, will detain us but a
very short time; as in all its general properties it very strongly
resembles potash; indeed, so great is their similitude, that they have
been long confounded, and they can now scarcely be distinguished, except
by the difference of the salts which they form with acids.
The great source of this alkali is the sea, where,
34
combined with a peculiar acid, it forms the salt with which the waters
of the ocean are so strongly impregnated.
EMILY.
Is not that the common table salt?
MRS. B.
The very same; but again we must postpone entering into the
particulars of this interesting combination, till we treat of the
neutral salts. Soda may be obtained from common salt; but the easiest
and most usual method of procuring it is by the combustion of marine
plants, an operation perfectly analogous to that by which potash is
obtained from vegetables.
EMILY.
From what does soda derive its name?
MRS. B.
From a plant called by us soda, and by the Arabs kali,
which affords it in great abundance. Kali has, indeed, given its name to
the alkalies in general.
CAROLINE.
Does soda form glass and soap in the same manner as potash?
MRS. B.
Yes, it does; it is of equal importance in the
35
arts, and is even preferred to potash for some purposes; but you will
not be able to distinguish their properties till we examine the compound
salts which they form with acids; we must therefore leave soda for the
present, and proceed to AMMONIA, or
the VOLATILE ALKALI.
EMILY.
I long to hear something of this alkali; is it not of the same nature
as hartshorn?
MRS. B.
Yes, it is, as you will see by-and-bye. This alkali is seldom found
in nature in its pure state; it is most commonly extracted from a
compound salt, called sal ammoniac, which was formerly imported
from Ammonia, a region of Libya, from which both these salts
and the alkali derive their names. The crystals contained in this bottle
are specimens of this salt, which consists of a combination of ammonia
and muriatic acid.
CAROLINE.
Then it should be called muriat of ammonia; for though I am
ignorant what muriatic acid is, yet I know that its combination with
ammonia cannot but be so called; and I am surprised to see sal ammoniac
inscribed on the label.
36
MRS. B.
That is the name by which it has been so long known, that the modern
chemists have not yet succeeded in banishing it altogether; and it is
still sold under that name by druggists, though by scientific chemists
it is more properly called muriat of ammonia.
CAROLINE.
Both the popular and the common name should be inscribed on
labels—this would soon introduce the new nomenclature.
EMILY.
By what means can the ammonia be separated from the muriatic
acid?
MRS. B.
By chemical attractions; but this operation is too complicated for
you to understand, till you are better acquainted with the agency of
affinities.
EMILY.
And when extracted from the salt, what kind of substance is
ammonia?
MRS. B.
Its natural form, at the temperature of the atmosphere, when free
from combination, is that of gas; and in this state it is called
ammoniacal gas.
37
But it mixes very readily with water, and can be thus obtained in a
liquid form.
CAROLINE.
You said that ammonia was more complicated in its composition than
the other alkalies; pray of what principles does it consist?
MRS. B.
It was discovered a few years since, by Berthollet, a celebrated
French chemist, that it consisted of about one part of hydrogen to four
parts of nitrogen. Having heated ammoniacal gas under a receiver, by
causing the electrical spark to pass repeatedly through it, he found
that it increased considerably in bulk, lost all its alkaline
properties, and was actually converted into hydrogen and nitrogen gases;
and from the latest and most accurate experiments, the proportions
appear to be, one volume of nitrogen gas to three of hydrogen gas.
CAROLINE.
Ammonia, therefore, has not, like the two other alkalies,
a metallic basis?
MRS. B.
It is believed it has, though it is extremely difficult to reconcile
that idea with what I have just stated of its chemical nature. But the
fact is, that
38
although this supposed metallic basis of ammonia has never been obtained
distinct and separate, yet both Professor Berzelius, of Stockholm, and
Sir H. Davy, have succeeded in forming a combination of mercury with the
basis of ammonia, which has so much the appearance of an amalgam, that
it strongly corroborates the idea of ammonia having a metallic basis.* But
these theoretical points are full of difficulties and doubts, and it
would be useless to dwell any longer upon them.
Let us therefore return to the properties of volatile alkali.
Ammoniacal gas is considerably lighter than oxygen gas, and only about
half the weight of atmospherical air. It possesses most of the
properties of the fixed alkalies; but cannot be of so much use in the
arts on account of its volatile nature. It is, therefore, never employed
in the manufacture of glass, but it forms soap with oils equally as well
as potash and soda; it resembles them likewise in its strong attraction
for water; for which reason it can be collected in a receiver over
mercury only.
39
CAROLINE.
I do not understand this?
MRS. B.
Do you recollect the method which we used to collect gases in a
glass-receiver over water?
CAROLINE.
Perfectly.
MRS. B.
Ammoniacal gas has so strong a tendency to unite with water, that,
instead of passing through that fluid, it would be instantaneously
absorbed by it. We can therefore neither use water for that purpose, nor
any other liquid of which water is a component part; so that, in order
to collect this gas, we are obliged to have recourse to mercury,
(a liquid which has no action upon it,) and a mercurial bath is
used instead of a water bath, such as we employed on former occasions.
Water impregnated with this gas is nothing more than the fluid which you
mentioned at the beginning of the conversation—hartshorn; it is
the ammoniacal gas escaping from the water which gives it so powerful a
smell.
EMILY.
But there is no appearance of effervescence in hartshorn.
40
MRS. B.
Because the particles of gas that rise from the water are too subtle
and minute for their effect to be visible.
Water diminishes in density, by being impregnated with ammoniacal
gas; and this augmentation of bulk increases its capacity for
caloric.
EMILY.
In making hartshorn, then, or impregnating water with ammonia, heat
must be absorbed, and cold produced?
MRS. B.
That effect would take place if it was not counteracted by another
circumstance; the gas is liquefied by incorporating with the water, and
gives out its latent heat. The condensation of the gas more than
counterbalances the expansion of the water; therefore, upon the whole,
heat is produced.—But if you dissolve ammoniacal gas with ice or
snow, cold is produced.—Can you account for that?
EMILY.
The gas, in being condensed into a liquid, must give out heat; and,
on the other hand, the snow or ice, in being rarefied into a liquid,
must absorb heat; so that, between the opposite effects, I should
have supposed the original temperature would have been preserved.
41
MRS. B.
But you have forgotten to take into the account the rarefaction of
the water (or melted ice) by the impregnation of the gas; and this is
the cause of the cold which is ultimately produced.
CAROLINE.
Is the sal volatile (the smell of which so strongly resembles
hartshorn) likewise a preparation of ammonia?
MRS. B.
It is carbonat of ammonia dissolved in water; and which, in its
concrete state, is commonly called salts of hartshorn. Ammonia is
caustic, like the fixed alkalies, as you may judge by the pungent
effects of hartshorn, which cannot be taken internally, nor applied to
delicate external parts, without being plentifully diluted with
water.—Oil and acids are very excellent antidotes for alkaline
poisons; can you guess why?
CAROLINE.
Perhaps, because the oil combines with the alkali, and forms soap,
and thus destroys its caustic properties; and the acid converts it into
a compound salt, which, I suppose, is not so pernicious as caustic
alkali.
MRS. B.
Precisely so.
42
Ammoniacal gas, if it be mixed with atmospherical air, and a burning
taper repeatedly plunged into it, will burn with a large flame of a
peculiar yellow colour.
EMILY.
But pray tell me, can ammonia be procured from this Lybian salt
only?
MRS. B.
So far from it, that it is contained in, and may be extracted from,
all animal substances whatever. Hydrogen and nitrogen are two of the
chief constituents of animal matter; it is therefore not surprising that
they should occasionally meet and combine in those proportions that
compose ammonia. But this alkali is more frequently generated by the
spontaneous decomposition of animal substances; the hydrogen and
nitrogen gases that arise from putrefied bodies combine, and form the
volatile alkali.
Muriat of ammonia, instead of being exclusively brought from Lybia,
as it originally was, is now chiefly prepared in Europe, by chemical
processes. Ammonia, although principally extracted from this salt, can
also be produced by a great variety of other substances. The horns of
cattle, especially those of deer, yield it in abundance, and it is from
this circumstance that a solution of ammonia in water has been called
hartshorn. It may likewise
43
be procured from wool, flesh, and bones; in a word, any animal substance
whatever yields it by decomposition.
We shall now lay aside the alkalies, however important the subject
may be, till we treat of their combination with acids. The next time we
meet we shall examine the earths.
44

MRS. B.
The EARTHS, which we are to-day to examine, are nine in
number:
SILEX,
ALUMINE,
BARYTES,
LIME,
MAGNESIA,
STRONTITES,
YTTRIA,
GLUCINA,
ZIRCONIA.
The last three are of late discovery; their properties are but
imperfectly known; and, as they have not yet been applied to use, it
will be unnecessary to enter into any particulars respecting them; we
shall confine our remarks, therefore, to the first five. They are
composed, as you have already learnt, of a metallic basis combined with
oxygen; and, from this circumstance, are incombustible.
45
CAROLINE.
Yet I have seen turf burnt in the country, and it makes an excellent
fire; the earth becomes red hot, and produces a very great quantity of
heat.
MRS. B.
It is not the earth that burns, my dear, but the roots, grass, and
other remnants of vegetables that are intermixed with it. The caloric,
which is produced by the combustion of these substances, makes the earth
red hot, and this being a bad conductor of heat, retains its caloric a
long time; but were you to examine it when cooled, you would find that
it had not absorbed one particle of oxygen, nor suffered any alteration
from the fire. Earth is, however, from the circumstance just mentioned,
an excellent radiator of heat, and owes its utility, when mixed with
fuel, solely to that property. It is in this point of view that Count
Rumford has recommended balls of incombustible substances to be arranged
in fire-places, and mixed with the coals, by which means the caloric
disengaged by the combustion of the latter is more perfectly reflected
into the room, and an expense of fuel is saved.
EMILY.
I expected that the list of earths would be much more considerable.
When I think of the great
46
variety of soils, I am astonished that there is not a greater
number of earths to form them.
MRS. B.
You might, indeed, almost confine that number to four; for barytes,
strontites, and the others of late discovery, act but so small a part in
this great theatre, that they cannot be reckoned as essential to the
general formation of the globe. And you must not confine your idea of
earths to the formation of soil; for rock, marble, chalk, slate, sand,
flint, and all kinds of stones, from the precious jewels to the
commonest pebbles; in a word, all the immense variety of mineral
products, may be referred to some of these earths, either in a simple
state, or combined the one with the other, or blended with other
ingredients.
CAROLINE.
Precious stones composed of earth! That seems very difficult to
conceive.
EMILY.
Is it more extraordinary than that the most precious of all jewels,
diamond, should be composed of carbon? But diamond forms an exception,
Mrs. B.; for, though a stone, it is not composed of earth.
47
MRS. B.
I did not specify the exception, as I knew you were so well
acquainted with it. Besides, I would call a diamond a mineral
rather than a stone, as the latter term always implies the presence of
some earth.
CAROLINE.
I cannot conceive how such coarse materials can be converted into
such beautiful productions.
MRS. B.
We are very far from understanding all the secret resources of
nature; but I do not think the spontaneous formation of the crystals,
which we call precious stones, one of the most difficult phenomena to
comprehend.
By the slow and regular work of ages, perhaps of hundreds of ages,
these earths may be gradually dissolved by water, and as gradually
deposited by their solvent in the undisturbed process of
crystallisation. The regular arrangement of their particles, during
their reunion in a solid mass, gives them that brilliancy, transparency,
and beauty, for which they are so much admired; and renders them in
appearance so totally different from their rude and primitive
ingredients.
48
CAROLINE.
But how does it happen that they are spontaneously dissolved, and
afterwards crystallised?
MRS. B.
The scarcity of many kinds of crystals, as rubies, emeralds, topazes,
&c. shows that their formation is not an operation very easily
carried on in nature. But cannot you imagine that when water, holding in
solution some particles of earth, filters through the crevices of hills
or mountains, and at length dribbles into some cavern, each successive
drop may be slowly evaporated, leaving behind it the particle of earth
which it held in solution? You know that crystallisation is more regular
and perfect, in proportion as the evaporation of the solvent is slow and
uniform; nature, therefore, who knows no limit of time, has, in all
works of this kind, an infinite advantage over any artist who attempts
to imitate such productions.
EMILY.
I can now conceive that the arrangement of the particles of earth,
during crystallisation, may be such as to occasion transparency, by
admitting a free passage to the rays of light; but I cannot understand
why crystallised earths should assume such beautiful colours as most of
them do. Sapphire,
49
for instance, is of a celestial blue; ruby, a deep red; topaz,
a brilliant yellow?
MRS. B.
Nothing is more simple than to suppose that the arrangement of their
particles is such, as to transmit some of the coloured rays of light,
and to reflect others, in which case the stone must appear of the colour
of the rays which it reflects. But besides, it frequently happens that
the colour of a stone is owing to a mixture of some metallic matter.
CAROLINE.
Pray, are the different kinds of precious stones each composed of one
individual earth, or are they formed of a combination of several
earths?
MRS. B.
A great variety of materials enters into the composition of most of
them; not only several earths, but sometimes salts and metals. The
earths, however, in their simple state, frequently form very beautiful
crystals; and, indeed, it is in that state only that they can be
obtained perfectly pure.
EMILY.
Is not the Derbyshire spar produced by the crystallisation of earths,
in the way you have just
50
explained? I have been in some of the subterraneous caverns where
it is found, which are similar to those you have described.
MRS. B.
Yes; but this spar is a very imperfect specimen of crystallisation;
it consists of a variety of ingredients confusedly blended together, as
you may judge by its opacity, and by the various colours and appearances
which it exhibits.
But, in examining the earths in their most perfect and agreeable
form, we must not lose sight of that state in which they are commonly
found, and which, if less pleasing to the eye, is far more interesting
by its utility.
All the earths are more or less endowed with alkaline properties; but
there are four, barytes, magnesia, lime, and strontites, which are
called alkaline earths, because they possess those qualities in
so great a degree, as to entitle them, in most respects, to the rank of
alkalies. They combine and form compound salts with acids, in the same
way as alkalies; they are, like them, susceptible of a considerable
degree of causticity, and are acted upon in a similar manner by chemical
tests.—The remaining earths, silex and alumine, with one or two
others of late discovery, are in some degree more earthy, that is to
say, they possess more completely the properties common to all the
earths, which
51
are, insipidity, dryness, unalterableness in the fire,
infusibility, &c.
CAROLINE.
Yet, did you not tell us that silex, or siliceous earth, when mixed
with an alkali, was fusible, and run into glass?
MRS. B.
Yes, my dear; but the characteristic properties of earths, which I
have mentioned, are to be considered as belonging to them in a state of
purity only; a state in which they are very seldom to be met with
in nature.—Besides these general properties, each earth has its
own specific characters, by which it is distinguished from any other
substance.—Let us therefore review them separately.
Silex, or SILICA, abounds in flint, sand, sandstone, agate,
jasper, &c.; it forms the basis of many precious stones, and
particularly of those which strike fire with steel. It is rough to the
touch, scratches and wears away metals; it is acted upon by no acid but
the fluoric, and is not soluble in water by any known process; but
nature certainly dissolves it by means with which we are unacquainted,
and thus produces a variety of siliceous crystals, and amongst these
rock crystal,
52
which is the purest specimen of this earth. Silex appears to have been
intended by Providence to form the solid basis of the globe, to serve as
a foundation for the original mountains, and give them that hardness and
durability which has enabled them to resist the various revolutions
which the surface of the earth has successively undergone. From these
mountains siliceous rocks have, during the course of ages, been
gradually detached by torrents of water, and brought down in fragments;
these, in the violence and rapidity of their descent, are sometimes
crumbled to sand, and in this state form the beds of rivers and of the
sea, chiefly composed of siliceous materials. Sometimes the fragments
are broken without being pulverised by their fall, and assume the form
of pebbles, which gradually become rounded and polished.
EMILY.
Pray what is the true colour of silex, which forms such a variety of
different coloured substances? Sand is brown, flint is nearly black, and
precious stones are of all colours.
MRS. B.
Pure silex, such as is found only in the chemist’s laboratory, is
perfectly white, and the various colours which it assumes, in the
different substances
53
you have just mentioned, proceed from the different ingredients with
which it is mixed in them.
CAROLINE.
I wonder that silex is not more valuable, since it forms the basis of
so many precious stones.
MRS. B.
You must not forget that the value we set upon precious stones
depends in a great measure upon the scarcity with which nature affords
them; for, were those productions either common or perfectly imitable by
art, they would no longer, notwithstanding their beauty, be so highly
esteemed. But the real value of siliceous earth, in many of the most
useful arts, is very extensive. Mixed with clay, it forms the basis of
all the various kinds of earthen ware, from the most common utensils to
the most refined ornaments.
EMILY.
And we must recollect its importance in the formation of glass with
potash.
MRS. B.
Nor should we omit to mention, likewise, many other important uses of
silex, such as being the chief ingredient of some of the most durable
cements, of mortar, &c.
54
I said before, that siliceous earth combined with no acid but the
fluoric; it is for this reason that glass is liable to be attacked by
that acid only, which, from its strong affinity for silex, forces that
substance from its combination with the potash, and thus destroys the
glass.
We will now hasten to proceed to the other earths, for I am rather
apprehensive of your growing weary of this part of our subject.
CAROLINE.
The history of the earths is not quite so entertaining as that of the
simple substances.
MRS. B.
Perhaps not; but it is absolutely indispensable that you should know
something of them; for they form the basis of so many interesting and
important compounds, that their total omission would throw great
obscurity on our general outline of chemical science. We shall, however,
review them in as cursory a manner as the subject can admit of.
Alumine derives its name from a
compound salt called alum, of which it forms the basis.
CAROLINE.
But it ought to be just the contrary, Mrs. B.;
55
the simple body should give, instead of taking, its name from the
compound.
MRS. B.
That is true; but as the compound salt was known long before its
basis was discovered, it was very natural that when the earth was at
length separated from the acid, it should derive its name from the
compound from which it was obtained. However, to remove your scruples,
we will call the salt according to the new nomenclature, sulphat of
alumine. From this combination, alumine may be obtained in its pure
state; it is then soft to the touch, makes a paste with water, and
hardens in the fire. In nature, it is found chiefly in clay, which
contains a considerable proportion of this earth; it is very abundant in
fuller’s earth, slate, and a variety of other mineral productions. There
is indeed scarcely any mineral substance more useful to mankind than
alumine. In the state of clay, it forms large strata of the earth, gives
consistency to the soil of valleys, and of all low and damp spots, such
as swamps and marshes. The beds of lakes, ponds, and springs, are almost
entirely of clay; instead of allowing of the filtration of water, as
sand does, it forms an impenetrable bottom, and by this means water is
accumulated in the caverns of the earth, producing
56
those reservoirs whence springs issue, and spout out at the surface.
EMILY.
I always thought that these subterraneous reservoirs of water were
bedded by some hard stone, or rock, which the water could not
penetrate.
MRS. B.
That is not the case; for in the course of time water would
penetrate, or wear away silex, or any other kind of stone, while it is
effectually stopped by clay, or alumine.
The solid compact soils, such as are fit for corn, owe their
consistence in a great measure to alumine; this earth is therefore used
to improve sandy or chalky soils, which do not retain a sufficient
quantity of water for the purpose of vegetation.
Alumine is the most essential ingredient in all potteries. It enters
into the composition of brick, as well as that of the finest porcelain;
the addition of silex and water hardens it, renders it susceptible of a
degree of vitrification, and makes it perfectly fit for its various
purposes.
CAROLINE.
I can scarcely conceive that brick and china should be made of the
same materials.
57
MRS. B.
Brick consists almost entirely of baked clay; but a certain
proportion of silex is essential to the formation of earthen or stone
ware. In common potteries sand is used for that purpose; a more
pure silex is, I believe, necessary for the composition of
porcelain, as well as a finer kind of clay; and these materials are, no
doubt, more carefully prepared, and curiously wrought, in the one case
than in the other. Porcelain owes its beautiful semitransparency to a
commencement of vitrification.
EMILY.
But the commonest earthen-ware, though not transparent, is covered
with a kind of glazing.
MRS. B.
That precaution is equally necessary for use as for beauty, as the
ware would be liable to be spoiled and corroded by a variety of
substances, if not covered with a coating of this kind. In porcelain it
consists of enamel, which is a fine white opake glass, formed of
metallic oxyds, sand, salts, and such other materials as are susceptible
of vitrification. The glazing of common earthen-ware is made chiefly of
oxyd of lead, or sometimes merely of salt, which, when thinly spread
over earthen vessels, will, at a certain heat, run into opake glass.
58
CAROLINE.
And of what nature are the colours which are used for painting
porcelain?
MRS. B.
They are all composed of metallic oxyds, so that these colours,
instead of receiving injury from the application of fire, are
strengthened and developed by its action, which causes them to undergo
different degrees of oxydation.
Alumine and silex are not only often combined by art, but they have
in nature a very strong tendency to unite, and are found combined, in
different proportions, in various gems and other minerals. Indeed, many
of the precious stones, such as ruby, oriental sapphire, amethyst,
&c. consist chiefly of alumine.
We may now proceed to the alkaline earths, I shall say but a few
words on BARYTES, as it is hardly ever
used, except in chemical laboratories. It is remarkable for its great
weight, and its strong alkaline properties, such as destroying animal
substances, turning green some blue vegetable colours, and showing a
powerful attraction for acids; this last property it possesses to such a
degree, particularly with regard to the sulphuric acid, that it will
always detect its presence in any substance or combination whatever, by
immediately uniting with it, and forming a sulphat of barytes. This
59
renders it a very valuable chemical test. It is found pretty abundantly
in nature in the state of carbonat, from which the pure earth can be
easily separated.
The next earth we have to consider is LIME. This is a substance of too great and general
importance to be passed over so slightly as the last.
Lime is strongly alkaline. In nature it is not met with in its simple
state, as its affinity for water and carbonic acid is so great, that it
is always found combined with these substances, with which it forms the
common lime-stone; but it is separated in the kiln from these
ingredients, which are volatilised whenever a sufficient degree of heat
is applied.
EMILY.
Pure lime, then, is nothing but lime-stone, which has been deprived,
in the kiln, of its water and carbonic acid?
MRS. B.
Precisely: in this state it is called quick-lime, and it is so
caustic, that it is capable of decomposing the dead bodies of animals
very rapidly, without their undergoing the process of
putrefaction.—I have here some quick lime, which is kept
carefully corked up in a bottle to prevent the access
60
of air; for were it at all exposed to the atmosphere, it would absorb
both moisture and carbonic acid gas from it, and be soon slaked. Here is
also some lime-stone—we shall pour a little water on each, and
observe the effects that result from it.
CAROLINE.
How the quick-lime hisses! It is become excessively hot!—It
swells, and now it bursts and crumbles to powder, while the water
appears to produce no kind of alteration on the lime-stone.
MRS. B.
Because the lime-stone is already saturated with water, whilst the
quick-lime, which has been deprived of it in the kiln, combines with it
with very great avidity, and produces this prodigious disengagement of
heat, the cause of which I formerly explained to you; do you
recollect it?
EMILY.
Yes; you said that the heat did not proceed from the lime, but from
the water which was solidified, and thus parted with its heat of
liquidity.
MRS. B.
Very well. If we continue to add successive quantities of water to
the lime after being slaked
61
and crumbled as you see, it will then gradually be diffused in the
water, till it will at length be dissolved in it, and entirely
disappear; but for this purpose it requires no less than 700 times its
weight of water. This solution is called lime-water.
CAROLINE.
How very small, then, is the proportion of lime dissolved!
MRS. B.
Barytes is still of more difficult solution; it dissolves only in 900
times its weight of water: but it is much more soluble in the state of
crystals. The liquid contained in this bottle is lime-water; it is often
used as a medicine, chiefly, I believe, for the purpose of
combining with, and neutralising, the superabundant acid which it meets
with in the stomach.
EMILY.
I am surprised that it is so perfectly clear; it does not at all
partake of the whiteness of the lime.
MRS. B.
Have you forgotten that, in solutions, the solid body is so minutely
subdivided by the fluid as to become invisible, and therefore will not
in the least degree impair the transparency of the solvent?
62
I said that the attraction of lime for carbonic acid was so strong,
that it would absorb it from the atmosphere. We may see this effect by
exposing a glass of lime-water to the air; the lime will then separate
from the water, combine with the carbonic acid, and re-appear on the
surface in the form of a white film, which is carbonat of lime, commonly
called chalk.
CAROLINE.
Chalk is, then, a compound salt! I never should have supposed
that those immense beds of chalk, that we see in many parts of the
country, were a salt.—Now, the white film begins to appear on the
surface of the water; but it is far from resembling hard solid
chalk.
MRS. B.
That is owing to its state of extreme division; in a little time it
will collect into a more compact mass, and subside at the bottom of the
glass.
If you breathe into lime-water, the carbonic acid, which is mixed
with the air that you expire, will produce the same effect. It is an
experiment very easily made;—I shall pour some lime-water
into this glass tube, and, by breathing repeatedly into it, you will
soon perceive a precipitation of chalk—
63
EMILY.
I see already a small white cloud formed.
MRS. B.
It is composed of minute particles of chalk; at present it floats in
the water, but it will soon subside.
Carbonat of lime, or chalk, you see, is insoluble in water, since the
lime which was dissolved re-appears when converted into chalk; but you
must take notice of a very singular circumstance, which is, that chalk
is soluble in water impregnated with carbonic acid.
CAROLINE.
It is very curious, indeed, that carbonic acid gas should render lime
soluble in one instance, and insoluble in the other!
MRS. B.
I have here a bottle of Seltzer water, which, you know, is strongly
impregnated with carbonic acid:—let us pour a little of it into a
glass of lime-water. You see that it immediately forms a precipitation
of carbonat of lime?
EMILY.
Yes, a white cloud appears.
64
MRS. B.
I shall now pour an additional quantity of the Seltzer water into the
lime-water—
EMILY.
How singular! The cloud is re-dissolved, and the liquid is again
transparent.
MRS. B.
All the mystery depends upon this circumstance, that carbonat of lime
is soluble in carbonic acid, whilst it is insoluble in water; the first
quantity of carbonic acid, therefore, which I introduce into the
lime-water, was employed in forming the carbonat of lime, which remained
visible, until an additional quantity of carbonic acid dissolved it.
Thus, you see, when the lime and carbonic acid are in proper proportions
to form chalk, the white cloud appears, but when the acid predominates,
the chalk is no sooner formed than it is dissolved.
CAROLINE.
That is now the case; but let us try whether a further addition of
lime-water will again precipitate the chalk.
EMILY.
It does, indeed! The cloud re-appears, because,
65
I suppose, there is now no more of the carbonic acid than is
necessary to form chalk; and, in order to dissolve the chalk,
a superabundance of acid is required.
MRS. B.
We have, I think, carried this experiment far enough; every
repetition would but exhibit the same appearances.
Lime combines with most of the acids, to which the carbonic (as being
the weakest) readily yields it; but these combinations we shall have an
opportunity of noticing more particularly hereafter. It unites with
phosphorus, and with sulphur, in their simple state; in short, of all
the earths, lime is that which nature employs most frequently, and most
abundantly, in its innumerable combinations. It is the basis of all
calcareous earths and stones; we find it likewise in the animal and the
vegetable creations.
EMILY.
And in the arts is not lime of very great utility?
MRS. B.
Scarcely any substance more so; you know that it is a most essential
requisite in building, as it constitutes the basis of all cements, such
as mortar, stucco, plaister, &c.
66
Lime is also of infinite importance in agriculture; it lightens and
warms soils that are too cold, and compact, in consequence of too great
a proportion of clay.—But it would be endless to enumerate the
various purposes for which it is employed; and you know enough of it to
form some idea of its importance; we shall, therefore, now proceed to
the third alkaline earth, MAGNESIA.
CAROLINE.
I am already pretty well acquainted with that earth; it is a
medicine.
MRS. B.
It is in the state of carbonat that magnesia is usually employed
medicinally; it then differs but little in appearance from its simple
form, which is that of a very fine light white powder. It dissolves in
2000 times its weight of water, but forms with acids extremely soluble
salts. It has not so great an attraction for acids as lime, and
consequently yields them to the latter. It is found in a great variety
of mineral combinations, such as slate, mica, amianthus, and more
particularly in a certain lime stone, which has lately been discovered
by Mr. Tennant to contain it in very great quantities. It does not
attract and solidify water, like lime: but when mixed with water and
exposed to the atmosphere, it slowly absorbs
67
carbonic acid from the latter, and thus loses its causticity. Its chief
use in medicine is, like that of lime, derived from its readiness to
combine with, and neutralise, the acid which it meets with in the
stomach.
EMILY.
Yet, you said that it was taken in the state of carbonat, in which
case it has already combined with an acid?
MRS. B.
Yes; but the carbonic is the last of all the acids in the order of
affinities; it will therefore yield the magnesia to any of the others.
It is, however, frequently taken in its caustic state as a remedy for
flatulence. Combined with sulphuric acid, magnesia forms another and
more powerful medicine, commonly called Epsom salt.
CAROLINE.
And properly, sulphat of magnesia, I suppose? Pray why
was it ever called Epsom salt?
MRS. B.
Because there is a spring in the neighbourhood of Epsom which
contains this salt in great abundance.
The last alkaline earth which we have to mention is STRONTIAN, or STRONTITES, discovered by
68
Dr. Hope a few years ago. It so strongly resembles barytes in its
properties, and is so sparingly found in nature, and of so little use in
the arts, that it will not be necessary to enter into any particulars
respecting it. One of the remarkable characteristic properties of
strontites is, that its salts, when dissolved in spirit of wine, tinge
the flame of a deep red, or blood colour.
69

MRS. B.
We may now proceed to the acids. Of the
metallic oxyds, you have already acquired some general notions. This
subject, though highly interesting in its details, is not of sufficient
importance to our concise view of chemistry, to be particularly treated
of; but it is absolutely necessary that you should be better acquainted
with the acids, and likewise with their combinations with the alkalies,
which form the triple compounds called NEUTRAL SALTS.
The class of acids is characterised by very distinct properties. They
all change blue vegetable infusions to a red colour: they are all more
or less sour to the taste; and have a general tendency to combine with
the earths, alkalies, and metallic oxyds.
You have, I believe, a clear idea of the nomenclature by which
the base (or radical) of the acid, and the various degrees of
acidification, are expressed?
EMILY.
Yes, I think so; the acid is distinguished by the
70
name of its base, and its degree of oxydation, that is, the quantity of
oxygen it contains, by the termination of that name in ous or
ic; thus sulphureous acid is that formed by the smallest
proportion of oxygen combined with sulphur; sulphuric acid that
which results from the combination of sulphur with the greatest quantity
of oxygen.
MRS. B.
A still greater latitude may, in many cases, be allowed to the
proportions of oxygen than can be combined with acidifiable radicals; for
several of these radicals are susceptible of uniting with a quantity of
oxygen so small as to be insufficient to give them the properties of
acids; in these cases, therefore, they are converted into oxyds. Such is
sulphur, which by exposure to the atmosphere with a degree of heat
inadequate to produce inflammation, absorbs a small proportion of
oxygen, which colours it red or brown. This, therefore, is the first
degree of oxygenation of sulphur; the 2d converts it into
sulphurous acid; the 3d into the sulphuric acid; and
4thly, if it was found capable of combining with a still larger
proportion of oxygen, it would then be termed super-oxygenated
sulphuric acid.
EMILY.
Are these various degrees of oxygenation common to all the acids?
71
MRS. B.
No; they vary much in this respect: some are susceptible of only one
degree of oxygenation; others, of two, or three; there are but very few
that will admit of more.
CAROLINE.
The modern nomenclature must be of immense advantage in pointing out
so easily the nature of the acids, and their various degrees of
oxygenation.
MRS. B.
Till lately many of the acids had not been decomposed; but analogy
afforded so strong a proof of their compound nature, that I never could
reconcile myself to classing them with the simple bodies, though this
division has been adopted by several chemical writers. At present there
are only the muriatic and the fluoric acids, which have not had their
bases distinctly separated.
CAROLINE.
We have heard of a great variety of acids; pray how many are there in
all?
MRS. B.
I believe there are reckoned at present thirty-four, and their number
is constantly increasing, as the science improves; but the most
important,
72
and those to which we shall almost entirely confine our attention, are
but few. I shall, however, give you a general view of the whole;
and then we shall more particularly examine those that are the most
essential.
This class of bodies was formerly divided into mineral, vegetable,
and animal acids, according to the substances from which they were
commonly obtained.
CAROLINE.
That, I should think, must have been an excellent arrangement; why
was it altered?
MRS. B.
Because in many cases it produced confusion. In which class, for
instance, would you place carbonic acid?
CAROLINE.
Now I see the difficulty. I should be at a loss where to place it, as
you have told us that it exists in the animal, vegetable, and mineral
kingdoms.
EMILY.
There would be the same objection with respect to phosphoric acid,
which, though obtained chiefly from bones, can also, you said, be found
in small quantities in stones, and likewise in some plants.
MRS. B.
You see, therefore, the propriety of changing
73
this mode of classification. These objections do not exist in the
present nomenclature; for the composition and nature of each individual
acid is in some degree pointed out, instead of the class of bodies from
which it is extracted; and, with regard to the more general division of
acids, they are classed under these three heads:
First, Acids of known or supposed simple bases, which are formed by
the union of these bases with oxygen. They are the following:
| The | Sulphuric |
|
Acids, of known and simple bases. |
| Carbonic |
| Nitric |
| Phosphoric |
| Arsenical |
| Tungstenic |
| Molybdenic |
| Boracic |
| Fluoric |
| Muriatic |
This class comprehends the most anciently known and most important
acids. The sulphuric, nitric, and muriatic were formerly, and are still
frequently, called mineral acids.
2dly, Acids that have double or binary radicals, and which
consequently consist of triple combinations. These are the vegetable
acids, whose common radical is a compound of hydrogen and carbon.
74
CAROLINE.
But if the basis of all the vegetable acids be the same, it should
form but one acid; it may indeed combine with different proportions of
oxygen, but the nature of the acid must be the same.
MRS. B.
The only difference that exists in the basis of vegetable acids, is
the various proportions of hydrogen and carbon from which they are
severally composed. But this is enough to produce a number of acids
apparently very dissimilar. That they do not, however, differ
essentially, is proved by their susceptibility of being converted into
each other, by the addition or subtraction of a portion of hydrogen or
of carbon. The names of these acids are,
| The | Acetic |
|
Acids, of double bases, being of vegetable origin. |
| Oxalic |
| Tartarous |
| Citric |
| Malic |
| Gallic |
| Mucous |
| Benzoic |
| Succinic |
| Camphoric |
| Suberic |
75
The 3d class of acids consists of those which have triple radicals,
and are therefore of a still more compound nature. This class
comprehends the animal acids, which are,
| The | Lactic |
|
Acids, of triple bases, or animal acids. |
| Prussic |
| Formic |
| Bombic |
| Sebacic |
| Zoonic |
| Lithic |
I have given you this summary account or enumeration of the acids, as
you may find it more satisfactory to have at once an outline or a
general notion of the extent of the subject; but we shall now confine
ourselves to the first class, which requires our more immediate
attention; and defer the few remarks which we shall have to make on the
others, till we treat of the chemistry of the animal and vegetable
kingdoms.
The acids of simple and known radicals are all capable of being
decomposed by combustible bodies, to which they yield their oxygen. If,
for instance, I pour a drop of sulphuric acid on this piece of
iron, it will produce a spot of rust, you know what that is?
76
CAROLINE.
Yes; it is an oxyd, formed by the oxygen of the acid combining with
the iron.
MRS. B.
In this case you see the sulphur deposits the oxygen by which it was
acidified on the metal. And again, if we pour some acid on a compound
combustible substance, (we shall try it on this piece of wood,) it will
combine with one or more of the constituents of that substance, and
occasion a decomposition.
EMILY.
It has changed the colour of the wood to black. How is that?
MRS. B.
The oxygen deposited by the acid has burnt it; you know that wood in
burning becomes black before it is reduced to ashes. Whether it derives
the oxygen which burns it from the atmosphere, or from any other source,
the chemical effect on the wood is the same. In the case of real
combustion, wood becomes black, because it is reduced to the state of
charcoal by the evaporation of its other constituents. But can you tell
me the reason why wood turns black when burnt by the application of an
acid?
77
CAROLINE.
First, tell me what are the ingredients of wood?
MRS. B.
Hydrogen and carbon are the chief constituents of wood, as of all
other vegetable substances.
CAROLINE.
Well, then, I suppose that the oxygen of the acid combines with the
hydrogen of the wood, to form water; and that the carbon of the wood,
remaining alone, appears of its usual black colour.
MRS. B.
Very well indeed, my dear; that is certainly the most plausible
explanation.
EMILY.
Would not this be a good method of making charcoal?
MRS. B.
It would be an extremely expensive, and, I believe, very
imperfect method; for the action of the acid on the wood, and the heat
produced by it, are far from sufficient to deprive the wood of all its
evaporable parts.
CAROLINE.
What is the reason that vinegar, lemon, and the acid of fruits, do
not produce this effect on wood?
78
MRS. B.
They are vegetable acids, whose bases are composed of hydrogen and
carbon; the oxygen, therefore, will not be disposed to quit this
radical, where it is already united with hydrogen. The strongest of
these may, perhaps, yield a little of their oxygen to the wood, and
produce a stain upon it; but the carbon will not be sufficiently
uncovered to assume its black colour. Indeed, the several mineral acids
themselves possess this power of charring wood in very different degrees.
EMILY.
Cannot vegetable acids be decomposed, by any combustibles?
MRS. B.
No; because their radical is composed of two substances which have a
greater attraction for oxygen than any known body.
CAROLINE.
And are those strong acids, which burn and decompose wood, capable of
producing similar effects on the skin and flesh of animals?
MRS. B.
Yes; all the mineral acids, and one of them more especially, possess
powerful caustic qualities. They actually corrode and destroy the
79
skin and flesh; but they do not produce upon these exactly the same
alteration they do on wood, probably because there is a great proportion
of nitrogen and other substances in animal matter, which prevents the
separation of carbon from being so conspicuous.
80
OF THE SULPHURIC AND PHOSPHORIC ACIDS; OR THE COMBINATIONS OF OXYGEN
WITH SULPHUR AND PHOSPHORUS; AND OF THE SULPHATS AND PHOSPHATS.

MRS. B.
In addition to the general survey which we
have taken of acids, I think you will find it interesting to
examine individually a few of the most important of them, and likewise
some of their principal combinations with the alkalies, alkaline earths,
and metals. The first of the acids, in point of importance, is the SULPHURIC, formerly called oil of
vitriol.
CAROLINE.
I have known it a long time by that name, but had no idea that it was
the same fluid as sulphuric acid. What resemblance or connection can
there be between oil of vitriol and this acid?
MRS. B.
Vitriol is the common name for sulphat of iron, a salt which is
formed by the combination of sulphuric acid and iron; the sulphuric acid
was formerly
81
obtained by distillation from this salt, and it very naturally received
its name from the substance which afforded it.
CAROLINE.
But it is still usually called oil of vitriol?
MRS. B.
Yes; a sufficient length of time has not yet elapsed, since the
invention of the new nomenclature, for it to be generally disseminated;
but, as it is adopted by all scientific chemists, there is every reason
to suppose that it will gradually become universal. When I received this
bottle from the chemists, oil of vitriol was inscribed on the
label; but, as I knew you were very punctilious in regard to the
nomenclature, I changed it, and substituted the words sulphuric
acid.
EMILY.
This acid has neither colour nor smell, but it appears much thicker
than water.
MRS. B.
It is nearly twice as heavy as water, and has, you see, an oily
consistence.
CAROLINE.
And it is probably from this circumstance that
82
it has been called an oil, for it can have no real claim to that name,
as it does not contain either hydrogen or carbon, which are the
essential constituents of oil.
MRS. B.
Certainly; and therefore it would be the more absurd to retain a name
which owed its origin to such a mistaken analogy.
Sulphuric acid, in its purest state, would probably be a concrete
substance, but its attraction for water is such, that it is impossible
to obtain that acid perfectly free from it; it is, therefore, always
seen in a liquid form, such as you here find it. One of the most
striking properties of sulphuric acid is that of evolving a considerable
quantity of heat when mixed with water; this I have already shown
you.
EMILY.
Yes, I recollect it; but what was the degree of heat produced by that
mixture?
MRS. B.
The thermometer may be raised by it to 300 degrees, which is
considerably above the temperature of boiling water.
CAROLINE.
Then water might be made to boil in that mixture?
83
MRS. B.
Nothing more easy, provided that you employ sufficient quantities of
acid and of water, and in the due proportions. The greatest heat is
produced by a mixture of one part of water to four of the acid: we shall
make a mixture of these proportions, and immerse in it this thin glass
tube, which is full of water.
CAROLINE.
The vessel feels extremely hot, but the water does not boil yet.
MRS. B.
You must allow some time for the heat to penetrate the tube, and
raise the temperature of the water to the boiling point—
CAROLINE.
Now it boils—and with increasing violence.
MRS. B.
But it will not continue boiling long; for the mixture gives out heat
only while the particles of the water and the acid are mutually
penetrating each other: as soon as the new arrangement of those
particles is effected, the mixture will gradually cool, and the water
return to its former temperature.
You have seen the manner in which sulphuric
84
acid decomposes all combustible substances, whether animal, vegetable,
or mineral, and burns them by means of its oxygen?
CAROLINE.
I have very unintentionally repeated the experiment on my gown, by
letting a drop of the acid fall upon it, and it has made a stain, which,
I suppose, will never wash out.
MRS. B.
No, certainly; for before you can put it into water, the spot will
become a hole, as the acid has literally burnt the muslin.
CAROLINE.
So it has, indeed! Well, I will fasten the stopper, and put the
bottle away, for it is a dangerous substance.—Oh, now I have done
worse still, for I have spilt some on my hand!
MRS. B.
It is then burned, as well as your gown, for you know that oxygen
destroys animal as well as vegetable matters; and, as far as the
decomposition of the skin of your finger is effected, there is no
remedy; but by washing it immediately in water, you will dilute the
acid, and prevent any further injury.
85
CAROLINE.
It feels extremely hot, I assure you.
MRS. B.
You have now learned, by experience, how cautiously this acid must be
used. You will soon become acquainted with another acid, the nitric,
which, though it produces less heat on the skin, destroys it still
quicker, and makes upon it an indelible stain. You should never handle
any substances of this kind, without previously dipping your fingers in
water, which will weaken their caustic effects. But, since you will not
repeat the experiment, I must put in the stopper, for the acid
attracts the moisture from the atmosphere, which would destroy its
strength and purity.
EMILY.
Pray, how can sulphuric acid be extracted from sulphat of iron by
distillation?
MRS. B.
The process of distillation, you know, consists in separating
substances from one another by means of their different degrees of
volatility, and by the introduction of a new chemical agent, caloric.
Thus, if sulphat of iron be exposed in a retort to a proper degree of
heat, it will be decomposed, and the sulphuric acid will be
volatilised.
86
EMILY.
But now that the process of forming acids by the combustion of their
radicals is known, why should not this method be used for making
sulphuric acid?
MRS. B.
This is actually done in most manufactures; but the usual method of
preparing sulphuric acid does not consist in burning the sulphur in
oxygen gas (as we formerly did by the way of experiment), but in heating
it together with another substance, nitre, which yields oxygen in
sufficient abundance to render the combustion in common air rapid and
complete.
CAROLINE.
This substance, then, answers the same purpose as oxygen gas?
MRS. B.
Exactly. In manufactures the combustion is performed in a leaden
chamber, with water at the bottom, to receive the vapour and assist its
condensation. The combustion is, however, never so perfect but that a
quantity of sulphureous acid is formed at the same time; for you
recollect that the sulphureous acid, according to the chemical
nomenclature, differs from the sulphuric only by containing less
oxygen.
From its own powerful properties, and from the various combinations
into which it enters, sulphuric
87
acid is of great importance in many of the arts.
It is used also in medicine in a state of great dilution; for were it
taken internally, in a concentrated state, it would prove a most
dangerous poison.
CAROLINE.
I am sure it would burn the throat and stomach.
MRS. B.
Can you think of any thing that would prove an antidote to this
poison?
CAROLINE.
A large draught of water to dilute it.
MRS. B.
That would certainly weaken the caustic power of the acid, but it
would increase the heat to an intolerable degree. Do you recollect
nothing that would destroy its deleterious properties more
effectually?
EMILY.
An alkali might, by combining with it; but, then, a pure alkali
is itself a poison, on account of its causticity.
MRS. B.
There is no necessity that the alkali should be
88
caustic. Soap, in which it is combined with oil; or magnesia, either in
the state of carbonat, or mixed with water, would prove the best
antidotes.
EMILY.
In those cases then, I suppose, the potash and the magnesia would
quit their combinations to form salts with the sulphuric acid?
MRS. B.
Precisely.
We may now make a few observations on the sulphureous acid,
which we have found to be the product of sulphur slowly and imperfectly
burnt. This acid is distinguished by its pungent smell, and its gaseous
form.
CAROLINE.
Its aëriform state is, I suppose, owing to the smaller proportion of
oxygen, which renders it lighter than sulphuric acid?
MRS. B.
Probably; for by adding oxygen to the weaker acid, it may be
converted into the stronger kind. But this change of state may also be
connected with a change of affinity with regard to caloric.
EMILY.
And may sulphureous acid be obtained from sulphuric acid by a
diminution of oxygen?
89
MRS. B.
Yes; it can be done by bringing any combustible substance in contact
with the acid. This decomposition is most easily performed by some of
the metals; these absorb a portion of the oxygen from the sulphuric
acid, which is thus converted into the sulphureous, and flies off in its
gaseous form.
CAROLINE.
And cannot the sulphureous acid itself be decomposed and reduced to
sulphur?
MRS. B.
Yes; if this gas be heated in contact with charcoal, the oxygen of
the gas will combine with it, and the pure sulphur is regenerated.
Sulphureous acid is readily absorbed by water; and in this liquid
state it is found particularly useful in bleaching linen and woollen
cloths, and is much used in manufactures for those purposes. I can
show you its effect in destroying colours, by taking out vegetable
stains—I think I see a spot on your gown, Emily, on which we may
try the experiment.
EMILY.
It is the stain of mulberries; but I shall be almost afraid of
exposing my gown to the experiment, after seeing the effect which the
sulphuric acid produced on that of Caroline—
90
MRS. B.
There is no such danger from the sulphureous; but the experiment must
be made with great caution, for, during the formation of sulphureous
acid by combustion, there is always some sulphuric produced.
CAROLINE.
But where is your sulphureous acid?
MRS. B.
We may easily prepare some ourselves, simply by burning a match; we
must first wet the stain with water, and now hold it in this way, at a
little distance, over the lighted match: the vapour that arises from it
is sulphureous acid, and the stain, you see, gradually disappears.
EMILY.
I have frequently taken out stains by this means, without
understanding the nature of the process. But why is it necessary to wet
the stain before it is exposed to the acid fumes?
MRS. B.
The moisture attracts and absorbs the sulphureous acid; and it serves
likewise to dilute any particles of sulphuric acid which might injure
the linen.
Sulphur is susceptible of a third combination
91
with oxygen, in which the proportion of the latter is too small to
render the sulphur acid. It acquires this slight oxygenation by mere
exposure to the atmosphere, without any elevation of temperature: in
this case, the sulphur does not change its natural form, but is only
discoloured, being changed to red or brown; and in this state it is an
oxyd of sulphur.
Before we take leave of the sulphuric acid, we shall say a few words
of its principal combinations. It unites with all the alkalies, alkaline
earths and metals, to form compound salts.
CAROLINE.
Pray, give me leave to interrupt you for a moment: you have never
mentioned any other salts than the compound or neutral salts; is there
no other kind?
MRS. B.
The term salt has been used, from time immemorial, as a kind
of general name for any substance that has savour, odour, is soluble in
water, and crystallisable, whether it be of an acid, an alkaline, or
compound nature; but the compound salts alone retain that appellation in
modern chemistry.
The most important of the salts, formed by the combinations of the
sulphuric acid, are, first, sulphat of potash, formerly called
sal polychrest: this
92
is a very bitter salt, much used in medicine; it is found in the ashes
of most vegetables, but it may be prepared artificially by the immediate
combination of sulphuric acid and potash. This salt is easily soluble in
boiling water. Solubility is, indeed, a property common to all
salts; and they always produce cold in melting.
EMILY.
That must be owing to the caloric which they absorb in passing from a
solid to a fluid form.
MRS. B.
That is, certainly, the most probable explanation.
Sulphat of soda, commonly called Glauber’s salt, is another
medicinal salt, which is still more bitter than the preceding. We must
prepare some of these compounds, that you may observe the phenomena
which take place during their formation. We need only pour some
sulphuric acid over the soda which I have put into this glass.
CAROLINE.
What an amazing heat is disengaged!—I thought you said that
cold was produced by the melting of salts?
MRS. B.
But you must observe that we are now making,
93
not melting a salt. Heat is disengaged during the formation of
compound salts, and a faint light is also emitted, which may sometimes
be perceived in the dark.
EMILY.
And is this heat and light produced by the union of the opposite
electricities of the alkali and the acid?
MRS. B.
No doubt it is, if that theory be true.
CAROLINE.
The union of an acid and an alkali is then an actual combustion?
MRS. B.
Not precisely, though there is certainly much analogy in these
processes.
CAROLINE.
Will this sulphat of soda become solid?
MRS. B.
We have not, I suppose, mixed the acid and the alkali in the exact
proportions that are required for the formation of the salt, otherwise
the mixture would have been almost immediately changed to a solid mass;
but, in order to obtain it in crystals, as you see it in this bottle, it
would be necessary first to dilute it with water, and afterwards
94
to evaporate the water, during which operation the salt would gradually
crystallise.
CAROLINE.
But of what use is the addition of water, if it is afterwards to be
evaporated?
MRS. B.
When suspended in water, the acid and the alkali are more at liberty
to act on each other, their union is more complete, and the salt assumes
the regular form of crystals during the slow evaporation of its
solvent.
Sulphat of soda liquefies by heat, and effloresces in the air.
EMILY.
Pray what is the meaning of the word effloresces? I do
not recollect your having mentioned it before.
MRS. B.
A salt is said to effloresce when it loses its water of
crystallisation on being exposed to the atmosphere, and is thus
gradually converted into a dry powder: you may observe that these
crystals of sulphat of soda are far from possessing the transparency
which belongs to their crystalline state; they are covered with a white
powder, occasioned by their having been exposed to the atmosphere, which
has deprived their surface of its lustre, by
95
absorbing its water of crystallisation. Salts are, in general, either
efflorescent or deliquescent: this latter property is
precisely the reverse of the former; that is to say, deliquescent salts
absorb water from the atmosphere, and are moistened and gradually melted
by it. Muriat of lime is an instance of great deliquescence.
EMILY.
But are there no salts that have the same degree of attraction for
water as the atmosphere, and that will consequently not be affected
by it?
MRS. B.
Yes; there are many such salts, as, for instance, common salt,
sulphat of magnesia, and a variety of others.
Sulphat of lime is very frequently met with in nature, and
constitutes the well-known substance called gypsum, or plaster
of Paris.
Sulphat of magnesia, commonly called Epsom salt, is
another very bitter medicine, which is obtained from sea-water and from
several springs, or may be prepared by the direct combination of its
ingredients.
We have formerly mentioned sulphat of alumine as constituting
the common alum; it is found in nature chiefly in the
neighbourhood of volcanos, and is particularly useful in the arts, from
its
96
strong astringent qualities. It is chiefly employed by dyers and
calico-printers, to fix colours; and is used also in the manufacture of
some kinds of leather.
Sulphuric acid combines also with the metals.
CAROLINE.
One of these combinations, sulphat of iron, we are already
well acquainted with.
MRS. B.
That is the most important metallic salt formed by sulphuric acid,
and the only one that we shall here notice. It is of great use in the
arts; and, in medicine, it affords a very valuable tonic: it is of this
salt that most of those preparations called steel medicines are
composed.
CAROLINE.
But does any carbon enter into these compositions to form steel?
MRS. B.
Not an atom: they are, therefore, very improperly called steel: but
it is the vulgar appellation, and medical men themselves often comply
with the general custom.
Sulphat of iron may be prepared, as you have seen, by dissolving iron
in sulphuric acid; but it
97
is generally obtained from the natural production called Pyrites,
which being a sulphuret of iron, requires only exposure to the
atmosphere to be oxydated, in order to form the salt; this, therefore,
is much the most easy way of procuring it on a large scale.
EMILY.
I am surprised to find that both acids and compound salts are
generally obtained from their various combinations, rather than from the
immediate union of their ingredients.
MRS. B.
Were the simple bodies always at hand, their combinations would
naturally be the most convenient method of forming compounds; but you
must consider that, in most instances, there is great difficulty and
expense in obtaining the simple ingredients from their combinations; it
is, therefore, often more expedient to procure compounds from the
decomposition of other compounds. But, to return to the sulphat of
iron.—There is a certain vegetable acid called Gallic acid,
which has the remarkable property of precipitating this salt
black—I shall pour a few drops of the gallic acid into this
solution of sulphat of iron—
CAROLINE.
It is become as black as ink!
98
MRS. B.
And it is ink in reality. Common writing ink is a precipitate of
sulphat of iron by gallic acid; the black colour is owing to the
formation of gallat of iron, which being insoluble, remains suspended in
the fluid.
This acid has also the property of altering the colour of iron in its
metallic state. You may frequently see its effect on the blade of a
knife, that has been used to cut certain kinds of fruits.
CAROLINE.
True; and that is, perhaps, the reason that a silver knife is
preferred to cut fruits; the gallic acid, I suppose, does not act
upon silver.—Is this acid found in all fruits?
MRS. B.
It is contained, more or less, in the rind of most fruits and roots,
especially the radish, which, if scraped with a steel or iron knife, has
its bright red colour changed to a deep purple, the knife being at the
same time blackened. But the vegetable substance in which the gallic
acid most abounds is nutgall, a kind of excrescence that
grows on oaks, and from which the acid is commonly obtained for its
various purposes.

99
MRS. B.
We now come to the PHOSPHORIC and
PHOSPHOROUS ACIDS. In treating of
phosphorus, you have seen how these acids may be obtained from it by
combustion?
EMILY.
Yes; but I should be much surprised if it was the usual method of
obtaining them, since it is so very difficult to procure phosphorus in
its pure state.
MRS. B.
You are right, my dear; the phosphoric acid, for general purposes, is
extracted from bones, in which it is contained in the state of phosphat
of lime; from this salt the phosphoric acid is separated by means of the
sulphuric, which combines with the lime. In its pure state, phosphoric
acid is either liquid or solid, according to its degree of
concentration.
Among the salts formed by this acid, phosphat of lime is the
only one that affords much interest; and this, we have already observed,
constitutes the basis of all bones. It is also found in very small
quantities in some vegetables.
100
OF THE NITRIC AND CARBONIC ACIDS: OR THE COMBINATIONS OF OXYGEN WITH
NITROGEN AND CARBON; AND OF THE NITRATS AND CARBONATS.

MRS. B.
I
am almost afraid of introducing the subject of the NITRIC ACID, as I am sure that I shall be blamed by
Caroline for not having made her acquainted with it before.
CAROLINE.
Why so, Mrs. B.?
MRS. B.
Because you have long known its radical, which is nitrogen or azote;
and in treating of that element, I did not even hint that it was
the basis of an acid.
CAROLINE.
And what could be your reason for not mentioning this acid
sooner?
MRS. B.
I do not know whether you will think the reason
101
sufficiently good to acquit me; but the omission, I assure you, did
not proceed from negligence. You may recollect that nitrogen was one of
the first simple bodies which we examined; you were then ignorant of the
theory of combustion, which I believe was, for the first time, mentioned
in that lesson; and therefore it would have been in vain, at that time,
to have attempted to explain the nature and formation of acids.
CAROLINE.
I wonder, however, that it never occurred to us to enquire whether
nitrogen could be acidified; for, as we knew it was classed among the
combustible bodies, it was natural to suppose that it might produce an
acid.
MRS. B.
That is not a necessary consequence; for it might combine with oxygen
only in the degree requisite to form an oxyd. But you will find that
nitrogen is susceptible of various degrees of oxygenation, some of which
convert it merely into an oxyd, and others give it all the acid
properties.
The acids, resulting from the combination of oxygen and nitrogen, are
called the NITROUS and NITRIC acids. We will begin with the NITRIC, in which nitrogen is in the highest state of
oxygenation. This acid naturally exists in the form of
102
gas; but is so very soluble in water, and has so great an affinity for
it, that one grain of water will absorb and condense ten grains of acid
gas, and form the limpid fluid which you see in this bottle.
CAROLINE.
What a strong offensive smell it has!
MRS. B.
This acid contains a greater abundance of oxygen than any other, but
it retains it with very little force.
EMILY.
Then it must be a powerful caustic, both from the facility with which
it parts with its oxygen, and the quantity which it affords?
MRS. B.
Very well, Emily; both cause and effect are exactly such as you
describe: nitric acid burns and destroys all kinds of organised matter.
It even sets fire to some of the most combustible substances.—We
shall pour a little of it over this piece of dry warm charcoal—you
see it inflames it immediately; it would do the same with oil of
turpentine, phosphorus, and several other very combustible bodies. This
shows you how easily this acid is decomposed by combustible bodies,
103
since these effects must depend upon the absorption of its oxygen.
Nitric acid has been used in the arts from time immemorial, but it is
only within these twenty-five years that its chemical nature has been
ascertained. The celebrated Mr. Cavendish discovered that it consisted
of about 10 parts of nitrogen and 25 of oxygen.* These principles, in their
gaseous state, combine at a high temperature; and this may be effected
by repeatedly passing the electrical spark through a mixture of the two
gases.
EMILY.
The nitrogen and oxygen gases, of which the atmosphere is composed,
do not combine, I suppose, because their temperature is not
sufficiently elevated?
CAROLINE.
But in a thunder-storm, when the lightning repeatedly passes through
them, may it not produce nitric acid? We should be in a strange
situation, if a violent storm should at once convert the atmosphere into
nitric acid.
MRS. B.
There is no danger of it, my dear; the lightning can affect but a
very small portion of the atmosphere,
104
and though it were occasionally to produce a little nitric acid, yet
this never could happen to such an extent as to be perceivable.
EMILY.
But how could the nitric acid be known, and used, before the method
of combining its constituents was discovered?
MRS. B.
Before that period the nitric acid was obtained, and it is indeed
still extracted, for the common purposes of art, from the compound salt
which it forms with potash, commonly called nitre.
CAROLINE.
Why is it so called? Pray, Mrs. B., let these old unmeaning names be
entirely given up, by us at least; and let us call this salt nitrat
of potash.
MRS. B.
With all my heart; but it is necessary that I should, at least,
mention the old names, and more especially those which are yet in common
use; otherwise, when you meet with them, you would not be able to
understand their meaning.
EMILY.
And how is the acid obtained from this salt?
105
MRS. B.
By the intervention of sulphuric acid, which combines with the
potash, and sets the nitric acid at liberty. This I can easily show you,
by mixing some nitrat of potash and sulphuric acid in this retort, and
heating it over a lamp; the nitric acid will come over in the form of
vapour, which we shall collect in a glass bell. This acid, diluted in
water, is commonly called aqua fortis, if Caroline will allow me
to mention that name.
CAROLINE.
I have often heard that aqua fortis will dissolve almost all metals;
it is no doubt because it yields its oxygen so easily.
MRS. B.
Yes; and from this powerful solvent property, it derived the name of
aqua fortis, or strong water. Do you not recollect that we oxydated, and
afterwards dissolved, some copper in this acid?
EMILY.
If I remember right, the nitrat of copper was the first instance you
gave us of a compound salt.
CAROLINE.
Can the nitric acid be completely decomposed and converted into
nitrogen and oxygen?
106
EMILY.
That cannot be the case, Caroline; since the acid can be decomposed
only by the combination of its constituents with other bodies.
MRS. B.
True; but caloric is sufficient for this purpose. By making the acid
pass through a red hot porcelain tube, it is decomposed; the nitrogen
and oxygen regain the caloric which they had lost in combining, and are
thus both restored to their gaseous state.
The nitric acid may also be partly decomposed, and is by this means
converted into NITROUS ACID.
CAROLINE.
This conversion must be easily effected, as the oxygen is so slightly
combined with the nitrogen.
MRS. B.
The partial decomposition of nitric acid is readily effected by most
metals; but it is sufficient to expose the nitric acid to a very strong
light to make it give out oxygen gas, and thus be converted into nitrous
acid. Of this acid there are various degrees, according to the
proportions of oxygen which it contains; the strongest, and that into
which the nitric is first converted, is of a yellow colour, as you see
in this bottle.
107
CAROLINE.
How it fumes when the stopper is taken out!
MRS. B.
The acid exists naturally in a gaseous state, and is here so strongly
concentrated in water, that it is constantly escaping.
Here is another bottle of nitrous acid, which, you see, is of an
orange red; this acid is weaker, the nitrogen being combined with a
smaller quantity of oxygen; and with a still less proportion of oxygen
it is an olive-green colour, as it appears in this third bottle. In
short, the weaker the acid, the deeper is its colour.
Nitrous acid acts still more powerfully on some inflammable
substances than the nitric.
EMILY.
I am surprised at that, as it contains less oxygen.
MRS. B.
But, on the other hand, it parts with its oxygen much more readily:
you may recollect that we once inflamed oil with this acid.
The next combinations of nitrogen and oxygen form only oxyds of
nitrogen, the first of which is commonly called nitrous air; or
more properly nitric oxyd gas. This may be obtained from nitric
acid, by exposing the latter to the action of metals,
108
as in dissolving them it does not yield the whole of its oxygen, but
retains a portion of this principle sufficient to convert it into this
peculiar gas, a specimen of which I have prepared, and preserved
within this inverted glass bell.
EMILY.
It is a perfectly invisible elastic fluid.
MRS. B.
Yes; and it may be kept any length of time in this manner over water,
as it is not, like the nitric and nitrous acids, absorbable by it. It is
rather heavier than atmospherical air, and is incapable of supporting
either combustion or respiration. I am going to incline the glass
gently on one side, so as to let some of the gas escape—
EMILY.
How very curious!—It produces orange fumes like the nitrous
acid! that is the more extraordinary, as the gas within the glass is
perfectly invisible.
MRS. B.
It would give me much pleasure if you could make out the reason of
this curious change without requiring any further explanation.
CAROLINE.
It seems, by the colour and smell, as if it were
109
converted into nitrous acid gas: yet that cannot be, unless it combines
with more oxygen; and how can it obtain oxygen the very instant it
escapes from the glass?
EMILY.
From the atmosphere, no doubt. Is it not so, Mrs. B.?
MRS. B.
You have guessed it; as soon as it comes in contact with the
atmosphere, it absorbs from it the additional quantity of oxygen
necessary to convert it into nitrous acid gas. And, if I now remove the
bottle entirely from the water, so as to bring at once the whole of the
gas into contact with the atmosphere, this conversion will appear still
more striking—
EMILY.
Look, Caroline, the whole capacity of the bottle is instantly tinged
of an orange colour!
MRS. B.
Thus, you see, it is the most easy process imaginable to convert
nitrous oxyd gas into nitrous acid gas. The property of
attracting oxygen from the atmosphere, without any elevation of
temperature, has occasioned this gaseous oxyd being used as a test for
ascertaining the degree
110
of purity of the atmosphere. I am going to show you how it is
applied to this purpose.—You see this graduated glass tube, which
is closed at one end, (Plate X. Fig. 2.)—I first
fill it with water, and then introduce a certain measure of nitrous gas,
which, not being absorbable by water, passes through it, and occupies
the upper part of the tube. I must now add rather above two-thirds
of oxygen gas, which will just be sufficient to convert the nitrous oxyd
gas into nitrous acid gas.
CAROLINE.
So it has!—I saw it turn of an orange colour; but it
immediately afterwards disappeared entirely, and the water, you see, has
risen, and almost filled the tube.
MRS. B.
That is because the acid gas is absorbable by water, and in
proportion as the gas impregnates the water, the latter rises in the
tube. When the oxygen gas is very pure, and the required proportion of
nitrous oxyd gas very exact, the whole is absorbed by the water; but if
any other gas be mixed with the oxygen, instead of combining with the
nitrous oxygen, it will remain and occupy the upper part of the tube;
or, if the gases be not in the due proportion, there will be a residue
of that which predominates.—Before we leave this
111
subject, I must not forget to remark that nitrous acid may be
formed by dissolving nitrous oxyd gas in nitric acid. This solution may
be effected simply by making bubbles of nitrous oxyd gas pass through
nitric acid.
EMILY.
That is to say, that nitrogen at its highest degree of oxygenation,
being mixed with nitrogen at its lowest degree of oxygenation, will
produce a kind of intermediate substance, which is nitrous acid.
MRS. B.
You have stated the fact with great precision.—There are
various other methods of preparing nitrous oxyd, and of obtaining it
from compound bodies; but it is not necessary to enter into these
particulars. It remains for me only to mention another curious
modification of oxygenated nitrogen, which has been distinguished by the
name of gaseous oxyd of nitrogen. It is but lately that this gas
has been accurately examined, and its properties have been investigated
chiefly by Sir H. Davy. It has obtained also the name of
exhilarating gas, from the very singular property which that
gentleman has discovered in it, of elevating the animal spirits, when
inhaled into the lungs, to a degree sometimes resembling delirium or
intoxication.
112
CAROLINE.
Is it respirable, then?
MRS. B.
It can scarcely be called respirable, as it would not support life
for any length of time; but it may be breathed for a few moments without
any other effects, than the singular exhilaration of spirits I have just
mentioned. It affects different people, however, in a very different
manner. Some become violent, even outrageous: others experience a
languor, attended with faintness; but most agree in opinion, that the
sensations it excites are extremely pleasant.
CAROLINE.
I think I should like to try it—how do you breathe it?
MRS. B.
By collecting the gas in a bladder, to which a short tube with a
stop-cock is adapted; this is applied to the mouth with one hand, whilst
the nostrils are kept closed with the other, that the common air may
have no access. You then alternately inspire, and expire the gas, till
you perceive its effects. But I cannot consent to your making the
experiment; for the nerves are sometimes unpleasantly affected by it,
and I would not run any risk of that kind.
113
EMILY.
I should like, at least, to see somebody breathe it; but pray by what
means is this curious gas obtained?
MRS. B.
It is procured from nitrat of ammonia, an artificial salt
which yields this gas on the application of a gentle heat. I have
put some of the salt into a retort, and by the aid of a lamp the gas
will be extricated.—
CAROLINE.
Bubbles of air begin to escape through the neck of the retort into
the water apparatus; will you not collect them?
MRS. B.
The gas that first comes over need not be preserved, as it consists
of little more than the common air that was in the retort; besides,
there is always in this experiment a quantity of watery vapour which
must come away before the nitrous oxyd appears.
EMILY.
Watery vapour! Whence does that proceed? There is no water in nitrat
of ammonia?
MRS. B.
You must recollect that there is in every salt a quantity of water of
crystallisation, which may
114
be evaporated by heat alone. But, besides this, water is actually
generated in this experiment, as you will see presently. First tell me,
what are the constituent parts of nitrat of ammonia?
EMILY.
Ammonia, and nitric acid: this salt, therefore, contains three
different elements, nitrogen and hydrogen, which produce the ammonia;
and oxygen, which, with nitrogen, forms the acid.
MRS. B.
Well then, in this process the ammonia is decomposed; the hydrogen
quits the nitrogen to combine with some of the oxygen of the nitric
acid, and forms with it the watery vapour which is now coming over. When
that is effected, what will you expect to find?
EMILY.
Nitrous acid instead of nitric acid, and nitrogen instead of
ammonia.
MRS. B.
Exactly so; and the nitrous acid and nitrogen combine, and form the
gaseous oxyd of nitrogen, in which the proportion of oxygen is 37 parts
to 63 of nitrogen.
You may have observed, that for a little while
115
no bubbles of air have come over, and we have perceived only a stream of
vapour condensing as it issued into the water.—Now bubbles of air
again make their appearance, and I imagine that by this time all the
watery vapour is come away, and that we may begin to collect the gas. We
may try whether it is pure, by filling a phial with it, and plunging a
taper into it—yes, it will do now, for the taper burns brighter
than in the common air, and with a greenish flame.
CAROLINE.
But how is that? I thought no gas would support combustion but oxygen
or chlorine.
MRS. B.
Or any gas that contains oxygen, and is ready to yield it, which is
the case with this in a considerable degree; it is not, therefore,
surprising that it should accelerate the combustion of the taper.
You see that the gas is now produced in great abundance; we shall
collect a large quantity of it, and I dare say that we shall find some
of the family who will be curious to make the experiment of respiring
it. Whilst this process is going on, we may take a general survey of the
most important combinations of the nitric and nitrous acids with the
alkalies.
116
The first of these is nitrat of potash, commonly called
nitre or saltpetre.
CAROLINE.
Is not that the salt with which gunpowder is made?
MRS. B.
Yes. Gunpowder is a mixture of five parts of nitre to one of sulphur,
and one of charcoal.—Nitre from its great proportion of oxygen,
and from the facility with which it yields it, is the basis of most
detonating compositions.
EMILY.
But what is the cause of the violent detonation of gunpowder when set
fire to?
MRS. B.
Detonation may proceed from two causes; the sudden formation or
destruction of an elastic fluid. In the first case, when either a solid
or liquid is instantaneously converted into an elastic fluid, the
prodigious and sudden expansion of the body strikes the air with great
violence, and this concussion produces the sound called detonation.
CAROLINE.
That I comprehend very well; but how can a similar effect be produced
by the destruction of a gas?
117
MRS. B.
A gas can be destroyed only by condensing it to a liquid or solid
state; when this takes place suddenly, the gas, in assuming a new and
more compact form, produces a vacuum, into which the surrounding air
rushes with great impetuosity; and it is by that rapid and violent
motion that the sound is produced. In all detonations, therefore, gases
are either suddenly formed, or destroyed. In that of gunpowder, can you
tell me which of these two circumstances takes place?
EMILY.
As gunpowder is a solid, it must, of course, produce the gases in its
detonation; but how, I cannot tell.
MRS. B.
The constituents of gunpowder, when heated to a certain degree, enter
into a number of new combinations, and are instantaneously converted
into a variety of gases, the sudden expansion of which gives rise to the
detonation.
CAROLINE.
And in what instance does the destruction or condensation of gases
produce detonation?
MRS. B.
I can give you one with which you are well
118
acquainted; the sudden combination of the oxygen and hydrogen gases.
CAROLINE.
True; I recollect perfectly that hydrogen detonates with oxygen when
the two gases are converted into water.
MRS. B.
But let us return to the nitrat of potash.—This salt is
decomposed when exposed to heat, and mixed with any combustible body,
such as carbon, sulphur, or metals, these substances oxydating rapidly
at the expense of the nitrat. I must show you an instance of
this.—I expose to the fire some of the salt in a small iron
ladle, and, when it is sufficiently heated, add to it some powdered
charcoal; this will attract the oxygen from the salt, and be converted
into carbonic acid.—
EMILY.
But what occasions that crackling noise, and those vivid flashes that
accompany it?
MRS. B.
The rapidity with which the carbonic acid gas is formed occasions a
succession of small detonations, which, together with the emission of
flame, is called deflagration.
Nitrat of ammonia we have already noticed,
119
on account of the gaseous oxyd of nitrogen which is obtained
from it.
Nitrat of silver is the lunar caustic, so remarkable for its
property of destroying animal fibre, for which purpose it is often used
by surgeons.—We have said so much on a former occasion, on the
mode in which caustics act on animal matter, that I shall not detain you
any longer on this subject.

We now come to the CARBONIC ACID,
which we have already had many opportunities of noticing. You recollect
that this acid may be formed by the combustion of carbon, whether in its
imperfect state of charcoal, or in its purest form of diamond. And it is
not necessary, for this purpose, to burn the carbon in oxygen gas, as we
did in the preceding lecture; for you need only light a piece of
charcoal and suspend it under a receiver on the water bath. The charcoal
will soon be extinguished, and the air in the receiver will be found
mixed with carbonic acid. The process, however, is much more expeditious
if the combustion be performed in pure oxygen gas.
CAROLINE.
But how can you separate the carbonic acid,
120
obtained in this manner, from the air with which it is mixed?
MRS. B.
The readiest mode is to introduce under the receiver a quantity of
caustic lime, or caustic alkali, which soon attracts the whole of the
carbonic acid to form a carbonat.—The alkali is found increased in
weight, and the volume of the air is diminished by a quantity equal to
that of the carbonic acid which was mixed with it.
EMILY.
Pray is there no method of obtaining pure carbon from carbonic
acid?
MRS. B.
For a long time it was supposed that carbonic acid was not
decompoundable; but Mr. Tennant discovered, a few years ago, that
this acid may be decomposed by burning phosphorus in a closed vessel
with carbonat of soda or carbonat of lime: the phosphorus absorbs the
oxygen from the carbonat, whilst the carbon is separated in the form of
a black powder. This decomposition, however, is not effected simply by
the attraction of the phosphorus for oxygen, since it is weaker than
that of charcoal; but the attraction of the alkali of lime for the
phosphoric acid, unites its power at the same time.
121
CAROLINE.
Cannot we make that experiment?
MRS. B.
Not easily; it requires being performed with extreme nicety, in order
to obtain any sensible quantity of carbon, and the experiment is much
too delicate for me to attempt it. But there can be no doubt of the
accuracy of Mr. Tennant’s results; and all chemists now agree, that one
hundred parts of carbonic acid gas consists of about twenty-eight parts
of carbon to seventy-two of oxygen gas. But if you recollect, we
decomposed carbonic acid gas the other day by burning potassium
in it.
CAROLINE.
True, so we did; and found the carbon precipitated on the regenerated
potash.
MRS. B.
Carbonic acid gas is found very abundantly in nature; it is supposed
to form about one thousandth part of the atmosphere, and is constantly
produced by the respiration of animals; it exists in a great variety of
combinations, and is exhaled from many natural decompositions. It is
contained in a state of great purity in certain caves, such as the
Grotto del Cane, near Naples.
122
EMILY.
I recollect having read an account of that grotto, and of the cruel
experiments made on the poor dogs, to gratify the curiosity of
strangers. But I understood that the vapour exhaled by this cave was
called fixed air.
MRS. B.
That is the name by which carbonic acid was known before its chemical
composition was discovered.—This gas is more destructive of life
than any other; and if the poor animals that are submitted to its
effects are not plunged into cold water as soon as they become
senseless, they do not recover. It extinguishes flame instantaneously.
I have collected some in this glass, which I will pour over the
candle.
CAROLINE.
This is extremely singular—it seems to extinguish it as it were
by enchantment, as the gas is invisible. I never should have
imagined that gas could have been poured like a liquid.
MRS. B.
It can be done with carbonic acid only, as no other gas is
sufficiently heavy to be susceptible of being poured out in the
atmospherical air without mixing with it.
123
EMILY.
Pray by what means did you obtain this gas?
MRS. B.
I procured it from marble. Carbonic acid gas has so strong an
attraction for all the alkalies and alkaline earths, that these are
always found in nature in the state of carbonats. Combined with lime,
this acid forms chalk, which may be considered as the basis of all kinds
of marbles, and calcareous stones. From these substances carbonic acid
is easily separated, as it adheres so slightly to its combinations, that
the carbonats are all decomposable by any of the other acids. I can
easily show you how I obtained this gas; I poured some diluted
sulphuric acid over pulverised marble in this bottle (the same which we
used the other day to prepare hydrogen gas), and the gas escaped through
the tube connected with it; the operation still continues, as you may
easily perceive—
EMILY.
Yes, it does; there is a great fermentation in the glass vessel. What
singular commotion is excited by the sulphuric acid taking possession of
the lime, and driving out the carbonic acid!
CAROLINE.
But did the carbonic acid exist in a gaseous state in the marble?
124
MRS. B.
Certainly not; the acid, when in a state of combination, is capable
of existing in a solid form.
CAROLINE.
Whence, then, does it obtain the caloric necessary to convert it into
gas?
MRS. B.
It may be supplied in this case from the mixture of sulphuric acid
and water, which produces an evolution of heat, even greater than is
required for the purpose; since, as you may perceive by touching the
glass vessel, a considerable quantity of the caloric disengaged
becomes sensible. But a supply of caloric may be obtained also from a
diminution of capacity for heat, occasioned by the new combination which
takes place; and, indeed, this must be the case when other acids are
employed for the disengagement of carbonic acid gas, which do not, like
the sulphuric, produce heat on being mixed with water. Carbonic acid may
likewise be disengaged from its combinations by heat alone, which
restores it to its gaseous state.
CAROLINE.
It appears to me very extraordinary that the same gas, which is
produced by the burning of wood and coals, should exist also in such
bodies
125
as marble, and chalk, which are incombustible substances.
MRS. B.
I will not answer that objection, Caroline, because I think I can put
you in a way of doing it yourself. Is carbonic acid combustible?
CAROLINE.
Why, no—because it is a body that has been already burnt; it is
carbon only, and not the acid, that is combustible.
MRS. B.
Well, and what inference do you draw from this?
CAROLINE.
That carbonic acid cannot render the bodies with which it is united
combustible; but that simple carbon does, and that it is in this
elementary state that it exists in wood, coals, and a great variety of
other combustible bodies.—Indeed, Mrs. B., you are very
ungenerous; you are not satisfied with convincing me that my objections
are frivolous, but you oblige me to prove them so myself.
MRS. B.
You must confess, however, that I make ample amends for the detection
of error, when I enable
126
you to discover the truth. You,
understand, now, I hope, that carbonic acid
is equally produced by the decomposition of chalk, or by the combustion
of charcoal. These processes are certainly of a very different nature;
in the first case the acid is already formed, and requires nothing more
than heat to restore it to its gaseous state; whilst, in the latter, the
acid is actually made by the process of combustion.
CAROLINE.
I understand it now perfectly. But I have just been thinking of
another difficulty, which, I hope, you will excuse my not being
able to remove myself. How does the immense quantity of calcareous
earth, which is spread all over the globe, obtain the carbonic acid with
which it is combined?
MRS. B.
The question is, indeed, not very easy to answer; but I conceive that
the general carbonisation of calcareous matter may have been the effect
of a general combustion, occasioned by some revolution of our globe, and
producing an immense supply of carbonic acid, with which the calcareous
matter became impregnated; or that this may have been effected by a
gradual absorption of carbonic acid from the atmosphere.—But this
would lead us to discussions which we cannot indulge
127
in, without deviating too much from our subject.
EMILY.
How does it happen that we do not perceive the pernicious effects of
the carbonic acid which is floating in the atmosphere?
MRS. B.
Because of the state of very great dilution in which it exists there.
But can you tell me, Emily, what are the sources which keep the
atmosphere constantly supplied with this acid?
EMILY.
I suppose the combustion of wood, coals, and other substances, that
contain carbon.
MRS. B.
And also the breath of animals.
CAROLINE.
The breath of animals! I thought you said that this gas was not at
all respirable, but on the contrary, extremely poisonous.
MRS. B.
So it is; but although animals cannot breathe in carbonic acid gas,
yet, in the process of respiration, they have the power of forming this
gas in
128
their lungs; so that the air which we expire, or reject from the
lungs, always contains a certain proportion of carbonic acid, which is
much greater than that which is commonly found in the atmosphere.
CAROLINE.
But what is it that renders carbonic acid such a deadly poison?
MRS. B.
The manner in which this gas destroys life, seems to be merely by
preventing the access of respirable air; for carbonic acid gas, unless
very much diluted with common air, does not penetrate into the lungs, as
the windpipe actually contracts and refuses it admittance.—But we
must dismiss this subject at present, as we shall have an opportunity of
treating of respiration much more fully, when we come to the chemical
functions of animals.
EMILY.
Is carbonic acid as destructive to the life of vegetables as it is to
that of animals?
MRS. B.
If a vegetable be completely immersed in it, I believe it
generally proves fatal to it; but mixed in certain proportions with
atmospherical air, it is, on the contrary, very favourable to
vegetation.
129
You remember, I suppose, our mentioning the mineral waters, both
natural and artificial, which contain carbonic acid gas?
CAROLINE.
You mean the Seltzer water?
MRS. B.
That is one of those which are the most used; there are, however,
a variety of others into which carbonic acid enters as an
ingredient: all these waters are usually distinguished by the name of
acidulous or gaseous mineral waters.
The class of salts called carbonats is the most numerous in
nature; we must pass over them in a very cursory manner, as the subject
is far too extensive for us to enter on it in detail. The state of
carbonat is the natural state of a vast number of minerals, and
particularly of the alkalies and alkaline earths, as they have so great
an attraction for the carbonic acid, that they are almost always found
combined with it; and you may recollect that it is only by separating
them from this acid, that they acquire that causticity and those
striking qualities which I have formerly described. All marbles, chalks,
shells, calcareous spars, and lime-stones of every description, are
neutral salts, in which lime, their common basis, has lost all
its characteristic properties.
130
EMILY.
But if all these various substances are formed by the union of lime
with carbonic acid, whence arises their diversity of form and
appearance?
MRS. B.
Both from the different proportions of their component parts, and
from a variety of foreign ingredients which may be occasionally blended
with them: the veins and colours of marbles, for instance, proceed from
a mixture of metallic substances; silex and alumine also frequently
enter into these combinations. The various carbonats, therefore, that I
have enumerated, cannot be considered as pure unadulterated neutral
salts, although they certainly belong to that class of bodies.
131
ON THE BORACIC, FLUORIC, MURIATIC, AND OXYGENATED MURIATIC ACIDS; AND ON
MURIATS.—ON IODINE AND IODIC ACID.

MRS. B.
We now come to the three remaining acids
with simple bases, the compound nature of which, though long suspected,
has been but recently proved. The chief of these is the muriatic; but I
shall first describe the two others, as their bases have been obtained
more distinctly than that of the muriatic acid.
You may recollect I mentioned the BORACIC
ACID. This is found very sparingly in some parts of Europe, but
for the use of manufactures we have always received it from the remote
country of Thibet, where it is found in some lakes, combined with soda.
It is easily separated from the soda by sulphuric acid, and appears in
the form of shining scales, as you see here.
CAROLINE.
I am glad to meet with an acid which we need
132
not be afraid to touch; for I perceive, from your keeping it in a piece
of paper, that it is more innocent than our late acquaintance, the
sulphuric and nitric acids.
MRS. B.
Certainly; but being more inert, you will not find its properties so
interesting. However, its decomposition, and the brilliant spectacle it
affords when its basis again unites with oxygen, atones for its want of
other striking qualities.
Sir H. Davy succeeded in decomposing the boracic acid, (which had
till then been considered as undecompoundable,) by various methods. On
exposing this acid to the Voltaic battery, the positive wire gave out
oxygen, and on the negative wire was deposited a black substance, in
appearance resembling charcoal. This was the basis of the acid, which
Sir H. Davy has called Boracium, or Boron.
The same substance was obtained in more considerable quantities, by
exposing the acid to a great heat in an iron gun-barrel.
A third method of decomposing the boracic acid consisted in burning
potassium in contact with it in vacuo. The potassium attracts the oxygen
from the acid, and leaves its basis in a separate state.
The recomposition of this acid I shall show
133
you, by burning some of its basis, which you see here, in a retort full
of oxygen gas. The heat of a candle is all that is required for this
combustion.—
EMILY.
The light is astonishingly brilliant, and what beautiful sparks it
throws out!
MRS. B.
The result of this combustion is the boracic acid, the nature of
which, you see, is proved both by analytic and synthetic means. Its
basis has not, it is true, a metallic appearance; but it makes very
hard alloys with other metals.
EMILY.
But pray, Mrs. B., for what purpose is the boracic acid used in
manufactures?
MRS. B.
Its principal use is in conjunction with soda, that is, in the state
of borat of soda, which in the arts is commonly called borax.
This salt has a peculiar power of dissolving metallic oxyds, and of
promoting the fusion of substances capable of being melted; it is
accordingly employed in various metallic arts; it is used, for example,
to remove the oxyd from the surface of metals, and
134
is often employed in the assaying of metallic ores.
Let us now proceed to the FLUORIC
ACID. This acid is obtained from a substance which is found
frequently in mines, and particularly in those of Derbyshire, called
fluor, a name which it acquired from the circumstance of its
being used to render the ores of metals more fluid when heated.
CAROLINE.
Pray is not this the Derbyshire spar, of which so many ornaments are
made?
MRS. B.
The same; but though it has long been employed for a variety of
purposes, its nature was unknown until Scheele, the great Swedish
chemist, discovered that it consisted of lime united with a peculiar
acid, which obtained the name of fluoric acid. It is easily
separated from the lime by the sulphuric acid, and unless condensed in
water, ascends in the form of gas. A very peculiar property of this
acid is its union with siliceous earths, which I have already mentioned.
If the distillation of this acid is performed in glass vessels, they are
corroded, and the siliceous part of the glass comes over, united with
the gas; if water
135
is then admitted, part of the silex is deposited, as you may observe in
this jar.
CAROLINE.
I see white flakes forming on the surface of the water; is that
silex?
MRS. B.
Yes it is. This power of corroding glass has been used for engraving,
or rather etching, upon it. The glass is first covered with a coat of
wax, through which the figures to be engraved are to be scratched with a
pin; then pouring the fluoric acid over the wax, it corrodes the glass
where the scratches have been made.
CAROLINE.
I should like to have a bottle of this acid, to make engravings.
MRS. B.
But you could not have it in a glass bottle, for in that case
the acid would be saturated with silex, and incapable of executing an
engraving; the same thing would happen were the acid kept in vessels of
porcelain or earthen-ware; this acid must therefore be both prepared and
preserved in vessels of silver.
136
If it be distilled from fluor spar and vitriolic acid, in silver or
leaden vessels, the receiver being kept very cold during the
distillation, it assumes the form of a dense fluid, and in that state is
the most intensely corrosive substance known. This seems to be the acid
combined with a little water. It may be called hydro-fluoric
acid; and Sir H. Davy has been led, from some late experiments on
the subject, to consider pure fluoric acid as a compound of a
certain unknown principle, which he calls fluorine, with
hydrogen.
Sir H. Davy has also attempted to decompose the fluoric acid by
burning potassium in contact with it; but he has not yet been able by
this or any other method, to obtain its basis in a distinct separate
state.
We shall conclude our account of the acids with that of the MURIATIC ACID, which is perhaps the most curious
and interesting of all of them. It is found in nature combined with
soda, lime, and magnesia. Muriat of soda is the common sea-salt,
and from this substance the acid is usually disengaged by means of the
sulphuric acid. The natural state of the muriatic acid is that of an
invisible permanent gas, at the common temperature of the atmosphere;
but it has a remarkably strong attraction for water, and assumes
137
the form of a whitish cloud whenever it meets any moisture to combine
with. This acid is remarkable for its peculiar and very pungent smell,
and possesses, in a powerful degree, most of the acid properties. Here
is a bottle containing muriatic acid in a liquid state.
CAROLINE.
And how is it liquefied?
MRS. B.
By impregnating water with it; its strong attraction for water makes
it very easy to obtain it in a liquid form. Now, if I open the phial,
you may observe a kind of vapour rising from it, which is muriatic acid
gas, of itself invisible, but made apparent by combining with the
moisture of the atmosphere.
EMILY.
Have you not any of the pure muriatic acid gas?
MRS. B.
This jar is full of that acid in its gaseous state—it is
inverted over mercury instead of water, because, being absorbable by
water, this gas cannot be confined by it.—I shall now raise
the jar a little on one side, and suffer some of the gas to
138
escape.—You see that it immediately becomes visible in the form of
a cloud.
EMILY.
It must be, no doubt, from its uniting with the moisture of the
atmosphere, that it is converted into this dewy vapour.
MRS. B.
Certainly; and for the same reason, that is to say, its extreme
eagerness to unite with water, this gas will cause snow to melt as
rapidly as an intense fire.
This acid proved much more refractory when Sir H. Davy attempted to
decompose it than the other two undecompounded acids. It is singular
that potassium will burn in muriatic acid, and be converted into potash,
without decomposing the acid, and the result of this combustion is a
muriat of potash; for the potash, as soon as it is regenerated,
combines with the muriatic acid.
CAROLINE.
But how can the potash be regenerated if the muriatic acid does not
oxydate the potassium?
MRS. B.
The potassium, in this process, obtains oxygen from the moisture with
which the muriatic acid is
139
always combined, and accordingly hydrogen, resulting from the
decomposition of the moisture, is invariably evolved.
EMILY.
But why not make these experiments with dry muriatic acid?
MRS. B.
Dry acids cannot be acted on by the Voltaic battery, because acids
are non-conductors of electricity, unless moistened. In the course of a
number of experiments which Sir H. Davy made upon acids in a state of
dryness, he observed that the presence of water appeared always
necessary to develop the acid properties, so that acids are not even
capable of reddening vegetable blues if they have been carefully
deprived of moisture. This remarkable circumstance led him to suspect,
that water, instead of oxygen, may be the acidifying principle; but this
he threw out rather as a conjecture than as an established point.
Sir H. Davy obtained very curious results from burning potassium in a
mixture of phosphorus and muriatic acid, and also of sulphur and
muriatic acid; the latter detonates with great violence. All his
experiments, however, failed in presenting to his view the basis of the
muriatic acid, of which he was in search; and he was at last induced to
140
form an opinion respecting the nature of this acid, which I shall
presently explain.
EMILY.
Is this acid susceptible of different degrees of oxygenation?
MRS. B.
Yes, for though we cannot deoxygenate this acid, yet we may add
oxygen to it.
CAROLINE.
Why, then, is not the least degree of oxygenation of the acid called
the muriatous, and the higher degree the muriatic
acid?
MRS. B.
Because, instead of becoming, like other acids, more dense, and more
acid by an addition of oxygen, it is rendered on the contrary more
volatile, more pungent, but less acid, and less absorbable by water.
These circumstances, therefore, seem to indicate the propriety of making
an exception to the nomenclature. The highest degree of oxygenation of
this acid has been distinguished by the additional epithet of
oxygenated, or, for the sake of brevity, oxy, so that it
is called the oxygenated, or oxy-muriatic acid. This
likewise exists in a gaseous form, at the temperature of the atmosphere;
it is also susceptible of being absorbed
141
by water, and can be congealed, or solidified, by a certain degree of
cold.
EMILY.
And how do you obtain the oxy-muriatic acid?
MRS. B.
In various ways; but it may be most conveniently obtained by
distilling liquid muriatic acid over oxyd of manganese, which supplies
the acid with the additional oxygen. One part of the acid being put into
a retort, with two parts of the oxyd of manganese, and the heat of a
lamp applied, the gas is soon disengaged, and may be received over
water, as it is but sparingly absorbed by it.—I have
collected some in this jar—
CAROLINE.
It is not invisible, like the generality of gases; for it is of a
yellowish colour.
MRS. B.
The muriatic acid extinguishes flame, whilst, on the contrary, the
oxy-muriatic makes the flame larger, and gives it a dark red colour. Can
you account for this difference in the two acids?
EMILY.
Yes, I think so; the muriatic acid will not supply
142
the flame with the oxygen necessary for its support; but when this acid
is further oxygenated, it will part with its additional quantity of
oxygen, and in this way support combustion.
MRS. B.
That is exactly the case; indeed the oxygen added to the muriatic
acid, adheres so slightly to it, that it is separated by mere exposure
to the sun’s rays. This acid is decomposed also by combustible bodies,
many of which it burns, and actually inflames, without any previous
increase of temperature.
CAROLINE.
That is extraordinary, indeed! I hope you mean to indulge us with
some of these experiments?
MRS. B.
I have prepared several glass jars of oxy-muriatic acid gas for that
purpose. In the first we shall introduce some Dutch gold leaf.—Do
you observe that it takes fire?
EMILY.
Yes, indeed it does—how wonderful it is! It became immediately
red hot, but was soon smothered in a thick vapour.
CAROLINE.
What a disagreeable smell!
143
MRS. B.
We shall try the same experiment with phosphorus in another jar of
this acid.—You had better keep your handkerchief to your nose when
I open it—now let us drop into it this little piece of
phosphorus—
CAROLINE.
It burns really; and almost as brilliantly as in oxygen gas! But,
what is most extraordinary, these combustions take place without the
metal or phosphorus being previously lighted, or even in the least
heated.
MRS. B.
All these curious effects are owing to the very great facility with
which this acid yields oxygen to such bodies as are strongly disposed to
combine with it. It appears extraordinary indeed to see bodies, and
metals in particular, melted down and inflamed, by a gas without any
increase of temperature, either of the gas, or of the combustible. The
phenomenon, however, is, you see, well accounted for.
EMILY.
Why did you burn a piece of Dutch gold leaf rather than a piece of
any other metal?
MRS. B.
Because, in the first place, it is a composition
144
of metals (consisting chiefly of copper) which burns readily; and I use
a thin metallic leaf in preference to a lump of metal, because it offers
to the action of the gas but a small quantity of matter under a large
surface. Filings, or shavings, would answer the purpose nearly as well;
but a lump of metal, though the surface would oxydate with great
rapidity, would not take fire. Pure gold is not inflamed by oxy-muriatic
acid gas, but it is rapidly oxydated, and dissolved by it; indeed, this
acid is the only one that will dissolve gold.
EMILY.
This, I suppose, is what is commonly called aqua regia, which
you know is the only thing that will act upon gold.
MRS. B.
That is not exactly the case either; for aqua regia is composed of a
mixture of muriatic acid and nitric acid.—But, in fact, the result
of this mixture is the formation of oxy-muriatic acid, as the muriatic
acid oxygenates itself at the expence of the nitric; this mixture,
therefore, though it bears the name of nitro-muriatic acid, acts
on gold merely in virtue of the oxy-muriatic acid which it contains.
Sulphur, volatile oils, and many other substances, will burn in the
same manner in oxy-muriatic
145
acid gas; but I have not prepared a sufficient quantity of it, to show
you the combustion of all these bodies.
CAROLINE.
There are several jars of the gas yet remaining.
MRS. B.
We must reserve these for future experiments. The oxy-muriatic acid
does not, like other acids, redden the blue vegetable colours; but it
totally destroys any colour, and turns all vegetables perfectly white.
Let us collect some vegetable substances to put into this glass, which
is full of gas.
EMILY.
Here is a sprig of myrtle—
CAROLINE.
And here some coloured paper—
MRS. B.
We shall also put in this piece of scarlet riband, and a
rose—
EMILY.
Their colours begin to fade immediately! But how does the gas produce
this effect?
MRS. B.
The oxygen combines with the colouring matter of these substances,
and destroys it; that is to
146
say, destroys the property which these colours had of reflecting only
one kind of rays, and renders them capable of reflecting them all,
which, you know, will make them appear white. Old prints may be cleaned
by this acid, for the paper will be whitened without injury to the
impression, as printer’s ink is made of materials (oil and lamp black)
which are not acted upon by acids.
This property of the oxy-muriatic acid has lately been employed in
manufactures in a variety of bleaching processes; but for these purposes
the gas must be dissolved in water, as the acid is thus rendered much
milder and less powerful in its effects; for, in a gaseous state, it
would destroy the texture, as well as the colour of the substance
submitted to its action.
CAROLINE.
Look at the things which we put into the gas; they have now entirely
lost their colour!
MRS. B.
The effect of the acid is almost completed; and, if we were to
examine the quantity that remains, we should find it to consist chiefly
of muriatic acid.
The oxy-muriatic acid has been used to purify the air in fever
hospitals and prisons, as it burns and destroys putrid effluvia of every
kind. The
147
infection of the small-pox is likewise destroyed by this gas, and matter
that has been submitted to its influence will no longer generate that
disorder.
CAROLINE.
Indeed, I think the remedy must be nearly as bad as the disease; the
oxy-muriatic acid has such a dreadfully suffocating smell.
MRS. B.
It is certainly extremely offensive; but by keeping the mouth shut,
and wetting the nostrils with liquid ammonia, in order to neutralize the
vapour as it reaches the nose, its prejudicial effects may be in some
degree prevented. At any rate, however, this mode of disinfection can
hardly be used in places that are inhabited. And as the vapour of nitric
acid, which is scarcely less efficacious for this purpose, is not at all
prejudicial, it is usually preferred on such occasions.
CAROLINE.
You have not told us yet what is Sir H. Davy’s new opinion respecting
the nature of muriatic acid, to which you alluded a few minutes ago?
MRS. B.
True; I avoided noticing it then, because you could not have
understood it without some previous
148
knowledge of the oxy-muriatic acid, which I have but just introduced to
your acquaintance.
Sir H. Davy’s idea is that muriatic acid, instead of being a
compound, consisting of an unknown basis and oxygen, is formed by the
union of oxy-muriatic gas with hydrogen.
EMILY.
Have you not told us just now that oxy-muriatic gas was itself a
compound of muriatic acid and oxygen?
MRS. B.
Yes; but according to Sir H. Davy’s hypothesis, oxy-muriatic gas is
considered as a simple body, which contains no oxygen—as a
substance of its own kind, which has a great analogy to oxygen in most
of its properties, though in others it differs entirely from
it.—According to this view of the subject, the name of
oxy-muriatic acid can no longer be proper, and therefore Sir H.
Davy has adopted that of chlorine, or chlorine gas,
a name which is simply expressive of its greenish colour; and in
compliance with that philosopher’s theory, we have placed chlorine in
our table among the simple bodies.
CAROLINE.
But what was Sir H. Davy’s reason for adopting an opinion so contrary
to that which had hitherto prevailed?
149
MRS. B.
There are many circumstances which are favourable to the new
doctrine; but the clearest and simplest fact in its support is, that if
hydrogen gas and oxy-muriatic gas be mixed together, both these gases
disappear, and muriatic acid gas is formed.
EMILY.
That seems to be a complete proof; is it not considered as perfectly
conclusive?
MRS. B.
Not so decisive as it appears at first sight; because it is argued by
those who still incline to the old doctrine, that muriatic acid gas,
however dry it may be, always contains a certain quantity of water,
which is supposed essential to its formation. So that, in the experiment
just mentioned, this water is supplied by the union of the hydrogen gas
with the oxygen of the oxy-muriatic acid; and therefore the mixture
resolves itself into the base of muriatic acid and water, that is,
muriatic acid gas.
CAROLINE.
I think the old theory must be the true one; for otherwise how could
you explain the formation of oxy-muriatic gas, from a mixture of
muriatic acid and oxyd of manganese?
150
MRS. B.
Very easily; you need only suppose that in this process the muriatic
acid is decomposed; its hydrogen unites with the oxygen of the manganese
to form water, and the chlorine appears in its separate state.
EMILY.
But how can you explain the various combustions which take place in
oxy-muriatic gas, if you consider it as containing no oxygen?
MRS. B.
We need only suppose that combustion is the result of intense
chemical action; so that chlorine, like oxygen, in combining with
bodies, forms compounds which have less capacity for caloric than their
constituent principles, and, therefore, caloric is evolved at the moment
of their combination.
EMILY.
If, then, we may explain every thing by either theory, to which of
the two shall we give the preference?
MRS. B.
It will, perhaps, be better to wait for more positive proofs, if such
can be obtained, before we decide positively upon the subject. The new
doctrine has certainly gained ground very rapidly, and may be considered
as nearly established; but several
151
competent judges still refuse their assent to it, and until that theory
is very generally adopted, it may be as well for us still occasionally
to use the language to which chemists have long been
accustomed.—But let us proceed to the examination of salts formed
by muriatic acid.
Among the compound salts formed by muriatic acid, the muriat of
soda, or common salt, is the most interesting.* The uses and properties of
this salt are too well known to require much comment. Besides the
pleasant flavour it imparts to the food, it is very wholesome, when not
used to excess, as it assists the process of digestion.
Sea-water is the great source from which muriat of soda is extracted
by evaporation. But it is also found in large solid masses in the bowels
of the earth, in England, and in many other parts of the world.
EMILY.
I thought that salts, when solid, were always in the state of
crystals; but the common table-salt is in the form of a coarse white
powder.
152
MRS. B.
Crystallisation depends, as you may recollect, on the slow and
regular reunion of particles dissolved in a fluid; common sea-salt is
only in a state of imperfect crystallisation, because the process by
which it is prepared is not favourable to the formation of regular
crystals. But if you dissolve it, and afterwards evaporate the water
slowly, you will obtain a regular crystallisation.
Muriat of ammonia is another combination of this acid, which
we have already mentioned as the principal source from which ammonia is
derived.
I can at once show you the formation of this salt by the immediate
combination of muriatic acid with ammonia.—These two glass jars
contain, the one muriatic acid gas, the other ammoniacal gas, both of
which are perfectly invisible—now, if I mix them together, you see
they immediately form an opake white cloud, like smoke.—If a
thermometer was placed in the jar in which these gases are mixed, you
would perceive that some heat is at the same time produced.
EMILY.
The effects of chemical combinations are, indeed,
wonderful!—How extraordinary it is that two invisible bodies
should become visible by their union!
153
MRS. B.
This strikes you with astonishment, because it is a phenomenon which
nature seldom exhibits to our view; but the most common of her
operations are as wonderful, and it is their frequency only that
prevents our regarding them with equal admiration. What would be more
surprising, for instance, than combustion, were it not rendered so
familiar by custom?
EMILY.
That is true.—But pray, Mrs. B., is this white cloud the salt
that produces ammonia? How different it is from the solid muriat of
ammonia which you once showed us!
MRS. B.
It is the same substance which first appears in the state of vapour,
but will soon be condensed by cooling against the sides of the jar, in
the form of very minute crystals.
We may now proceed to the oxy-muriats. In this class of salts
the oxy-muriat of potash is the most worthy of our attention, for
its striking properties. The acid, in this state of combination,
contains a still greater proportion of oxygen than when alone.
CAROLINE.
But how can the oxy-muriatic acid acquire an increase of oxygen by
combining with potash?
154
MRS. B.
It does not really acquire an additional quantity of oxygen, but it
loses some of the muriatic acid, which produces the same effect, as the
acid which remains is proportionably super-oxygenated.*
If this salt be mixed, and merely rubbed together with sulphur,
phosphorus, charcoal, or indeed any other combustible, it explodes
strongly.
CAROLINE.
Like gun-powder, I suppose, it is suddenly converted into elastic
fluids?
MRS. B.
Yes; but with this remarkable difference, that no increase of
temperature, any further than is produced by gentle friction, is
required in this instance. Can you tell me what gases are generated by
the detonation of this salt with charcoal?
EMILY.
Let me consider . . . . . The oxy-muriatic acid parts with its excess
of oxygen to the charcoal, by which means it is converted into muriatic
acid gas; whilst the charcoal, being burnt by the oxygen, is
155
changed to carbonic acid gas.—What becomes of the potash I cannot
tell.
MRS. B.
That is a fixed product which remains in the vessel.
CAROLINE.
But since the potash does not enter into the new combinations,
I do not understand of what use it is in this operation. Would not
the oxy-muriatic acid and the charcoal produce the same effect
without it?
MRS. B.
No; because there would not be that very great concentration of
oxygen which the combination with the potash produces, as I have just
explained.
I mean to show you this experiment, but I would advise you not to
repeat it alone; for if care be not taken to mix only very small
quantities at a time, the detonation will be extremely violent, and may
be attended with dangerous effects. You see I mix an exceedingly small
quantity of the salt with a little powdered charcoal, in this Wedgwood
mortar, and rub them together with the pestle—
CAROLINE.
Heavens! How can such a loud explosion be produced by so small a
quantity of matter?
156
MRS. B.
You must consider that an extremely small quantity of solid substance
may produce a very great volume of gases; and it is the sudden evolution
of these which occasions the sound.
EMILY.
Would not oxy-muriat of potash make stronger gunpowder than nitrat of
potash?
MRS. B.
Yes; but the preparation, as well as the use of this salt, is
attended with so much danger, that it is never employed for that
purpose.
CAROLINE.
There is no cause to regret it, I think; for the common gunpowder is
quite sufficiently destructive.
MRS. B.
I can show you a very curious experiment with this salt; but it must
again be on condition that you will never attempt to repeat it by
yourselves. I throw a small piece of phosphorus into this glass of
water; then a little oxy-muriat of potash; and, lastly, I pour in
(by means of this funnel, so as to bring it in contact with the two
other ingredients at the bottom of the glass) a small quantity of
sulphuric acid—
157
CAROLINE.
This is, indeed, a beautiful experiment! The phosphorus takes fire
and burns from the bottom of the water.
EMILY.
How wonderful it is to see flame bursting out under water, and rising
through it! Pray, how is this accounted for?
MRS. B.
Cannot you find it out, Caroline?
EMILY.
Stop—I think I can explain it. Is it not because the sulphuric
acid decomposes the salt by combining with the potash, so as to liberate
the oxy-muriatic acid gas by which the phosphoric is set on fire?
MRS. B.
Very well, Emily; and with a little more reflection you would have
discovered another concurring circumstance, which is, that an increase
of temperature is produced by the mixture of the sulphuric acid and
water, which assists in promoting the combustion of the phosphorus.
I must, before we part, introduce to your acquaintance the
newly-discovered substance IODINE,
which you may recollect we placed next to oxygen and chlorine in our
table of simple bodies.
158
CAROLINE.
Is this also a body capable of maintaining combustion like oxygen and
chlorine?
MRS. B.
It is; and although it does not so generally disengage light and heat
from inflammable bodies, as oxygen and chlorine do, yet it is capable of
combining with most of them; and sometimes, as in the instance of
potassium and phosphorus, the combination is attended with an actual
appearance of light and heat.
CAROLINE.
But what sort of a substance is iodine: what is its form, and
colour?
MRS. B.
It is a very singular body, in many respects. At the ordinary
temperature of the atmosphere, it commonly appears in the form of
blueish black crystalline scales, such as you see in this tube.
CAROLINE.
They shine like black lead, and some of the scales have the shape of
lozenges.
MRS. B.
That is actually the form which the crystals of iodine often assume.
But if we heat them gently,
159
by holding the tube over the flame of a candle, see what a change takes
place in them.
CAROLINE.
How curious! They seem to melt, and the tube immediately fills with a
beautiful violet vapour. But look, Mrs. B., the same scales are now
appearing at the other end of the tube.
MRS. B.
This is in fact a sublimation of iodine, from one part of the tube to
another; but with this remarkable peculiarity, that, while in the
gaseous state, iodine assumes that bright violet colour, which, as you
may already perceive, it loses as the tube cools, and the substance
resumes its usual solid form.—It is from the violet colour of the
gas that iodine has obtained its name.
CAROLINE.
But how is this curious substance obtained?
MRS. B.
It is found in the ley of ashes of sea-weeds, after the soda has been
separated by crystallisation; and it is disengaged by means of sulphuric
acid, which expels it from the alkaline ley in the form of a violet gas,
which may be collected and condensed in the way you have just
160
seen.—This interesting discovery was made in the year 1812, by M.
Courtois, a manufacturer of saltpetre at Paris.
CAROLINE.
And pray, Mrs. B., what is the proof of iodine being a simple
body?
MRS. B.
It is considered as a simple body, both because it is not capable of
being resolved into other ingredients; and because it is itself capable
of combining with other bodies, in a manner analogous to oxygen and
chlorine. The most curious of these combinations is that which it forms
with hydrogen gas, the result of which is a peculiar gaseous acid.
CAROLINE.
Just as chlorine and hydrogen gas form muriatic acid? In this respect
chlorine and iodine seem to bear a strong analogy to each other.
MRS. B.
That is indeed the case; so that if the theory of the constitution of
either of these two bodies be true, it must be true also in regard to
the other; if erroneous in the one, the theory must fall in both.
161
But it is now time to conclude; we have examined such of the acids
and salts as I conceived would appear to you most
interesting.—I shall not enter into any particulars
respecting the metallic acids, as they offer nothing sufficiently
striking for our present purpose.
162
CONVERSATION XX.
ON THE NATURE AND COMPOSITION OF
VEGETABLES.

MRS. B.
We have hitherto treated only of the
simplest combinations of elements, such as alkalies, earths, acids,
compound salts, stones, &c.; all of which belong to the mineral
kingdom. It is time now to turn our attention to a more complicated
class of compounds, that of ORGANISED
BODIES, which will furnish us with a new source of instruction
and amusement.
EMILY.
By organised bodies, I suppose, you mean the vegetable and animal
creation? I have, however, but a very vague idea of the word
organisation, and I have often wished to know more precisely what
it means.
163
MRS. B.
Organised bodies are such as are endowed by nature with various
parts, peculiarly constructed and adapted to perform certain functions
connected with life. Thus you may observe, that mineral compounds are
formed by the simple effect of mechanical or chemical attraction, and
may appear to some to be in a great measure the productions of chance;
whilst organised bodies bear the most striking and impressive marks of
design, and are eminently distinguished by that unknown principle,
called life, from which the various organs derive the power of
exercising their respective functions.
CAROLINE.
But in what manner does life enable these organs to perform their
several functions?
MRS. B.
That is a mystery which, I fear, is enveloped in such profound
darkness that there is very little hope of our ever being able to unfold
it. We must content ourselves with examining the effects of this
principle; as for the cause, we have been able only to give it a name,
without attaching any other meaning to it than the vague and
unsatisfactory idea of au unknown agent.
164
CAROLINE.
And yet I think I can form a very clear idea of life.
MRS. B.
Pray let me hear how you would define it?
CAROLINE.
It is perhaps more easy to conceive than to express—let me
consider—Is not life the power which enables both the animal and
the vegetable creation to perform the various functions which nature has
assigned to them?
MRS. B.
I have nothing to object to your definition; but you will allow me to
observe, that you have only mentioned the effects which the unknown
cause produces, without giving us any notion of the cause itself.
EMILY.
Yes, Caroline, you have told us what life does, but you have
not told us what it is.
MRS. B.
We may study its operations, but we should puzzle ourselves to no
purpose by attempting to form an idea of its real nature.
We shall begin with examining its effects in the
165
vegetable world, which constitutes the simplest class of organised
bodies; these we shall find distinguished from the mineral creation, not
only by their more complicated nature, but by the power which they
possess within themselves, of forming new chemical arrangements of their
constituent parts, by means of appropriate organs. Thus, though all
vegetables are ultimately composed of hydrogen, carbon, and oxygen,
(with a few other occasional ingredients,) they separate and combine
these principles by their various organs, in a thousand ways, and form,
with them, different kinds of juices and solid parts, which exist ready
made in vegetables, and may, therefore, be considered as their immediate
materials.
These are:
Sap,
Mucilage,
Sugar,
Fecula,
Gluten,
Fixed Oil,
Volatile Oil,
Camphor,
Resins,
Gum Resins,
Balsams,
Caoutchouc,
Extractive colouring Matter,
Tannin,
Woody Fibre,
Vegetable Acids, &c.
CAROLINE.
What a long list of names! I did not suppose
166
that a vegetable was composed of half so many ingredients.
MRS. B.
You must not imagine that every one of these materials is formed in
each individual plant. I only mean to say, that they are all
derived exclusively from the vegetable kingdom.
EMILY.
But does each particular part of the plant, such as the root, the
bark, the stem, the seeds, the leaves, consist of one of these
ingredients only, or of several of them combined together?
MRS. B.
I believe there is no part of a plant which can be said to consist
solely of any one particular ingredient; a certain number of
vegetable materials must always be combined for the formation of any
particular part, (of a seed for instance,) and these combinations are
carried on by sets of vessels, or minute organs, which select from other
parts, and bring together, the several principles required for the
development and growth of those particular parts which they are intended
to form and to maintain.
EMILY.
And are not these combinations always regulated by the laws of
chemical attraction?
167
MRS. B.
No doubt; the organs of plants cannot force principles to combine
that have no attraction for each other; nor can they compel superior
attractions to yield to those of inferior power; they probably act
rather mechanically, by bringing into contact such principles, and in
such proportions, as will, by their chemical combination, form the
various vegetable products.
CAROLINE.
We may then consider each of these organs as a curiously constructed
apparatus, adapted for the performance of a variety of chemical
processes.
MRS. B.
Exactly so. As long as the plant lives and thrives, the carbon,
hydrogen, and oxygen, (the chief constituents of its immediate
materials,) are so balanced and connected together, that they are not
susceptible of entering into other combinations; but no sooner does
death take place, than this state of equilibrium is destroyed, and new
combinations produced.
EMILY.
But why should death destroy it; for these principles must remain in
the same proportions, and consequently, I should suppose, in the
same order of attractions?
168
MRS. B.
You must remember, that in the vegetable, as well as in the animal
kingdom, it is by the principle of life that the organs are
enabled to act; when deprived of that agent or stimulus, their power
ceases, and an order of attractions succeeds similar to that which would
take place in mineral or unorganised matter.
EMILY.
It is this new order of attractions, I suppose, that destroys the
organisation of the plant after death; for if the same combinations
still continued to prevail, the plant would always remain in the state
in which it died?
MRS. B.
And that, you know, is never the case; plants may be partially
preserved for some time after death, by drying; but in the natural
course of events they all return to the state of simple elements;
a wise and admirable dispensation of Providence, by which dead
plants are rendered fit to enrich the soil, and become subservient to
the nourishment of living vegetables.
CAROLINE.
But we are talking of the dissolution of plants, before we have
examined them in their living state.
169
MRS. B.
That is true, my dear. But I wished to give you a general idea of the
nature of vegetation, before we entered into particulars. Besides, it is
not so irrelevant as you suppose to talk of vegetables in their dead
state, since we cannot analyse them without destroying life; and it is
only by hastening to submit them to examination, immediately after they
have ceased to live, that we can anticipate their natural decomposition.
There are two kinds of analysis of which vegetables are susceptible;
first, that which separates them into their immediate materials, such as
sap, resin, mucilage, &c.; secondly, that which decomposes them into
their primitive elements, as carbon, hydrogen, and oxygen.
EMILY.
Is there not a third kind of analysis of plants, which consists in
separating their various parts, as the stem, the leaves, and the several
organs of the flower?
MRS. B.
That, my dear, is rather the department of the botanist; we shall
consider these different parts of plants only, as the organs by which
the various secretions or separations are performed; but we must first
examine the nature of these secretions.
170
The sap is the principal material of vegetables, since it
contains the ingredients that nourish every part of the plant. The basis
of this juice, which the roots suck up from the soil, is water; this
holds in solution the various other ingredients required by the several
parts of the plant, which are gradually secreted from the sap by the
different organs appropriated to that purpose, as it passes them in
circulating through the plant.
Mucus, or mucilage, is a vegetable substance, which, like
all the others, is secreted from the sap; when in excess, it exudes from
trees in the form of gum.
CAROLINE.
Is that the gum so frequently used instead of paste or glue?
MRS. B.
It is; almost all fruit-trees yield some sort of gum, but that most
commonly used in the arts is obtained from a species of acacia-tree in
Arabia, and is called gum arabic; it forms the chief nourishment
of the natives of those parts, who obtain it in great quantities from
incisions which they make in the trees.
CAROLINE.
I did not know that gum was eatable.
171
MRS. B.
There is an account of a whole ship’s company being saved from
starving by feeding on the cargo, which was gum senegal. I should
not, however, imagine, that it would be either a pleasant or a
particularly eligible diet to those who have not, from their birth, been
accustomed to it. It is, however, frequently taken medicinally, and
considered as very nourishing. Several kinds of vegetable acids may be
obtained, by particular processes, from gum or mucilage, the principal
of which is called the mucous acid.
Sugar is not found in its simple state in plants, but is always
mixed with gum, sap, or other ingredients; this saccharine matter is to
be met with in every vegetable, but abounds most in roots, fruits, and
particularly in the sugar-cane.
EMILY.
If all vegetables contain sugar, why is it extracted exclusively from
the sugar-cane?
MRS. B.
Because it is both most abundant in that plant, and most easily
obtained from it. Besides, the sugars produced by other vegetables
differ a little in their nature.
172
During the late troubles in the West-Indies, when Europe was but
imperfectly supplied with sugar, several attempts were made to extract
it from other vegetables, and very good sugar was obtained from parsnips
and from carrots; but the process was too expensive to carry this
enterprize to any extent.
CAROLINE.
I should think that sugar might be more easily obtained from sweet
fruits, such as figs, dates, &c.
MRS. B.
Probably; but it would be still more expensive, from the high price
of those fruits.
EMILY.
Pray, in what manner is sugar obtained from the sugar-cane?
MRS. B.
The juice of this plant is first expressed by passing it between two
cylinders of iron. It is then boiled with lime-water, which makes a
thick scum rise to the surface. The clarified liquor is let off below
and evaporated to a very small quantity, after which it is suffered to
crystallise by standing in a vessel, the bottom of which is perforated
173
with holes, that are imperfectly stopped, in order that the syrup may
drain off. The sugar obtained by this process is a coarse brown powder,
commonly called raw or moist sugar; it undergoes another operation to be
refined and converted into loaf sugar. For this purpose it is dissolved
in water, and afterwards purified by an animal fluid called albumen.
White of eggs chiefly consist of this fluid, which is also one of the
constituent parts of blood; and consequently eggs, or bullocks’ blood,
are commonly used for this purpose.
The albuminous fluid being diffused through the syrup, combines with
all the solid impurities contained in it, and rises with them to the
surface, where it forms a thick scum; the clear liquor is then again
evaporated to a proper consistence, and poured into moulds, in which, by
a confused crystallisation, it forms loaf-sugar. But an additional
process is required to whiten it; to this effect the mould is inverted,
and its open base is covered with clay, through which water is made to
pass; the water slowly trickling through the sugar, combines with and
carries off the colouring matter.
CAROLINE.
I am very glad to hear that the blood that is used to purify sugar
does not remain in it; it would be a disgusting idea. I have heard
of some
174
improvements by the late Mr. Howard, in the process of refining sugar.
Pray what are they?
MRS. B.
It would be much too long to give you an account of the process in
detail. But the principal improvement relates to the mode of evaporating
the syrup, in order to bring it to the consistency of sugar. Instead of
boiling the syrup in a large copper, over a strong fire, Mr. Howard
carries off the water by means of a large air-pump, in a way similar to
that used in Mr. Leslie’s experiment for freezing water by evaporation;
that is, the syrup being exposed to a vacuum, the water evaporates
quickly, with no greater heat than that of a little steam, which is
introduced round the boiler. The air-pump is of course of large
dimensions, and is worked by a steam engine. A great saving is thus
obtained, and a striking instance afforded of the power of science in
suggesting useful economical improvements.
EMILY.
And pray how is sugar-candy and barley-sugar prepared?
MRS. B.
Candied sugar is nothing more than the regular crystals, obtained by
slow evaporation from a solution of sugar. Barley-sugar is sugar melted
by
175
heat, and afterwards cooled in moulds of a spiral form.
Sugar may be decomposed by a red heat, and, like all other vegetable
substances, resolved into carbonic acid and hydrogen. The formation and
the decomposition of sugar afford many very interesting particulars,
which we shall fully examine, after having gone through the other
materials of vegetables. We shall find that there is reason to suppose
that sugar is not, like the other materials, secreted from the sap by
appropriate organs; but that it is formed by a peculiar process with
which you are not yet acquainted.
CAROLINE.
Pray, is not honey of the same nature as sugar?
MRS. B.
Honey is a mixture of saccharine matter and gum.
EMILY.
I thought that honey was in some measure an animal substance, as it
is prepared by the bees.
MRS. B.
It is rather collected by them from flowers, and conveyed to their
store-houses, the hives. It is the wax only that undergoes a real
alteration in
176
the body of the bee, and is thence converted into an animal
substance.
Manna is another kind of sugar, which is united with a nauseous
extractive matter, to which it owes its peculiar taste and colour. It
exudes like gum from various trees in hot climates, some of which have
their leaves glazed by it.
The next of the vegetable materials is fecula; this is the
general name given to the farinaceous substance contained in all seeds,
and in some roots, as the potatoe, parsnip, &c. It is intended by
nature for the first aliment of the young vegetable; but that of one
particular grain is become a favourite and most common food of a large
part of mankind.
EMILY.
You allude, I suppose, to bread, which is made of wheat-flower?
MRS. B.
Yes. The fecula of wheat contains also another vegetable substance
which seems peculiar to that seed, or at least has not as yet been
obtained from any other. This is gluten, which is of a sticky,
ropy, elastic nature; and it is supposed to be owing to the viscous
qualities of this substance, that wheat-flour forms a much better paste
than any other.
177
EMILY.
Gluten, by your description, must be very like gum?
MRS. B.
In their sticky nature they certainly have some resemblance; but
gluten is essentially different from gum in other points, and especially
in its being insoluble in water, whilst gum, you know, is extremely
soluble.
The oils contained in vegetables all consist of hydrogen and
carbon in various proportions. They are of two kinds, fixed and
volatile, both of which we formerly mentioned. Do you remember in
what the difference between fixed and volatile oil consists?
EMILY.
If I recollect rightly, the former are decomposed by heat, whilst the
latter are merely volatilised by it.
MRS. B.
Very well. Fixed oil is contained only in the seeds of plants,
excepting in the olive, in which it is produced in, and expressed from,
the fruit. We have already observed that seeds contain also fecula;
these two substances, united with a little mucilage, form the white
substance contained in the seeds or kernels of plants, and is destined
for the nourishment of the young plant, to which the seed gives birth.
The milk of almonds, which is
178
expressed from the seed of that name, is composed of these three
substances.
EMILY.
Pray, of what nature is the linseed oil which is used in
painting?
MRS. B.
It is a fixed oil, obtained from the seed of flax. Nut oil, which is
frequently used for the same purpose, is expressed from walnuts.
Olive oil is that which is best adapted to culinary purposes.
CAROLINE.
And what are the oils used for burning?
MRS. B.
Animal oils most commonly; but the preference given to them is owing
to their being less expensive; for vegetable oils burn equally well, and
are more pleasant, as their smell is not offensive.
EMILY.
Since oil is so good a combustible, what is the reason that lamps so
frequently require trimming?
MRS. B.
This sometimes proceeds from the construction of the lamp, which may
not be sufficiently favourable to a perfect combustion; but there is
179
certainly a defect in the nature of oil itself, which renders it
necessary for the best-constructed lamps to be occasionally trimmed.
This defect arises from a portion of mucilage which it is extremely
difficult to separate from the oil, and which being a bad combustible,
gathers round the wick, and thus impedes its combustion, and
consequently dims the light.
CAROLINE.
But will not oils burn without a wick?
MRS. B.
Not unless their temperature be elevated to five or six hundred
degrees; the wick answers this purpose, as I think I once before
explained to you. The oil rises between the fibres of the cotton by
capillary attraction, and the heat of the burning wick volatilises it,
and brings it successively to the temperature at which it is
combustible.
EMILY.
I suppose the explanation which you have given with regard to the
necessity of trimming lamps, applies also to candles, which so often
require snuffing?
MRS. B.
I believe it does; at least, in some degree. But besides the
circumstance just explained, the common
180
sorts of oils are not very highly combustible, so that the heat produced
by a candle, which is a coarse kind of animal oil, being insufficient to
volatilise them completely, a quantity of soot is gradually
deposited on the wick, which dims the light, and retards the
combustion.
CAROLINE.
Wax candles then contain no incombustible matter, since they do not
require snuffing?
MRS. B.
Wax is a much better combustible than tallow, but still not perfectly
so, since it likewise contains some particles that are unfit for
burning; but when these gather round the wick, (which in a wax light is
comparatively small,) they weigh it down on one side, and fall off
together with the burnt part of the wick.
CAROLINE.
As oils are such good combustibles, I wonder that they should require
so great an elevation of temperature before they begin to burn?
MRS. B.
Though fixed oils will not enter into actual combustion below the
temperature of about four hundred degrees, yet they will slowly absorb
oxygen
181
at the common temperature of the atmosphere. Hence arises a variety of
changes in oils which modify their properties and uses in the arts.
If oil simply absorbs, and combines with oxygen, it thickens and
changes to a kind of wax. This change is observed to take place on the
external parts of certain vegetables, even during their life. But it
happens in many instances that the oil does not retain all the oxygen
which it attracts, but that part of it combines with, or burns, the
hydrogen of the oil, thus forming a quantity of water, which gradually
goes off by evaporation. In this case the alteration of the oil consists
not only in the addition of a certain quantity of oxygen, but in the
diminution of the hydrogen. These oils are distinguished by the name of
drying oils. Linseed, poppy, and nut-oils, are of this
description.
EMILY.
I am well acquainted with drying oils, as I continually use them in
painting. But I do not understand why the acquisition of oxygen on one
hand, and a loss of hydrogen on the other, should render them
drying?
MRS. B.
This, I conceive, may arise from two reasons; either from the oxygen
which is added being less
182
favourable to the state of fluidity than the hydrogen, which is
subtracted; or from this additional quantity of oxygen giving rise to
new combinations, in consequence of which the most fluid parts of the
oil are liberated and volatilised.
For the purpose of painting, the drying quality of oil is further
increased by adding a quantity of oxyd of lead to it, by which means it
is more rapidly oxygenated.
The rancidity of oil is likewise owing to their oxygenation. In this
case a new order of attraction takes place, from which a peculiar acid
is formed, called the sebacic acid.
CAROLINE.
Since the nature and composition of oil is so well known, pray could
not oil be actually made, by combining its principles?
MRS. B.
That is by no means a necessary consequence; for there are
innumerable varieties of compound bodies which we can decompose,
although we are unable to reunite their ingredients. This, however, is
not the case with oil, as it has very lately been discovered, that it is
possible to form oil, by a peculiar process, from the action of
oxygenated muriatic acid gas on hydro-carbonate.
183
We now pass to the volatile or essential oils. These
form the basis of all the vegetable perfumes, and are contained, more or
less, in every part of the plant excepting the seed; they are, at least,
never found in that part of the seed which contains the embrio
plant.
EMILY.
The smell of flowers, then, proceeds from volatile oil?
MRS. B.
Certainly; but this oil is often most abundant in the rind of fruits,
as in oranges, lemons, &c. from which it may be extracted by
the slightest pressure; it is found also in the leaves of plants, and
even in the wood.
CAROLINE.
Is it not very plentiful in the leaves of mint, and of thyme, and all
the sweet-smelling herbs?
MRS. B.
Yes, remarkably so; and in geranium leaves also, which have a much
more powerful odour than the flowers.
The perfume of sandal fans is an instance of its existence in wood.
In short, all vegetable odours or perfumes are produced by the
evaporation of particles of these volatile oils.
184
EMILY.
They are, I suppose, very light, and of very thin consistence, since
they are so volatile?
MRS. B.
They vary very much in this respect, some of them being as thick as
butter, whilst others are as fluid as water. In order to be prepared for
perfumes, or essences, these oils are first properly purified, and then
either distilled with spirit of wine, as in the case with lavender
water, or simply mixed with a large proportion of water, as is often
done with regard to peppermint. Frequently, also, these odoriferous
waters are prepared merely by soaking the plants in water, and
distilling. The water then comes over impregnated with the volatile
oil.
CAROLINE.
Such waters are frequently used to take spots of grease out of cloth,
or silk; how do they produce that effect?
MRS. B.
By combining with the substance that forms these stains; for volatile
oils, and likewise the spirit in which they are distilled, will dissolve
wax, tallow, spermaceti, and resins; if, therefore, the spot proceeds
from any of these substances, it
185
will remove it. Insects of every kind have a great aversion to perfumes,
so that volatile oils are employed with success in museums for the
preservation of stuffed birds and other species of animals.
CAROLINE.
Pray does not the powerful smell of camphor proceed from a volatile
oil?
MRS. B.
Camphor seems to be a substance of its own kind, remarkable by
many peculiarities. But if not exactly of the same nature as volatile
oil, it is at least very analogous to it. It is obtained chiefly from
the camphor-tree, a species of laurel which grows in China, and in
the Indian isles, from the stem and roots of which it is extracted.
Small quantities have also been distilled from thyme, sage, and other
aromatic plants; and it is deposited in pretty large quantities by some
volatile oils after long standing. It is extremely volatile and
inflammable. It is insoluble in water, but is soluble in oils, in which
state, as well as in its solid form, it is frequently applied to
medicinal purposes. Amongst the particular properties of camphor, there
is one too singular to be passed over in silence. If you take a small
piece of camphor, and place it on the surface of a bason of pure water,
it will immediately begin to move round
186
and round with great rapidity; but if you pour into the bason a single
drop of any odoriferous fluid, it will instantly put a stop to this
motion. You can at any time try this very simple experiment; but you
must not expect that I shall be able to account for this phenomenon, as
nothing satisfactory has yet been advanced for its explanation.
CAROLINE.
It is very singular indeed; and I will certainly try the experiment.
Pray what are resins, which you just now mentioned?
MRS. B.
They are volatile oils, that have been acted on, and peculiarly
modified, by oxygen.
CAROLINE.
They are, therefore, oxygenated volatile oils?
MRS. B.
Not exactly; for the process does not appear to consist so much in
the oxygenation of the oil, as in the combustion of a portion of its
hydrogen, and a small portion of its carbon. For when resins are
artificially made by the combination of volatile oils with oxygen, the
vessel in which the process is performed is bedewed with water, and the
air included within is loaded with carbonic acid.
187
EMILY.
This process must be, in some respects, similar to that for preparing
drying oils?
MRS. B.
Yes; and it is by this operation that both of them acquire a greater
degree of consistence. Pitch, tar, and turpentine, are the most common
resins; they exude from the pine and fir trees. Copal, mastic, and
frankincense, are also of this class of vegetable substances.
EMILY.
Is it of these resins that the mastic and copal varnishes, so much
used in painting, are made?
MRS. B.
Yes. Dissolved either in oil, or in alcohol, resins form varnishes.
From these solutions they may be precipitated by water, in which they
are insoluble. This I can easily show you.—If you will pour some
water into this glass of mastic varnish, it will combine with the
alcohol in which the resin is dissolved, and the latter will be
precipitated in the form of a white cloud—
EMILY.
It is so. And yet how is it that pictures or
188
drawings, varnished with this solution, may safely be washed with
water?
MRS. B.
As the varnish dries, the alcohol evaporates, and the dry varnish or
resin which remains, not being soluble in water, will not be acted on
by it.
There is a class of compound resins called gum-resins, which
are precisely what their name denotes, that is to say, resins combined
with mucilage. Myrrh and assafœtida are of this description.
CAROLINE.
Is it possible that a substance of so disagreeable a smell as
assafœtida can be formed from a volatile oil?
MRS. B.
The odour of volatile oils is by no means always grateful. Onions and
garlic derive their smell from volatile oils, as well as roses and
lavender.
There is still another form under which volatile oils present
themselves, which is that of balsams. These consist of resinous
juices combined with a peculiar acid, called the benzoic acid. Balsams
appear to have been originally volatile oils, the oxygenation of which
has converted one part into a resin, and the other part into an acid,
which,
189
combined together, form a balsam; such are the balsams of Peru,
Tolu, &c.
We shall now take leave of the oils and their various modifications, and
proceed to the next vegetable substance, which is caoutchouc.
This is a white milky glutinous fluid, which acquires consistence, and
blackens in drying, in which state it forms the substance with which you
are so well acquainted, under the name of gum-elastic.
CAROLINE.
I am surprised to hear that gum-elastic was ever white, or ever
fluid! And from what vegetable is it procured?
MRS. B.
It is obtained from two or three different species of trees, in the
East-Indies, and South-America, by making incisions in the stem. The
juice is collected as it trickles from these incisions, and moulds of
clay, in the form of little bottles of gum-elastic, are dipped into it.
A layer of this juice adheres to the clay and dries on it; and
several layers are successively added by repeating this till the bottle
is of sufficient thickness. It is then beaten to break down the clay,
which is easily shaken out. The natives of the countries where this
substance is produced sometimes make shoes and boots of it by a similar
process, and
190
they are said to be extremely pleasant and serviceable, both from their
elasticity, and their being water-proof.
The substance which comes next in our enumeration of the immediate
ingredients of vegetables, is extractive matter. This is a term,
which, in a general sense, may be applied to any substance extracted
from vegetables; but it is more particularly understood to relate to the
extractive colouring matter of plants. A great variety of
colours are prepared from the vegetable kingdom, both for the purposes
of painting and of dying; all the colours called lakes are of
this description; but they are less durable than mineral colours, for,
by long exposure to the atmosphere, they either darken or turn
yellow.
EMILY.
I know that in painting, the lakes are reckoned far less durable
colours than the ochres; but what is the reason of it?
MRS. B.
The change which takes place in vegetable colours is owing chiefly to
the oxygen of the atmosphere slowly burning their hydrogen, and leaving,
in some measure, the blackness of the carbon exposed.
191
Such change cannot take place in ochre, which is altogether a mineral
substance.
Vegetable colours have a stronger affinity for animal than for
vegetable substances, and this is supposed to be owing to a small
quantity of nitrogen which they contain. Thus, silk and worsted will
take a much finer vegetable dye than linen and cotton.
CAROLINE.
Dying, then, is quite a chemical process?
MRS. B.
Undoubtedly. The condition required to form a good dye is, that the
colouring matter should be precipitated, or fixed, on the substance to
be dyed, and should form a compound not soluble in the liquids to which
it will probably be exposed. Thus, for instance, printed or dyed linens
or cottons must be able to resist the action of soap and water, to which
they must necessarily be subject in washing; and woollens and silks
should withstand the action of grease and acids, to which they may
accidentally be exposed.
CAROLINE.
But if linen and cotton have not a sufficient affinity for colouring
matter, how are they made to resist the action of washing, which they
always do when they are well printed?
192
MRS. B.
When the substance to be dyed has either no affinity for the
colouring matter, or not sufficient power to retain it, the combination
is effected, or strengthened, by the intervention of a third substance,
called a mordant, or basis. The mordant must have a strong
affinity both for the colouring matter and the substance to be dyed, by
which means it causes them to combine and adhere together.
CAROLINE.
And what are the substances that perform the office of thus
reconciling the two adverse parties?
MRS. B.
The most common mordant is sulphat of alumine, or alum. Oxyds of tin
and iron, in the state of compound salts, are likewise used for that
purpose.
Tannin is another vegetable ingredient of great importance in
the arts. It is obtained chiefly from the bark of trees; but it is found
also in nut-galls, and in some other vegetables.
EMILY.
Is that the substance commonly called tan, which is used in
hot-houses?
193
MRS. B.
Tan is the prepared bark in which the peculiar substance, tannin, is
contained. But the use of tan in hot-houses is of much less importance
than in the operation of tanning, by which skin is converted into
leather.
EMILY.
Pray, how is this operation performed?
MRS. B.
Various methods are employed for this purpose, which all consist in
exposing skin to the action of tannin, or of substances containing this
principle, in sufficient quantities, and disposed to yield it to the
skin. The most usual way is to infuse coarsely powdered oak bark in
water, and to keep the skin immersed in this infusion for a certain
length of time. During this process, which is slow and gradual, the skin
is found to have increased in weight, and to have acquired a
considerable tenacity and impermeability to water. This effect may be
much accelerated by using strong saturations of the tanning principle
(which can be extracted from bark), instead of employing the bark
itself. But this quick mode of preparation does not appear to make
equally good leather.
Tannin is contained in a great variety of
194
astringent vegetable substances, as galls, the rose-tree, and wine; but
it is nowhere so plentiful as in bark. All these substances yield it to
water, from which it may be precipitated by a solution of isinglass, or
glue, with which it strongly unites and forms an insoluble compound.
Hence its valuable property of combining with skin (which consists
chiefly of glue), and of enabling it to resist the action of water.
EMILY.
Might we not see that effect by pouring a little melted isinglass
into a glass of wine, which you say contains tannin?
MRS. B.
Yes. I have prepared a solution of isinglass for that very
purpose.—Do you observe the thick muddy precipitate?—That is
the tannin combined with the isinglass.
CAROLINE.
This precipitate must then be of the same nature as leather?
MRS. B.
It is composed of the same ingredients; but the organisation and
texture of the skin being wanting, it has neither the consistence nor
the tenacity of leather.
195
CAROLINE.
One might suppose that men who drink large quantities of red wine
stand a chance of having the coats of their stomachs converted into
leather, since tannin has so strong an affinity for skin.
MRS. B.
It is not impossible but that the coats of their stomachs may be, in
some measure, tanned, or hardened by the constant use of this liquor;
but you must remember that where a number of other chemical agents are
concerned, and, above all, where life exists, no certain chemical
inference can be drawn.
I must not dismiss this subject, without mentioning a recent
discovery of Mr. Hatchett, which relates to it. This gentleman found
that a substance very similar to tannin, possessing all its leading
properties, and actually capable of tanning leather, may be produced by
exposing carbon, or any substance containing carbonaceous matter,
whether vegetable, animal, or mineral, to the action of nitric acid.
CAROLINE.
And is not this discovery very likely to be of use to
manufactures?
MRS. B.
That is very doubtful, because tannin, thus artificially
196
prepared, must probably always be more expensive than that which is
obtained from bark. But the fact is extremely curious, as it affords one
of those very rare instances of chemistry being able to imitate the
proximate principles of organised bodies.
The last of the vegetable materials is woody fibre; it is the
hardest part of plants. The chief source from which this substance is
derived is wood, but it is also contained, more or less, in every solid
part of that plant. It forms a kind of skeleton of the part to which it
belongs, and retains its shape after all the other materials have
disappeared. It consists chiefly of carbon, united with a small
proportion of salts, and the other constituents common to all
vegetables.
EMILY.
It is of woody fibre, then, that the common charcoal is made?
MRS. B.
Yes. Charcoal, as you may recollect, is obtained from wood, by the
separation of all its evaporable parts.
Before we take leave of the vegetable materials, it will be proper,
at least, to enumerate the several vegetable acids which we either have
had, or may
197
have occasion to mention. I believe I formerly told you that their
basis, or radical, was uniformly composed of hydrogen and carbon, and
that their difference consisted only in the various proportions of
oxygen which they contained.
The following are the names of the vegetable acids:
| The | Mucous Acid, |
obtained from gum or mucilage; |
| Suberic |
from cork; |
| Camphoric |
from camphor; |
| Benzoic |
from balsams; |
| Gallic |
from galls, bark, &c. |
| Malic |
from ripe fruits; |
| Citric |
from lemon juice; |
| Oxalic |
from sorrel; |
| Succinic |
from amber; |
| Tartarous |
from tartrit of potash: |
| Acetic |
from vinegar. |
They are all decomposable by heat, soluble in water, and turn
vegetable blue colours red. The succinic, the tartarous,
and the acetous acids, are the products of the decomposition of
vegetables; we shall,
therefore, reserve their examination for a future period.
The oxalic acid, distilled from sorrel, is the highest term of
vegetable acidification; for, if
198
more oxygen be added to it, it loses its vegetable nature, and is
resolved into carbonic acid and water; therefore, though all the other
acids may be converted into the oxalic by an addition of oxygen, the
oxalic itself is not susceptible of a further degree of oxygenation; nor
can it be made, by any chemical processes, to return to a state of lower
acidification.
To conclude this subject, I have only to add a few words on the
gallic acid. . . . .
CAROLINE.
Is not this the same acid before mentioned, which forms ink, by
precipitating sulphat of iron from its solution?
MRS. B.
Yes. Though it is usually extracted from galls, on account of its
being most abundant in that vegetable substance, it may also be obtained
from a great variety of plants. It constitutes what is called the
astringent principle of vegetables; it is generally combined with
tannin, and you will find that an infusion of tea, coffee, bark,
red-wine, or any vegetable substance that contains the astringent
principle, will make a black precipitate with a solution of sulphat of
iron.
199
CAROLINE.
But pray what are galls?
MRS. B.
They are excrescences which grow on the bark of young oaks, and are
occasioned by an insect which wounds the bark of trees, and lays its
eggs in the aperture. The lacerated vessels of the tree then discharge
their contents, and form an excrescence, which affords a defensive
covering for these eggs. The insect, when come to life, first feeds on
this excrescence, and some time afterward eats its way out, as it
appears from a hole which is formed in all gall-nuts that no longer
contain an insect. It is in hot climates only that strongly astringent
gall-nuts are found; those which are used for the purpose of making ink
are brought from Aleppo.
EMILY.
But are not the oak-apples, which grow on the leaves of the oak in
this country, of a similar nature?
MRS. B.
Yes; only they are an inferior species of galls, containing less of
the astringent principle, and therefore less applicable to useful
purposes.
200
CAROLINE.
Are the vegetable acids never found but in their pure uncombined
state?
MRS. B.
By no means; on the contrary, they are frequently met with in the
state of compound salts; these, however, are in general not fully
saturated with the salifiable bases, so that the acid predominates; and,
in this state, they are called acidulous salts. Of this kind is
the salt called cream of tartar.
CAROLINE.
Is not the salt of lemon, commonly used to take out ink-spots and
stains, of this nature?
MRS. B.
No; that salt consists of the oxalic acid, combined with a little
potash. It is found in that state in sorrel.
CAROLINE.
And pray how does it take out ink-spots?
MRS. B.
By uniting with the iron, and rendering it soluble in water.
Besides the vegetable materials which we have enumerated, a variety
of other substances, common
201
to the three kingdoms, are found in vegetables, such as potash, which
was formerly supposed to belong exclusively to plants, and was, in
consequence, called the vegetable alkali.
Sulphur, phosphorus, earths, and a variety of metallic oxyds, are
also found in vegetables, but only in small quantities. And we meet
sometimes with neutral salts, formed by the combination of these
ingredients.
202
CONVERSATION XXI.
ON THE DECOMPOSITION OF VEGETABLES.

CAROLINE.
The account which you have given us,
Mrs. B., of the materials of vegetables, is, doubtless, very
instructive; but it does not completely satisfy my curiosity.
I wish to know how plants obtain the principles from which their
various materials are formed; by what means these are converted into
vegetable matter, and how they are connected with the life of the
plant?
MRS. B.
This implies nothing less than a complete history of the chemistry
and physiology of vegetation, subjects on which we have yet but very
imperfect notions. Still I hope that I shall be able, in some measure,
to satisfy your curiosity. But, in order to render the subject more
intelligible, I must first
203
make you acquainted with the various changes which vegetables undergo,
when the vital power no longer enables them to resist the common laws of
chemical attraction.
The composition of vegetables being more complicated than that of
minerals, the former more readily undergo chemical changes than the
latter: for the greater the variety of attractions, the more easily is
the equilibrium destroyed, and a new order of combinations
introduced.
EMILY.
I am surprised that vegetables should be so easily susceptible of
decomposition; for the preservation of the vegetable kingdom is
certainly far more important than that of minerals.
MRS. B.
You must consider, on the other hand, how much more easily the former
is renewed than the latter. The decomposition of the vegetable takes
place only after the death of the plant, which, in the common course of
nature, happens when it has yielded fruit and seeds to propagate its
species. If, instead of thus finishing its career, each plant was to
retain its form and vegetable state, it would become an useless burden
to the earth and its inhabitants. When vegetables, therefore, cease to
be productive, they cease to live, and nature
204
then begins her process of decomposition, in order to resolve them into
their chemical constituents, hydrogen, carbon, and oxygen; those simple
and primitive ingredients, which she keeps in store for all her
combinations.
EMILY.
But since no system of combination can be destroyed, except by the
establishment of another order of attractions, how can the decomposition
of vegetables reduce them to their simple elements?
MRS. B.
It is a very long process, during which a variety of new combinations
are successively established and successively destroyed: but, in each of
these changes, the ingredients of vegetable matter tend to unite in a
more simple order of compounds, till they are at length brought to their
elementary state, or, at least, to their most simple order of
combinations. Thus you will find that vegetables are in the end almost
entirely reduced to water and carbonic acid; the hydrogen and carbon
dividing the oxygen between them, so as to form with it these two
substances. But the variety of intermediate combinations that take place
during the several stages of the decomposition of vegetables, present us
with a new set of compounds, well worthy of our examination.
205
CAROLINE.
How is it possible that vegetables, while putrefying, should produce
any thing worthy of observation?
MRS. B.
They are susceptible of undergoing certain changes before they arrive
at the state of putrefaction, which is the final term of decomposition;
and of these changes we avail ourselves for particular and important
purposes. But, in order to make you understand this subject, which is of
considerable importance, I must explain it more in detail.
The decomposition of vegetables is always attended by a violent
internal motion, produced by the disunion of one order of particles, and
the combination of another. This is called FERMENTATION. There are several periods at which
this process stops, so that a state of rest appears to be restored, and
the new order of compounds fairly established. But, unless means be used
to secure these new combinations in their actual state, their duration
will be but transient, and a new fermentation will take place, by which
the compound last formed will be destroyed;
and another, and less complex order, will succeed.
EMILY.
The fermentations, then, appear to be only the
206
successive steps by which a vegetable descends to its final
dissolution.
MRS. B.
Precisely so. Your definition is perfectly correct.
CAROLINE.
And how many fermentations, or new arrangements, does a vegetable
undergo before it is reduced to its simple ingredients?
MRS. B.
Chemists do not exactly agree in this point; but there are,
I think, four distinct fermentations, or periods, at which the
decomposition of vegetable matter stops and changes its course. But
every kind of vegetable matter is not equally susceptible of undergoing
all these fermentations.
There are likewise several circumstances required to produce
fermentation. Water and a certain degree of heat are both essential to
this process, in order to separate the particles, and thus weaken their
force of cohesion, that the new chemical affinities may be brought into
action.
CAROLINE.
In frozen climates, then, how can the spontaneous decomposition of
vegetables take place?
207
MRS. B.
It certainly cannot; and, accordingly, we find scarcely any vestiges
of vegetation where a constant frost prevails.
CAROLINE.
One would imagine that, on the contrary, such spots would be covered
with vegetables; for, since they cannot be decomposed, their number must
always increase.
MRS. B.
But, my dear, heat and water are quite as essential to the formation
of vegetables, as they are to their decomposition. Besides, it is from
the dead vegetables, reduced to their elementary principles, that the
rising generation is supplied with sustenance. No young plant,
therefore, can grow unless its predecessors contribute both to its
formation and support; and these not only furnish the seed from which
the new plant springs, but likewise the food by which it is
nourished.
CAROLINE.
Under the torrid zone, therefore, where water is never frozen, and
the heat is very great, both the processes of vegetation and of
fermentation must, I suppose, be extremely rapid?
MRS. B.
Not so much as you imagine: for in such climates
208
great part of the
water which it requires for these processes is in an aëriform state,
which is scarcely more conducive either to the growth or formation of
vegetables than that of ice. In those latitudes, therefore, it is only
in low damp situations, sheltered by woods from the sun’s rays, that the
smaller tribes of vegetables can grow and thrive during the dry season,
as dead vegetables seldom retain water enough to produce fermentation,
but are, on the contrary, soon dried up by the heat of the sun, which
enables them to resist that process; so that it is not till the fall of
the autumnal rains (which are very violent in such climates), that
spontaneous fermentation can take place.
The several fermentations derive their names from their principal
products. The first is called the saccharine fermentation,
because its product is sugar.
CAROLINE.
But sugar, you have told us, is found in all vegetables; it cannot,
therefore, be the product of their decomposition.
MRS. B.
It is true that this fermentation is not confined to the
decomposition of vegetables, as it continually takes place during their
life; and, indeed, this circumstance has, till lately, prevented it from
209
being considered as one of the fermentations. But the process appears so
analogous to the other fermentations, and the formation of sugar,
whether in living or dead vegetable matter is so evidently a new
compound, proceeding from the destruction of the previous order of
combinations, and essential to the subsequent fermentations, that it is
now, I believe, generally esteemed the first step, or necessary
preliminary, to decomposition, if not an actual commencement of that
process.
CAROLINE.
I recollect your hinting to us that sugar was supposed not to be
secreted from the sap, in the same manner as mucilage, fecula, oil, and
the other ingredients of vegetables.
MRS. B.
It is rather from these materials, than from the sap itself, that
sugar is formed; and it is developed at particular periods, as you may
observe in fruits, which become sweet in ripening, sometimes even after
they have been gathered. Life, therefore, is not essential to the
formation of sugar, whilst on the contrary, mucilage, fecula, and the
other vegetable materials that are secreted from the sap by appropriate
organs, whose powers immediately depend on the vital principle, cannot
be
210
produced but during the existence of that principle.
EMILY.
The ripening of fruits is, then, their first step to destruction, as
well as their last towards perfection?
MRS. B.
Exactly.—A process analogous to the saccharine fermentation
takes place also during the cooking of certain vegetables. This is the
case with parsnips, carrots, potatoes, &c. in which sweetness is
developed by heat and moisture; and we know that if we carried the
process a little farther, a more complete decomposition would
ensue. The same process takes place also in seeds previous to their
sprouting.
CAROLINE.
How do you reconcile this to your theory, Mrs. B.? Can you
suppose that a decomposition is the necessary precursor of life?
MRS. B.
That is indeed the case. The materials of the seed must be
decomposed, and the seed disorganized, before a plant can sprout from
it. Seeds, besides the embrio plant, contain (as we have already
observed) fecula, oil, and a little mucilage. These substances are
destined for the nourishment of the future plant; but they undergo some
change
211
before they can be fit for this function. The seeds, when buried in the
earth, with a certain degree of moisture and of temperature, absorb
water, which dilates them, separates their particles, and introduces a
new order of attractions, of which sugar is the product. The substance
of the seed is thus softened, sweetened, and converted into a sort of
white milky pulp, fit for the nourishment of the embrio plant.
The saccharine fermentation of seeds is artificially produced, for
the purpose of making malt, by the following
process:—A quantity of barley is first soaked in water for
two or three days: the water being afterwards drained off, the grain
heats spontaneously, swells, bursts, sweetens, shows a disposition to
germinate, and actually sprouts to the length of an inch, when the
process is stopped by putting it into a kiln, where it is well dried at
a gentle heat. In this state it is crisp and friable, and constitutes
the substance called malt, which is the principal ingredient of
beer.
EMILY.
But I hope you will tell us how malt is made into beer?
MRS. B.
Certainly; but I must first explain to you the nature of the second
fermentation, which is essential
212
to that operation. This is called the vinous fermentation,
because its product is wine.
EMILY.
How very different the decomposition of vegetables is from what I had
imagined! The products of their disorganisation appear almost superior
to those which they yield during their state of life and perfection.
MRS. B.
And do you not, at the same time, admire the beautiful economy of
Nature, which, whether she creates, or whether she destroys, directs all
her operations to some useful and benevolent purpose?—It appears
that the saccharine fermentation is extremely favourable, if not
absolutely essential, as a previous step, to the vinous fermentation; so
that if sugar be not developed during the life of the plant, the
saccharine fermentation must be artificially produced before the vinous
fermentation can take place. This is the case with barley, which does
not yield any sugar until it is made into malt; and it is in that state
only that it is susceptible of undergoing the vinous fermentation by
which it is converted into beer.
CAROLINE.
But if the product of the vinous fermentation
213
is always wine, beer cannot have undergone that process, for beer is
certainly not wine.
MRS. B.
Chemically speaking, beer may be considered as the wine of grain. For
it is the product of the fermentation of malt, just as wine is that of
the fermentation of grapes, or other fruits.
The consequence of the vinous fermentation is the decomposition of
the saccharine matter, and the formation of a spirituous liquor from the
constituents of the sugar. But, in order to promote this fermentation,
not only water and a certain degree of heat are necessary, but also some
other vegetable ingredients, besides the sugar, as fecula, mucilage,
acids, salts, extractive matter, &c. all of which seem to contribute
to this process; and give to the liquor its peculiar taste.
EMILY.
It is, perhaps, for this reason that wine is not obtained from the
fermentation of pure sugar; but that fruits are chosen for that purpose,
as they contain not only sugar, but likewise the other vegetable
ingredients which promote the vinous fermentation, and give the peculiar
flavour.
MRS. B.
Certainly. And you must observe also, that
214
the relative quantity of sugar is not the only circumstance to be
considered in the choice of vegetable juices for the formation of wine;
otherwise the sugar-cane would be best adapted for that purpose. It is
rather the manner and proportion in which the sugar is mixed with other
vegetable ingredients that influences the production and qualities of
wine. And it is found that the juice of the grape not only yields the
most considerable proportion of wine, but that it likewise affords it of
the most grateful flavour.
EMILY.
I have seen a vintage in Switzerland, and I do not recollect that
heat was applied, or water added, to produce the fermentation of the
grapes.
MRS. B.
The common temperature of the atmosphere in the cellars in which the
juice of the grape is fermented is sufficiently warm for this purpose;
and as the juice contains an ample supply of water, there is no occasion
for any addition of it. But when fermentation is produced in dry malt,
a quantity of water must necessarily be added.
EMILY.
But what are precisely the changes that happen during the vinous
fermentation?
215
MRS. B.
The sugar is decomposed, and its constituents are recombined into two
new substances; the one a peculiar liquid substance, called
alcohol or spirit of wine, which remains in the fluid; the
other, carbonic acid gas, which escapes during the fermentation. Wine,
therefore, as I before observed, in a general point of view, may be
considered as a liquid of which alcohol constitutes the essential part.
And the varieties of strength and flavour of the different kinds of wine
are to be attributed to the different qualities of the fruits from which
they are obtained, independently of the sugar.
CAROLINE.
I am astonished to hear that so powerful a liquid as spirit of wine
should be obtained from so mild a substance as sugar.
MRS. B.
Can you tell me in what the principal difference consists between
alcohol and sugar?
CAROLINE.
Let me reflect . . . . . Sugar consists of carbon, hydrogen, and
oxygen. If carbonic acid be subtracted from it, during the formation of
alcohol, the latter will contain less carbon and oxygen
216
than sugar does; therefore hydrogen must be the prevailing principle of
alcohol.
MRS. B.
It is exactly so. And this very large proportion of hydrogen accounts
for the lightness and combustible property of alcohol, and of spirits in
general, all of which consist of alcohol variously modified.
EMILY.
And can sugar be recomposed from the combination of alcohol and
carbonic acid?
MRS. B.
Chemists have never been able to succeed in effecting this; but from
analogy, I should suppose such a recomposition possible. Let us now
observe more particularly the phenomena that take place during the
vinous fermentation. At the commencement of this process, heat is
evolved, and the liquor swells considerably from the formation of the
carbonic acid, which is disengaged in such prodigious quantities as
would be fatal to any person who should unawares inspire it; an accident
which has sometimes happened. If the fermentation be stopped by putting
the liquor into barrels, before the whole of the carbonic acid is
evolved, the wine is brisk, like Champagne,
217
from the carbonic acid imprisoned in it, and it tastes sweet, like
cyder, from the sugar not being completely decomposed.
EMILY.
But I do not understand why heat should be evolved during this
operation. For, as there is a considerable formation of gas, in which a
proportionable quantity of heat must become insensible, I should
have imagined that cold, rather than heat, would have been produced.
MRS. B.
It appears so on first consideration; but you must recollect that
fermentation is a complicated chemical process; and that, during the
decompositions and recompositions attending it, a quantity of
chemical heat may be disengaged, sufficient both to develope the gas,
and to effect an increase of temperature. When the fermentation is
completed, the liquid cools and subsides, the effervescence ceases, and
the thick, sweet, sticky juice of the fruit is converted into a clear,
transparent, spirituous liquor, called wine.
EMILY.
How much I regret not having been acquainted with the nature of the
vinous fermentation, when I had an opportunity of seeing the
process!
218
MRS. B.
You have an easy method of satisfying yourself in that respect by
observing the process of brewing, which, in every essential
circumstance, is similar to that of making wine, and is really a very
curious chemical operation.
Although we cannot actually make wine at this moment, it will be easy
to show you the mode of analyzing it. This is done by distillation. When
wine of any kind is submitted to this operation, it is found to contain
brandy, water, tartar, extractive colouring matter, and some vegetable
acids. I have put a little port wine into this alembic of glass (Plate XIV. Fig. 1.), and
on placing the lamp under it, you will soon see the spirit and water
successively come over—
Plate XIV.
Vol. II. p. 213.
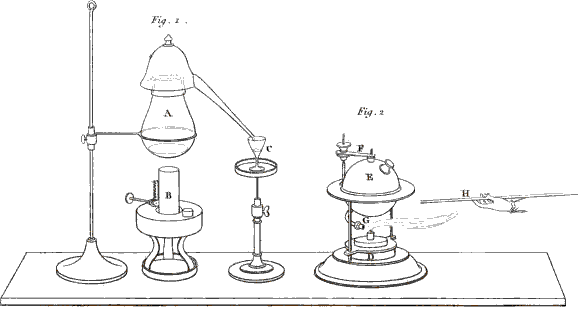
Fig. 1. A Alembic.
B Lamp.
C Wine glass.
Fig. 2. Alcohol blowpipe.
D the Lamp.
E the vessel in which the Alcohol is boiling.
F a safety valve.
G the inflamed jet or steam of alcohol directed towards a glass
tube H.
Larger
view
EMILY.
But you do not mention alcohol amongst the products of the
distillation of wine; and yet that is its most essential ingredient?
MRS. B.
The alcohol is contained in the brandy which is now coming over, and
dropping from the still. Brandy is nothing more than a mixture of
alcohol and water; and in order to obtain the alcohol pure, we must
again distil it from brandy.
219
CAROLINE.
I have just taken a drop on my finger; it tastes like strong brandy,
but it is without colour, whilst brandy is of a deep yellow.
MRS. B.
It is not so naturally; in its pure state brandy is colourless, and
it obtains the yellow tint you observe, by extracting the colouring
matter from the new oaken casks in which it is kept. But if it does not
acquire the usual tinge in this way, it is the custom to colour the
brandy used in this country artificially, with a little burnt sugar, in
order to give it the appearance of having been long kept.
CAROLINE.
And is rum also distilled from wine?
MRS. B.
By no means; it is distilled from the sugar-cane, a plant which
contains so great a quantity of sugar, that it yields more alcohol than
almost any other vegetable. After the juice of the cane has been pressed
out for making sugar, what still remains in the bruised cane is
extracted by water, and this watery solution of sugar is fermented, and
produces rum.
220
The spirituous liquor called arack is in a similar manner
distilled from the product of the vinous fermentation of rice.
EMILY.
But rice has no sweetness; does it contain any sugar?
MRS. B.
Like barley and most other seeds, it is insipid until it has
undergone the saccharine fermentation; and this, you must recollect, is
always a previous step to the vinous fermentation in those vegetables in
which sugar is not already formed. Brandy may in the same manner be
obtained from malt.
CAROLINE.
You mean from beer, I suppose; for the malt must have previously
undergone the vinous fermentation.
MRS. B.
Beer is not precisely the product of the vinous fermentation of malt.
For hops are a necessary ingredient for the formation of that liquor;
whilst brandy is distilled from pure fermented malt. But brandy might,
no doubt, be distilled from beer as well as from any other liquor that
has undergone the vinous fermentation; for since the basis of brandy is
alcohol, it may be obtained from any liquid that contains that
spirituous substance.
221
EMILY.
And pray, from what vegetable is the favourite spirit of the lower
orders of people, gin, extracted?
MRS. B.
The spirit (which is the same in all fermented liquors) may be
obtained from any kind of grain; but the peculiar flavour which
distinguishes gin is that of juniper berries, which are distilled
together with the grain—
I think the brandy contained in the wine which we are distilling
must, by this time, be all come over. Yes—taste the liquid that is
now dropping from the alembic—
CAROLINE.
It is perfectly insipid, like water.
MRS. B.
It is water, which, as I was telling you, is the second product of
wine, and comes over after all the spirit, which is the lightest part,
is distilled.—The tartar and extractive colouring matter we shall
find in a solid form at the bottom of the alembic.
EMILY.
They look very like the lees of wine.
MRS. B.
And in many respects they are of a similar nature;
222
for lees of wine consist chiefly of tartrit of potash; a salt which
exists in the juice of the grape, and in many other vegetables, and is
developed only by the vinous fermentation. During this operation it is
precipitated, and deposits itself on the internal surface of the cask in
which the wine is contained. It is much used in medicine, and in various
arts, particularly dying, under the name of cream of tartar, and
it is from this salt that the tartarous acid is obtained.
CAROLINE.
But the medicinal cream of tartar is in appearance quite different
from these dark-coloured dregs; it is perfectly colourless.
MRS. B.
Because it consists of the pure salts only, in its crystallised form;
whilst in the instance before us it is mixed with the deep-coloured
extractive matter, and other foreign ingredients.
EMILY.
Pray cannot we now obtain pure alcohol from the brandy which we have
distilled?
MRS. B.
We might; but the process would be tedious: for in order to obtain
alcohol perfectly free from
223
water, it is necessary to distil, or, as the distillers call it,
rectify it several times. You must therefore allow me to produce
a bottle of alcohol that has been thus purified. This is a very
important ingredient, which has many striking properties, besides its
forming the basis of all spirituous liquors.
EMILY.
It is alcohol, I suppose, that produces intoxication?
MRS. B.
Certainly; but the stimulus and momentary energy it gives to the
system, and the intoxication it occasions when taken in excess, are
circumstances not yet accounted for.
CAROLINE.
I thought that it produced these effects by increasing the rapidity
of the circulation of the blood; for drinking wine or spirits,
I have heard, always quickens the pulse.
MRS. B.
No doubt; the spirit, by stimulating the nerves, increases the action
of the muscles; and the heart, which is one of the strongest muscular
organs, beats with augmented vigour, and propels the blood with
accelerated quickness. After such a
224
strong excitation the frame naturally suffers a proportional degree of
depression, so that a state of debility and languor is the invariable consequence of
intoxication. But though these circumstances are well ascertained, they
are far from explaining why alcohol should produce such effects.
EMILY.
Liqueurs are the only kind of spirits which I think pleasant. Pray of
what do they consist?
MRS. B.
They are composed of alcohol, sweetened with syrup, and flavoured
with volatile oil.
The different kinds of odoriferous spirituous waters are likewise
solutions of volatile oil in alcohol, as lavender water, eau de
Cologne, &c.
The chemical properties of alcohol are important and numerous. It is
one of the most powerful chemical agents, and is particularly useful in
dissolving a variety of substances, which are soluble neither by water
nor heat.
EMILY.
We have seen it dissolve copal and mastic to form varnishes; and
these resins are certainly not soluble in water, since water
precipitates them from their solution in alcohol.
225
MRS. B.
I am happy to find that you recollect these circumstances so well.
The same experiment affords also an instance of another property of
alcohol,—its tendency to unite with water; for the resin is
precipitated in consequence of losing the alcohol, which abandons it
from its preference for water. It is attended also, as you may
recollect, with the same peculiar circumstance of a disengagement of
heat and consequent diminution of bulk, which we have supposed to be
produced by a mechanical penetration of particles by which latent heat
is forced out.
Alcohol unites thus readily not only with resins and with water, but
with oils and balsams; these compounds form the extensive class of
elixirs, tinctures, quintessences, &c.
EMILY.
I suppose that alcohol must be highly combustible, since it contains
so large a proportion of hydrogen?
MRS. B.
Extremely so; and it will burn at a very moderate temperature.
CAROLINE.
I have often seen both brandy and spirit of
226
wine burnt; they produce a great deal of flame, but not a proportional
quantity of heat, and no smoke whatever.
MRS. B.
The last circumstance arises from their combustion being complete;
and the disproportion between the flame and heat shows you that these
are by no means synonymous.
The great quantity of flame proceeds from the combustion of the
hydrogen to which, you know, that manner of burning is
peculiar.—Have you not remarked also that brandy and alcohol will
burn without a wick?—They take fire at so low a temperature, that
this assistance is not required to concentrate the heat and volatilise
the fluid.
CAROLINE.
I have sometimes seen brandy burnt by merely heating it in a
spoon.
MRS. B.
The rapidity of the combustion of alcohol may, however, be
prodigiously increased by first volatilising it. An ingenious instrument
has been constructed on this principle to answer the purpose of a
blow-pipe, which may be used for melting glass, or other chemical
purposes. It consists of a small metallic vessel (Plate XIV. Fig. 2.), of a
227
spherical shape, which contains the alcohol, and is heated by the lamp
beneath it; as soon as the alcohol is volatilised, it passes through the
spout of the vessel, and issues just above the wick of the lamp, which
immediately sets fire to the stream of vapour, as I shall show
you—
EMILY.
With what amazing violence it burns! The flame of alcohol, in the
state of vapour, is, I fancy, much hotter than when the spirit is
merely burnt in a spoon?
MRS. B.
Yes; because in this way the combustion goes on much quicker, and, of
course, the heat is proportionally increased.—Observe its effect
on this small glass tube, the middle of which I present to the extremity
of the flame, where the heat is greatest.
CAROLINE.
The glass, in that spot, is become red hot, and bends from its own
weight.
MRS. B.
I have now drawn it asunder, and am going to blow a ball at one of
the heated ends; but I must previously close it up, and flatten it with
this little metallic instrument, otherwise the breath would
228
pass through the tube without dilating any part of it.—Now,
Caroline, will you blow strongly into the tube whilst the closed end is
red hot.
EMILY.
You blowed too hard; for the ball suddenly dilated to a great size,
and then burst in pieces.
MRS. B.
You will be more expert another time; but I must caution you, should
you ever use this blow-pipe, to be very careful that the combustion of
the alcohol does not go on with too great violence, for I have seen the
flame sometimes dart out with such force as to reach the opposite wall
of the room, and set the paint on fire. There is, however, no danger of
the vessel bursting, as it is provided with a safety tube, which affords
an additional vent for the vapour of alcohol when required.
The products of the combustion of alcohol consist in a great
proportion of water, and a small quantity of carbonic acid. There is no
smoke or fixed remains whatever.—How do you account for that,
Emily?
EMILY.
I suppose that the oxygen which the alcohol absorbs in burning,
converts its hydrogen into water
229
and its carbon into carbonic acid gas, and thus it is completely
consumed.
MRS. B.
Very well.—Ether, the lightest of all fluids, and with
which you are well acquainted, is obtained from alcohol, of which it
forms the lightest and most volatile part.
EMILY.
Ether, then, is to alcohol, what alcohol is to brandy?
MRS. B.
No: there is an essential difference. In order to obtain alcohol from
brandy, you need only deprive the latter of its water; but for the
formation of ether, the alcohol must be decomposed, and one of its
constituents partly subtracted. I leave you to guess which of them
it is—
EMILY.
It cannot be hydrogen, as ether is more volatile than alcohol, and
hydrogen is the lightest of all its ingredients: nor do I suppose that
it can be oxygen, as alcohol contains so small a proportion of that
principle; it is, therefore, most probably, carbon, a diminution of
which would not fail to render the new compound more volatile.
230
MRS. B.
You are perfectly right. The formation of ether consists simply in
subtracting from the alcohol a certain proportion of carbon; this is
effected by the action of the sulphuric, nitric, or muriatic acids, on
alcohol. The acid and carbon remain at the bottom of the vessel, whilst
the decarbonised alcohol flies off in the form of a condensable vapour,
which is ether.
Ether is the most inflammable of all fluids, and burns at so slow a temperature
that the heat evolved during its combustion is more than is required for
its support, so that a quantity of ether is volatilised, which takes
fire, and gradually increases the violence of the combustion.
Sir Humphry Davy has lately discovered a very singular fact respecting
the vapour of ether. If a few drops of ether be poured into a
wine-glass, and a fine platina wire, heated almost to redness, be held
suspended in the glass, close to the surface of the ether, the wire soon
becomes intensely red-hot, and remains so for any length of time. We may
easily try the experiment. . . . .
CAROLINE.
How very curious! The wire is almost white hot, and a pungent smell
rises from the glass. Pray how is this accounted for?
231
MRS. B.
This is owing to a very peculiar property of the vapour of ether, and
indeed of many other combustible gaseous bodies. At a certain
temperature lower than that of ignition, these vapours undergo a slow
and imperfect combustion, which does not give rise, in any sensible
degree, to the phenomena of light and flame, and yet extricates a
quantity of caloric sufficient to react upon the wire and make it
red-hot, and the wire in its turn keeps up the effect as long as the
emission of vapour continues.
CAROLINE.
But why should not an iron or silver wire produce the same
effect?
MRS. B.
Because either iron or silver, being much better conductors of heat
than platina, the heat is carried off too fast by those metals to allow
the accumulation of caloric necessary to produce the effect in
question.
Ether is so light that it evaporates at the common temperature of the
atmosphere; it is therefore necessary to keep it confined by a well
ground glass stopper. No degree of cold known has ever
frozen it.
CAROLINE.
Is it not often taken medicinally?
232
MRS. B.
Yes; it is one of the most effectual antispasmodic medicines, and the
quickness of its effects, as such, probably depends on its being
instantly converted into vapour by the heat of the stomach, through the
intervention of which it acts on the nervous system. But the frequent
use of ether, like that of spirituous liquors, becomes prejudicial, and,
if taken to excess, it produces effects similar to those of
intoxication.
We may now take our leave of the vinous fermentation, of which,
I hope, you have acquired a clear idea; as well as of the several
products that are derived from it.
CAROLINE.
Though this process appears, at first sight, so much complicated, it
may, I think, be summed up in a few words, as it consists in the
conversion of sugar and fermentable bodies into alcohol and carbonic
acid, which give rise both to the formation of wine, and of all kinds of
spirituous liquors.
MRS. B.
We shall now proceed to the acetous fermentation, which is
thus called, because it converts wine into vinegar, by the formation of
the acetous acid, which is the basis or radical of vinegar.
233
CAROLINE.
But is not the acidifying principle of the acetous acid the same as
that of all other acids, oxygen?
MRS. B.
Certainly; and on that account the contact of air is essential to
this fermentation, as it affords the necessary supply of oxygen.
Vinegar, in order to obtain pure acetous acid from it, must be distilled
and rectified by certain processes.
EMILY.
But pray, Mrs. B., is not the acetous acid frequently formed without
this fermentation taking place? Is it not, for instance, contained in
acid fruits, and in every substance that becomes sour?
MRS. B.
No, not in fruits; you confound it with the citric, the malic, the
oxalic, and other vegetable acids, to which living vegetables owe their
acidity. But whenever a vegetable substance turns sour, after it has
ceased to live, the acetous acid is developed by means of the acetous
fermentation, in which the substance advances a step towards its final
decomposition.
Amongst the various instances of acetous fermentation, that of bread
is usually classed.
234
CAROLINE.
But the fermentation of bread is produced by yeast; how does that
effect it?
MRS. B.
It is found by experience that any substance that has already
undergone a fermentation, will readily excite it in one that is
susceptible of that process. If, for instance, you mix a little vinegar
with wine, that is intended to be acidified, it will absorb oxygen more
rapidly, and the process be completed much sooner, than if left to
ferment spontaneously. Thus yeast, which is a product of the
fermentation of beer, is used to excite and accelerate the fermentation
of malt, which is to be converted into beer, as well as that of paste
which is to be made into bread.
CAROLINE.
But if bread undergoes the acetous fermentation, why is it not
sour?
MRS. B.
It acquires a certain savour which corrects the heavy insipidity of
flour, and may be reckoned a first degree of acidification; or if the
process were carried further, the bread would become decidedly acid.
There are, however, some chemists who do not
235
consider the fermentation of bread as being of the acetous kind, but
suppose that it is a process of fermentation peculiar to that
substance.
The putrid fermentation is the final operation of Nature, and
her last step towards reducing organised bodies to their simplest
combinations. All vegetables spontaneously undergo this fermentation
after death, provided there be a sufficient degree of heat and moisture,
together with access of air; for it is well known that dead plants may
be preserved by drying, or by the total exclusion of air.
CAROLINE.
But do dead plants undergo the other fermentation previous to this
last; or do they immediately suffer the putrid fermentation?
MRS. B.
That depends on a variety of circumstances, such as the degrees of
temperature and of moisture, the nature of the plant itself, &c. But
if you were carefully to follow and examine the decomposition of plants
from their death to their final dissolution, you would generally find a
sweetness developed in the seeds, and a spirituous flavour in the fruits
(which have undergone the saccharine fermentation), previous to the
total disorganisation and separation of the parts.
236
EMILY.
I have sometimes remarked a kind of spirituous taste in fruits that
were over ripe, especially oranges; and this was just before they became
rotten.
MRS. B.
It was then the vinous fermentation which had succeeded the
saccharine, and had you followed up these changes attentively, you would
probably have found the spirituous taste followed by acidity, previous
to the fruit passing to the state of putrefaction.
When the leaves fall from the trees in autumn, they do not (if there
is no great moisture in the atmosphere) immediately undergo a
decomposition, but are first dried and withered; as soon, however, as
the rain sets in, fermentation commences, their gaseous products are
imperceptibly evolved into the atmosphere, and their fixed remains mixed
with their kindred earth.
Wood, when exposed to moisture, also undergoes the putrid
fermentation and becomes rotten.
EMILY.
But I have heard that the dry rot, which is so liable to
destroy the beams of houses, is prevented by a current of air; and yet
you said that air was essential to the putrid fermentation?
237
MRS. B.
True; but it must not be in such a proportion to the moisture as to
dissolve the latter, and this is generally the case when the rotting of
wood is prevented or stopped by the free access of air. What is commonly
called dry rot, however, is not I believe a true process of
putrefaction. It is supposed to depend on a peculiar kind of vegetation,
which, by feeding on the wood, gradually destroys it.
Straw and all other kinds of vegetable matter undergo the putrid
fermentation more rapidly when mixed with animal matter. Much heat is
evolved during this process, and a variety of volatile products are
disengaged, as carbonic acid and hydrogen gas, the latter of which is
frequently either sulphurated or phosphorated.—When all these
gases have been evolved, the fixed products, consisting of carbon,
salts, potash, &c. form a kind of vegetable earth, which makes very
fine manure, as it is composed of those elements which form the
immediate materials of plants.
CAROLINE.
Pray are not vegetables sometimes preserved from decomposition by
petrification? I have seen very curious specimens of petrified
vegetables, in which state they perfectly preserve their form
238
and organisation, though in appearance they are changed to stone.
MRS. B.
That is a kind of metamorphosis, which, now that you are tolerably
well versed in the history of mineral and vegetable substances,
I leave to your judgment to explain. Do you imagine that vegetables
can be converted into stone?
EMILY.
No, certainly; but they might perhaps be changed to a substance in
appearance resembling stone.
MRS. B.
It is not so, however, with the substances that are called petrified
vegetables; for these are really stone, and generally of the hardest
kind, consisting chiefly of silex. The case is this: when a vegetable is
buried under water, or in wet earth, it is slowly and gradually
decomposed. As each successive particle of the vegetable is destroyed,
its place is supplied by a particle of siliceous earth, conveyed thither
by the water. In the course of time the vegetable is entirely destroyed,
but the silex has completely replaced it, having assumed its form and
apparent texture, as if the vegetable itself were changed to stone.
239
CAROLINE.
That is very curious! and I suppose that petrified animal substances
are of the same nature?
MRS. B.
Precisely. It is equally impossible for either animal or vegetable
substances to be converted into stone. They may be reduced, as we find
they are, by decomposition, to their constituent elements, but cannot be
changed to elements, which do not enter into their composition.
There are, however, circumstances which frequently prevent the
regular and final decomposition of vegetables; as, for instance, when
they are buried either in the sea, or in the earth, where they cannot
undergo the putrid fermentation for want of air. In these cases they are
subject to a peculiar change, by which they are converted into a new
class of compounds, called bitumens.
CAROLINE.
These are substances I never heard of before.
MRS. B.
You will find, however, that some of them are very familiar to you.
Bitumens are vegetables so far decomposed as to retain no organic
appearance; but their origin is easily detected by their oily nature,
their combustibility, the products of
240
their analysis, and the impressions of the forms of leaves, grains,
fibres of wood, and even of animals, which they frequently bear.
They are sometimes of an oily liquid consistence, as the substance
called naptha, in which we preserved potassium; it is a fine
transparent colourless fluid, that issues out of clays in some parts of
Persia. But more frequently bitumens are solid, as asphaltum,
a smooth, hard, brittle substance, which easily melts, and forms,
in its liquid state, a beautiful dark brown colour for oil
painting. Jet, which is of a still harder texture, is a peculiar
bitumen, susceptible of so fine a polish, that it is used for many
ornamental purposes.
Coal is also a bituminous substance, to the composition of which
both the mineral and animal kingdoms seem to concur. This most useful
mineral appears to consist chiefly of vegetable matter, mixed with the
remains of marine animals and marine salts, and occasionally containing
a quantity of sulphuret of iron, commonly called pyrites.
EMILY.
It is, I suppose, the earthly, the metallic, and the saline parts of
coals, that compose the cinders or fixed products of their combustion;
whilst the
241
hydrogen and carbon, which they derive from vegetables, constitute their
volatile products.
CAROLINE.
Pray is not coke, (which I have heard is much used in some
manufactures,) also a bituminous substance?
MRS. B.
No; it is a kind of fuel artificially prepared from coals. It
consists of coals reduced to a substance analogous to charcoal, by the
evaporation of their bituminous parts. Coke, therefore, is composed of
carbon, with some earthy and saline ingredients.
Succin, or yellow amber, is a bitumen which the
ancients called electrum, from whence the word electricity is
derived, as that substance is peculiarly, and was once supposed to be
exclusively, electric. It is found either deeply buried in the bowels of
the earth, or floating on the sea, and is supposed to be a resinous body
which has been acted on by sulphuric acid, as its analysis shows it to
consist of ah oil and an acid. The oil is called oil of amber,
the acid the succinic.
EMILY.
That oil I have sometimes used in painting, as it is reckoned to
change less than the other kinds of oils.
242
MRS. B.
The last class of vegetable substances that have changed their nature
are fossil-wood, peat, and turf. These are composed
of wood and roots of shrubs, that are partly decomposed by being exposed
to moisture under ground, and yet, in some measure, preserve their form
and organic appearance. The peat, or black earth of the moors, retains
but few vestiges of the roots to which it owes its richness and
combustibility, these substances being in the course of time reduced to
the state of vegetable earth. But in turf the roots of plants are still
discernible, and it equally answers the purpose of fuel. It is the
combustible used by the poor in heathy countries, which supply it
abundantly.
It is too late this morning to enter upon the history of vegetation.
We shall reserve this subject, therefore, for our next interview, when I
expect that it will furnish us with ample matter for another
conversation.
243

MRS. B.
The VEGETABLE
KINGDOM may be considered as the link which unites the mineral
and animal creation into one common chain of beings; for it is through
the means of vegetation alone that mineral substances are introduced
into the animal system, since, generally speaking, it is from vegetables
that all animals ultimately derive their sustenance.
CAROLINE.
I do not understand that; the human species subsists as much on
animal as on vegetable food, and there are some carnivorous animals that
will eat only animal food.
MRS. B.
That is true; but you do not consider that those that live on animal
food, derive their sustenance equally, though not so immediately, from
244
vegetables. The meat that we eat is formed from the herbs of the field,
and the prey of carnivorous animals proceeds, either directly or
indirectly, from the same source. It is, therefore, through this channel
that the simple elements become a part of the animal frame. We should in
vain attempt to derive nourishment from carbon, hydrogen, and oxygen,
either in their separate state, or combined in the mineral kingdom; for
it is only by being united in the form of vegetable combination, that
they become capable of conveying nourishment.
EMILY.
Vegetation, then, seems to be the method which Nature employs to
prepare the food of animals?
MRS. B.
That is certainly its principal object. The vegetable creation does
not exhibit more wisdom in that admirable system of organisation, by
which it is enabled to answer its own immediate ends of preservation,
nutrition, and propagation, than in its grand and ultimate object of
forming those arrangements and combinations of principles, which are so
well adapted for the nourishment of animals.
EMILY.
But I am very curious to know whence vegetables
245
obtain those principles which form their immediate materials?
MRS. B.
This is a point on which we are yet so much in the dark, that I
cannot hope fully to satisfy your curiosity; but what little I know on
this subject, I will endeavour to explain to you.
The soil, which, at first view, appears to be the aliment of
vegetables, is found, on a closer investigation, to be little more than
the channel through which they receive their nourishment; so that it is
very possible to rear plants without any earth or soil.
CAROLINE.
Of that we have an instance in the hyacinth and other bulbous roots,
which will grow and blossom beautifully in glasses of water. But I
confess I should think it would be difficult to rear trees in a similar
manner.
MRS. B.
No doubt it would, as it is the burying of the roots in the earth
that supports the stem of the tree. But this office, besides that of
affording a vehicle for food, is far the most important part which the
earthy portion of the soil performs in the process of vegetation; for we
can discover, by
246
analysis, but an extremely small proportion of earth in vegetable
compounds.
CAROLINE.
But if earths do not afford nourishment, why is it necessary to be so
attentive to the preparation of the soil?
MRS. B.
In order to impart to it those qualities which render it a proper
vehicle for the food of the plant. Water is the chief nourishment of
vegetables; if, therefore, the soil be too sandy, it will not retain a
quantity of water sufficient to supply the roots of the plants. If, on
the contrary, it abound too much with clay, the water will lodge in such
quantities as to threaten a decomposition of the roots. Calcareous soils
are, upon the whole, the most favourable to the growth of plants: soils
are, therefore, usually improved by chalk, which, you may recollect, is
a carbonat of lime. Different vegetables, however, require different
kinds of soils. Thus rice demands a moist retentive soil; potatoes a
soft sandy soil; wheat a firm and rich soil. Forest trees grow better in
fine sand than in a stiff clay; and a light ferruginous soil is best
suited to fruit-trees.
CAROLINE.
But pray what is the use of manuring the soil?
247
MRS. B.
Manure consists of all kinds of substances, whether of vegetable or
animal origin, which have undergone the putrid fermentation, and are
consequently decomposed, or nearly so, into their elementary principles.
And it is requisite that these vegetable matters should be in a state of
decay, or approaching decomposition. The addition of calcareous earth,
in the state of chalk or lime, is beneficial to such soils, as it
accelerates the dissolution of vegetable bodies. Now, I ask you,
what is the utility of supplying the soil with these decomposed
substances?
CAROLINE.
It is, I suppose, in order to furnish vegetables with the principles
which enter into their composition. For manures not only contain carbon,
hydrogen, and oxygen, but by their decomposition supply the soil with
these principles in their elementary form.
MRS. B.
Undoubtedly; and it is for this reason that the finest crops are
produced in fields that were formerly covered with woods, because their
soil is composed of a rich mould, a kind of vegetable earth, which
abounds in those principles.
EMILY.
This accounts for the plentifulness of the crops
248
produced in America, where the country was but a few years since covered
with wood.
CAROLINE.
But how is it that animal substances are reckoned to produce the best
manure? Does it not appear much more natural that the decomposed
elements of vegetables should be the most appropriate to the formation
of new vegetables?
MRS. B.
The addition of a much greater proportion of nitrogen, which
constitutes the chief difference between animal and vegetable matter,
renders the composition of the former more complicated, and consequently
more favourable to decomposition. The use of animal substances is
chiefly to give the first impulse to the fermentation of the vegetable
ingredients that enter into the composition of manures. The manure of a
farm-yard is of that description; but there is scarcely any substance
susceptible of undergoing the putrid fermentation that will not make
good manure. The heat produced by the fermentation of manure is another
circumstance which is extremely favourable to vegetation; yet this heat
would be too great if the manure was laid on the ground during the
height of fermentation; it is used in this state only for hot-beds, to
produce melons, cucumbers,
249
and such vegetables as require a very high temperature.
CAROLINE.
A difficulty has just occurred to me which I do not know how to
remove. Since all organised bodies are, in the common course of nature,
ultimately reduced to their elementary state, they must necessarily in
that state enrich the soil, and afford food for vegetation. How is it,
then, that agriculture, which cannot increase the quantity of those
elements that are required to manure the earth, can increase its produce
so wonderfully as is found to be the case in all cultivated
countries?
MRS. B.
It is by suffering none of these decaying bodies to be dissipated,
but in applying them duly to the soil. It is by a judicious preparation
of the soil, which consists in fitting it either for the general
purposes of vegetation, or for that of the particular seed which is to
be sown. Thus, if the soil be too wet, it may be drained; if too loose
and sandy, it may be rendered more consistent and retentive of water by
the addition of clay or loam; it may be enriched by chalk, or any kind
of calcareous earth. On soils thus improved, manures will act with
double efficacy, and if attention be paid to spread them on the ground
at a proper season of the year, to mix them with
250
the soil so that they may be generally diffused through it, to destroy
the weeds which might appropriate these nutritive principles to their
own use, to remove the stones which would impede the growth of the
plant, &c. we may obtain a produce an hundred fold more abundant
than the earth would spontaneously supply.
EMILY.
We have a very striking instance of this in the scanty produce of
uncultivated commons, compared to the rich crops of meadows which are
occasionally manured.
CAROLINE.
But, Mrs. B., though experience daily proves the advantage of
cultivation, there is still a difficulty which I cannot get over.
A certain quantity of elementary principles exist in nature, which
it is not in the power of man either to augment or diminish. Of these
principles you have taught us that both the animal and vegetable
creation are composed. Now the more of them is taken up by the vegetable
kingdom, the less, it would seem, will remain for animals; and,
therefore, the more populous the earth becomes, the less it will
produce.
MRS. B.
Your reasoning is very plausible; but experience
251
every where contradicts the inference you would draw from it; for we
find that the animal and vegetable kingdoms, instead of thriving, as you
would suppose, at each other’s expense, always increase and multiply
together. For you should recollect that animals can derive the elements
of which they are formed only through the medium of vegetables. And you
must allow that your conclusion would be valid only if every particle of
the several principles that could possibly be spared from other purposes
were employed in the animal and vegetable creations. Now we have reason
to believe that a much greater proportion of these principles than is
required for such purposes remains either in an elementary state, or
engaged in a less useful mode of combination in the mineral kingdom.
Possessed of such immense resources as the atmosphere and the waters
afford us, for oxygen, hydrogen, and carbon, so far from being in danger
of working up all our simple materials, we cannot suppose that we shall
ever bring agriculture to such a degree of perfection as to require the
whole of what these resources could supply.
Nature, however, in thus furnishing us with an inexhaustible stock of
raw materials, leaves it in some measure to the ingenuity of man to
appropriate them to its own purposes. But, like a kind parent, she
stimulates him to exertion, by setting
252
the example and pointing out the way. For it is on the operations of
nature that all the improvements of art are founded. The art of
agriculture consists, therefore, in discovering the readiest method of
obtaining the several principles, either from their grand sources, air
and water, or from the decomposition of organised bodies; and in
appropriating them in the best manner to the purposes of vegetation.
EMILY.
But, among the sources of nutritive principles, I am surprised
that you do not mention the earth itself, as it contains abundance of
coals, which are chiefly composed of carbon.
MRS. B.
Though coals abound in carbon, they cannot, on account of their
hardness and impermeable texture, be immediately subservient to the
purposes of vegetation.
EMILY.
No; but by their combustion carbonic acid is produced; and this
entering into various combinations on the surface of the earth, may,
perhaps, assist in promoting vegetation.
MRS. B.
Probably it may in some degree; but at any
253
rate the quantity of nourishment which vegetables may derive from that
source can be but very trifling, and must entirely depend on local
circumstances.
CAROLINE.
Perhaps the smoky atmosphere of London is the cause of vegetation
being so forward and so rich in its vicinity?
MRS. B.
I rather believe that this circumstance proceeds from the very ample
supply of manure, assisted, perhaps, by the warmth and shelter which the
town affords. Far from attributing any good to the smoky atmosphere of
London, I confess I like to anticipate the time when we shall have
made such progress in the art of managing combustion, that every
particle of carbon will be consumed, and the smoke destroyed at the
moment of its production. We may then expect to have the satisfaction of
seeing the atmosphere of London as clear as that of the
country.—But to return to our subject: I hope that you are
now convinced that we shall not easily experience a deficiency of
nutritive elements to fertilise the earth, and that, provided we are but
industrious in applying them to the best advantage by improving the art
of agriculture, no limits can be assigned to the fruits that we may
expect to reap from our labours.
254
CAROLINE.
Yes; I am perfectly satisfied in that respect, and I can assure you
that I feel already much more interested in the progress and improvement
of agriculture.
EMILY.
I have frequently thought that the culture of the land was not
considered as a concern of sufficient importance. Manufactures always
take the lead; and health and innocence are frequently sacrificed to the
prospect of a more profitable employment. It has often grieved me to see
the poor manufacturers crowded together in close rooms, and confined for
the whole day to the most uniform and sedentary employment, instead of
being engaged in that innocent and salutary kind of labour, which Nature
seems to have assigned to man for the immediate acquirement of comfort,
and for the preservation of his existence. I am sure that you agree
with me in thinking so, Mrs. B.?
MRS. B.
I am entirely of your opinion, my dear, in regard to the importance
of agriculture; but as the conveniences of life, which we are all
enjoying, are not derived merely from the soil, I am far from
wishing to depreciate manufactures. Besides, as the labour of one man is
sufficient to produce food for several, those whose industry is not
255
required in tillage must do something in return for the food that is
provided for them. They exchange, consequently, the accommodations for
the necessaries of life. Thus the carpenter and the weaver lodge and
clothe the peasant, who supplies them with their daily bread. The
greater stock of provisions, therefore, which the husbandman produces,
the greater is the quantity of accommodation which the artificer
prepares. Such are the happy effects which naturally result from
civilised society. It would be wiser, therefore, to endeavour to improve
the situation of those who are engaged in manufactures, than to indulge
in vain declamations on the hardships to which they are too frequently
exposed.
But we must not yet take our leave of the subject of agriculture; we
have prepared the soil, it remains for us now to sow the seed. In this
operation we must be careful not to bury it too deep in the ground, as
the access of air is absolutely necessary to its germination; the earth
must, therefore, lie loose and light over it, in order that the air may
penetrate. Hence the use of ploughing and digging, harrowing and raking,
&c. A certain degree of heat and moisture, such as usually
takes place in the spring, is likewise necessary.
CAROLINE.
One would imagine you were going to describe
256
the decomposition of an old plant, rather than the formation of a new
one; for you have enumerated all the requisites of fermentation.
MRS. B.
Do you forget, my dear, that the young plant derives its existence
from the destruction of the seed, and that it is actually by the
saccharine fermentation that the latter is decomposed?
CAROLINE.
True; I wonder that I did not recollect that. The temperature and
moisture required for the germination of the seed is then employed in
producing the saccharine fermentation within it?
MRS. B.
Certainly. But, in order to understand the nature of germination, you
should be acquainted with the different parts of which the seed is
composed. The external covering or envelope contains, besides the germ
of the future plant, the substance which is to constitute its first
nourishment; this substance, which is called the parenchyma,
consists of fecula, mucilage, and oil, as we formerly observed.
The seed is generally divided into two compartments, called
lobes, or cotyledons, as is exemplified by this bean (Plate XV.
Fig. 1.)—the dark-coloured
257
kind of string which divides the lobes is called the radicle, as
it forms the root of the plant, and it is from a contiguous substance,
called plumula, which is enclosed within the lobes, that the stem
arises. The figure and size of the seed depend very much upon the
cotyledons; these vary in number in different seeds; some have only one,
as wheat, oats, barley, and all the grasses; some have three, others
six. But most seeds, as, for instance, all the varieties of beans, have
two cotyledons. When the seed is buried in the earth, at any temperature
above 40 degrees, it imbibes water, which softens and swells the lobes;
it then absorbs oxygen, which combines with some of its carbon, and is
returned in the form of carbonic acid. This loss of carbon increases the
comparative proportion of hydrogen and oxygen in the seed, and excites
the saccharine fermentation, by which the parenchymatous matter is
converted into a kind of sweet emulsion. In this form it is carried into
the radicle by vessels appropriated to that purpose; and in the mean
time, the fermentation having caused the seed to burst, the cotyledons
are rent asunder, the radicle strikes into the ground and becomes the
root of the plant, and hence the fermented liquid is conveyed to the
plumula, whose vessels have been previously distended by the heat of the
fermentation. The plumula being thus swelled, as it were, by the
258
emulsive fluid, raises itself and springs up to the surface of the
earth, bearing with it the cotyledons, which, as soon as they come in
contact with the air, spread themselves, and are transformed into
leaves.—If we go into the garden, we shall probably find some
seeds in the state which I have described—
Plate XV.
Vol. II. p. 250
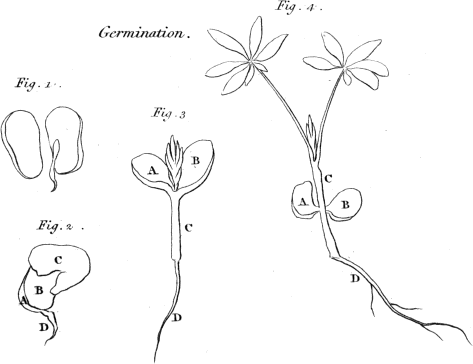
Fig. 1 & 2.
A.B Cotyledons.
C Envelope.
D Radicle.
Fig. 3.
A.B Cotyledons.
C Plumula.
D Radicle.
Fig. 4.
A.B. Cotyledons.
C Plumula.
D Radicle.
Larger view
(complete Plate)
EMILY.
Here are some lupines that are just making their appearance above
ground.
MRS. B.
We shall take up several of them to observe their different degrees
of progress in vegetation. Here is one that has but recently burst its
envelope—do you see the little radicle striking downwards? (Plate XV. Fig. 2.) In
this the plumula is not yet visible. But here is another in a greater
state of forwardness—the plumula, or stem, has risen out of the
ground, and the cotyledons are converted into seed leaves. (Plate XV. Fig. 3.)
CAROLINE.
These leaves are very thick and clumsy, and unlike the other leaves,
which I perceive are just beginning to appear.
MRS. B.
It is because they retain the remains of the parenchyma,
259
with which they still continue to nourish the young plant, as it has not
yet sufficient roots and strength to provide for its sustenance from the
soil.—But, in this third lupine (Plate XV. Fig. 4.), the radicle had sunk deep into the
earth, and sent out several shoots, each of which is furnished with a
mouth to suck up nourishment from the soil; the function of the original
leaves, therefore, being no longer required, they are gradually
decaying, and the plumula is become a regular stem, shooting out small
branches, and spreading its foliage.
EMILY.
There seems to be a very striking analogy between a seed and an egg;
both require an elevation of temperature to be brought to life; both at
first supply with aliment the organised being which they produce; and as
soon as this has attained sufficient strength to procure its own
nourishment, the egg-shell breaks, whilst in the plant the seed-leaves
fall off.
MRS. B.
There is certainly some resemblance between these processes; and when
you become acquainted with animal chemistry, you will frequently be
struck with its analogy to that of the vegetable kingdom.
260
As soon as the young plant feeds from the soil, it requires the
assistance of leaves, which are the organs by which it throws off its
super-abundant fluid; this secretion is much more plentiful in the
vegetable than in the animal creation, and the great extent of surface
of the foliage of plants is admirably calculated for carrying it on in
sufficient quantities. This transpired fluid consists of little more
than water. The sap, by this process, is converted into a liquid of
greater consistence, which is fit to be assimilated to its several
parts.
EMILY.
Vegetation, then, must be essentially injured by destroying the
leaves of the plant?
MRS. B.
Undoubtedly; it not only diminishes the transpiration, but also the
absorption by the roots; for the quantity of sap absorbed is always in
proportion to the quantity of fluid thrown off by transpiration. You
see, therefore, the necessity that a young plant should unfold its
leaves as soon as it begins to derive its nourishment from the soil;
and, accordingly, you will find that those lupines which have dropped
their seed-leaves, and are no longer fed by the parenchyma, have spread
their foliage, in order to perform the office just described.
261
But I should inform you that this function of transpiration seems to
be confined to the upper surface of the leaves, whilst, on the contrary,
the lower surface, which is more rough and uneven, and furnished with a
kind of hair or down, is destined to absorb moisture, or such other
ingredients as the plant derives from the atmosphere.
As soon as a young plant makes its appearance above ground, light, as
well as air, becomes necessary to its preservation. Light is essential
to the development of the colours, and to the thriving of the plant. You
may have often observed what a predilection vegetables have for the
light. If you make any plants grow in a room, they all spread their
leaves, and extend their branches towards the windows.
CAROLINE.
And many plants close up their flowers as soon as it is dark.
EMILY.
But may not this be owing to the cold and dampness of the evening
air?
MRS. B.
That does not appear to be the case; for in a course of curious
experiments, made by Mr. Senebier, of Geneva, on plants which he reared
by
262
lamp-light, he found that the flowers closed their petals whenever the
lamps were extinguished.
EMILY.
But pray, why is air essential to vegetation, plants do not breathe
it like animals?
MRS. B.
At least not in the same manner; but they certainly derive some
principles from the atmosphere, and yield others to it. Indeed, it is
chiefly owing to the action of the atmosphere and the vegetable kingdom
on each other, that the air continues always fit for respiration. But
you will understand this better when I have explained the effect of
water on plants.
I have said that water forms the chief nourishment of plants; it is
the basis not only of the sap, but of all the vegetable juices. Water is
the vehicle which carries into the plant the various salts and other
ingredients required for the formation and support of the vegetable
system. Nor is this all; part of the water itself is decomposed by the
organs of the plant; the hydrogen becomes a constituent part of oil, of
extract, of colouring matter, &c. whilst a portion of the oxygen
enters into the formation of mucilage, of fecula, of sugar, and of
vegetable acids. But the greater part of the oxygen, proceeding from the
decomposition
263
of the water, is converted into a gaseous state by the caloric
disengaged from the hydrogen during its condensation in the formation of
the vegetable materials. In this state the oxygen is transpired by the
leaves of plants when exposed to the sun’s rays. Thus you find that the
decomposition of water, by the organs of the plant, is not only a means
of supplying it with its chief ingredient, hydrogen, but at the same
time of replenishing the atmosphere with oxygen, a principle which
requires continual renovation, to make up for the great consumption of
it occasioned by the numerous oxygenations, combustions, and
respirations, that are constantly taking place on the surface of the
globe.
EMILY.
What a striking instance of the harmony of nature.
MRS. B.
And how admirable the design of Providence, who makes every different
part of the creation thus contribute to the support and renovation of
each other!
But the intercourse of the vegetable and animal kingdoms through the
medium of the atmosphere extends still further. Animals, in breathing,
not only consume the oxygen of the air, but load it with carbonic acid,
which, if accumulated in the atmosphere, would, in a short time, render
it
264
totally unfit for respiration. Here the vegetable kingdom again
interferes; it attracts and decomposes the carbonic acid, retains the
carbon for its own purposes, and returns the oxygen for ours.
CAROLINE.
How interesting this is! I do not know a more beautiful illustration
of the wisdom which is displayed in the laws of nature.
MRS. B.
Faint and imperfect as are the ideas which our limited perceptions
enable us to form of divine wisdom, still they cannot fail to inspire us
with awe and admiration. What, then, would be our feelings, were the
complete system of nature at once displayed before us! So magnificent a
scene would probably be too great for our limited and imperfect
comprehension, and it is no doubt among the wise dispensations of
Providence, to veil the splendour of a glory with which we should be
overpowered. But it is well suited to the nature of a rational being to
explore, step by step, the works of the creation, to endeavour to
connect them into harmonious systems; and, in a word, to trace in the
chain of beings, the kindred ties and benevolent design which unites its
various links, and secure its preservation.
265
CAROLINE.
But of what nature are the organs of plants which are endued with
such wonderful powers?
MRS. B.
They are so minute that their structure, as well as the mode in which
they perform their functions, generally elude our examination; but we
may consider them as so many vessels or apparatus appropriated to perform, with the
assistance of the principle of life, certain chemical processes, by
means of which these vegetable compounds are generated. We may, however,
trace the tannin, resins, gum, mucilage, and some other vegetable
materials, in the organised arrangement of plants, in which they form
the bark, the wood, the leaves, flowers, and seeds.
The bark is composed of the epidermis, the
parenchyma, and the cortical layers.
The epidermis is the external covering of the plant. It is a thin
transparent membrane, consisting of a number of slender fibres, crossing
each other, and forming a kind of net-work. When of a white glossy
nature, as in several species of trees, in the stems of corn and of
seeds, it is composed of a thin coating of siliceous earth, which
accounts for the strength and hardness of those long and slender stems.
Sir H. Davy was led to the discovery of the siliceous nature of the
epidermis of
266
such plants, by observing the singular phenomenon of sparks of fire
emitted by the collision of ratan canes with which two boys were
fighting in a dark room. On analysing the epidermis of the cane, he
found it to be almost entirely siliceous.
CAROLINE.
With iron then, a cane, I suppose, will strike fire very
easily?
MRS. B.
I understand that it will.—In ever-greens the epidermis is
mostly resinous, and in some few plants is formed of wax. The resin,
from its want of affinity for water, tends to preserve the plant from
the destructive effects of violent rains, severe climates, or inclement
seasons, to which this species of vegetables is peculiarly exposed.
EMILY.
Resin must preserve wood just like a varnish, as it is the essential
ingredient of varnishes?
MRS. B.
Yes; and by this means it prevents likewise all unnecessary
expenditure of moisture.
The parenchyma is immediately beneath the epidermis; it is that green
rind which appears when you strip a branch of any tree or shrub of
267
its external coat of bark. The parenchyma is not confined to the stem or
branches, but extends over every part of the plant. It forms the green
matter of the leaves, and is composed of tubes filled with a peculiar
juice.
The cortical layers are immediately in contact with the wood; they
abound with tannin and gallic acid, and consist of small vessels through
which the sap descends after being elaborated in the leaves. The
cortical layers are annually renewed, the old bark being converted into
wood.
EMILY.
But through what vessels does the sap ascend?
MRS. B.
That function is performed by the tubes of the alburnum, or wood,
which is immediately beneath the cortical layers. The wood is composed
of woody fibre, mucilage, and resin. The fibres are disposed in two
ways; some of them longitudinally, and these form what is called the
silver grain of the wood. The others, which are concentric, are called
the spurious grain. These last are disposed in layers, from the number
of which the age of the tree may be computed, a new one being
produced annually by the conversion of the bark into wood. The oldest,
and consequently most internal part of the alburnum, is called
268
heart-wood; it appears to be dead, at least no vital functions are
discernible in it. It is through the tubes of the living alburnum that
the sap rises. These, therefore, spread into the leaves, and there
communicate with the extremities of the vessels of the cortical layers,
into which they pour their contents.
CAROLINE.
Of what use, then, are the tubes of the parenchyma, since neither the
ascending nor descending sap passes through them?
MRS. B.
They are supposed to perform the important function of secreting from
the sap the peculiar juices from which the plant more immediately
derives its nourishment. These juices are very conspicuous, as the
vessels which contain them are much larger than those through which the
sap circulates. The peculiar juices of plants differ much in their
nature, not only in different species of vegetables, but frequently in
different parts of the same individual plant: they are sometimes
saccharine, as in the sugar-cane, sometimes resinous, as in firs and
evergreens, sometimes of a milky appearance, as in the laurel.
EMILY.
I have often observed, that in breaking a young
269
shoot, or in bruising a leaf of laurel, a milky juice will ooze out
in great abundance.
MRS. B.
And it is by making incisions in the bark that pitch, tar, and
turpentine are obtained from fir-trees. The durability of this species
of wood is chiefly owing to the resinous nature of its peculiar juices.
The volatile oils have, in a great measure, the same preservative
effects, as they defend the parts, with which they are connected, from
the attack of insects. This tribe seems to have as great an aversion to
perfumes, as the human species have delight in them. They scarcely ever
attack any odoriferous parts of plants, and it is not uncommon to see
every leaf of a tree destroyed by a blight, whilst the blossoms remain
untouched. Cedar, sandal, and all aromatic woods, are on this account of
great durability.
EMILY.
But the wood of the oak, which is so much esteemed for its
durability, has, I believe, no smell. Does it derive this quality
from its hardness alone?
MRS. B.
Not entirely; for the chesnut,
though considerably harder and firmer than the
oak, is not so lasting. The durability of the oak is, I believe,
270
in a great measure owing to its having very little heart-wood, the
alburnum preserving its vital functions longer than in other trees.
CAROLINE.
If incisions are made into the alburnum and cortical layers, may not
the ascending and descending sap be procured in the same manner as the
peculiar juice is from the vessels of the parenchyma?
MRS. B.
Yes; but in order to obtain specimens of these fluids, in any
quantity, the experiment must be made in the spring, when the sap
circulates with the greatest energy. For this purpose a small bent glass
tube should be introduced into the incision, through which the sap may
flow without mixing with any of the other juices of the tree. From the
bark the sap will flow much more plentifully than from the wood, as the
ascending sap is much more liquid, more abundant, and more rapid in its
motion than that which descends; for the latter having been deprived by
the operation of the leaves of a considerable part of its moisture,
contains a much greater proportion of solid matter, which retards its
motion. It does not appear that there is any excess of descending sap,
as none ever exudes from the roots of plants; this process, therefore,
seems to be carried on only in
271
proportion to the wants of the plant, and the sap descends no further,
and in no greater quantity, than is required to nourish the several
organs. Therefore, though the sap rises and descends in the plant, it
does not appear to undergo a real circulation.
The last of the organs of plants is the flower, or
blossom, which produces the fruits and seed. These
may be considered as the ultimate purpose of nature in the vegetable
creation. From fruits and seeds animals derive both a plentiful source
of immediate nourishment, and an ample provision for the reproduction of
the same means of subsistence.
The seed which forms the final product of mature plants, we have
already examined as constituting the first rudiments of future
vegetation.
These are the principal organs of vegetation, by means of which the
several chemical processes which are carried on during the life of the
plant are performed.
EMILY.
But how are the several principles which enter into the composition
of vegetables so combined by the organs of the plant as to be converted
into vegetable matter?
MRS. B.
By chemical processes, no doubt; but the apparatus in which they are
performed is so extremely
272
minute as completely to elude our examination. We can form an opinion,
therefore, only by the result of these operations. The sap is evidently
composed of water, absorbed by the roots, and holding in solution the
various principles which it derives from the soil. From the roots the
sap ascends through the tubes of the alburnum into the stem, and thence
branches out to every extremity of the plant. Together with the sap
circulates a certain quantity of carbonic acid, which is gradually
disengaged from the former by the internal heat of the plant.
CAROLINE.
What! have vegetables a peculiar heat, analogous to animal heat?
MRS. B.
It is a circumstance that has long been suspected; but late
experiments have decided beyond a doubt that vegetable heat is
considerably above that of unorganised matter in winter, and below it in
summer. The wood of a tree is about sixty degrees, when the thermometer
is seventy or eighty degrees. And the bark, though so much exposed, is
seldom below forty in winter.
It is from the sap, after it has been elaborated by the leaves, that
vegetables derive their nourishment; in its progress through the plant
from the
273
leaves to the roots, it deposits in the several sets of vessels with
which it communicates, the materials on which the growth and nourishment
of each plant depends. It is thus that the various peculiar juices,
saccharine, oily, mucous, acid, and colouring, are formed; as also the
more solid parts, fecula, woody fibre, tannin, resins, concrete salts;
in a word, all the immediate materials of vegetables, as well as the
organised parts of plants, which latter, besides the power of secreting
these from the sap for the general purpose of the plant, have also that
of applying them to their own particular nourishment.
EMILY.
But why should the process of vegetation take place only at one
season of the year, whilst a total inaction prevails during the
other?
MRS. B.
Heat is such an important chemical agent, that its effect, as such,
might perhaps alone account for the impulse which the spring gives to
vegetation. But, in order to explain the mechanism of that operation, it
has been supposed that the warmth of the spring dilates the vessels of
plants, and produces a kind of vacuum, into which the sap (which had
remained in a state of inaction in the trunk during the winter) rises:
this is followed by the
274
ascent of the sap contained in the roots, and room is thus made for
fresh sap, which the roots, in their turn, pump up from the soil. This
process goes on till the plant blossoms and bears fruit, which
terminates its summer career: but when the cold weather sets in, the
fibres and vessels contract, the leaves wither, and are no longer able
to perform their office of transpiration; and, as this secretion stops,
the roots cease to absorb sap from the soil. If the plant be an annual,
its life then terminates; if not, it remains in a state of torpid
inaction during the winter; or the only internal motion that takes place
is that of a small quantity of resinous juice, which slowly rises from
the stem into the branches, and enlarges their buds during the
winter.
CAROLINE.
Yet, in evergreens, vegetation must continue throughout the year.
MRS. B.
Yes; but in winter it goes on in a very imperfect manner, compared to
the vegetation of spring and summer.
We have dwelt much longer on the history of vegetable chemistry than
I had intended; but we have at length, I think, brought the subject
to a conclusion.
275
CAROLINE.
I rather wonder that you did not reserve the account of the
fermentations for the conclusion; for the decomposition of vegetables
naturally follows their death, and can hardly, it seems, be introduced
with so much propriety at any other period.
MRS. B.
It is difficult to determine at what point precisely it may be most
eligible to enter on the history of vegetation; every part of the
subject is so closely connected, and forms such an uninterrupted chain,
that it is by no means easy to divide it. Had I begun with the
germination of the seed, which, at first view, seems to be the most
proper arrangement, I could not have explained the nature and
fermentation of the seed, or have described the changes which manure
must undergo, in order to yield the vegetable elements. To understand
the nature of germination, it is necessary, I think, previously to
decompose the parent plant, in order to become acquainted with the
materials required for that purpose. I hope, therefore, that, upon
second consideration, you will find that the order which I have adopted,
though apparently less correct, is in fact the best calculated for the
elucidation of the subject.
276

MRS. B.
We are now come to the last branch of
chemistry, which comprehends the most complicated order of compound
beings. This is the animal creation, the history of which cannot but
excite the highest degree of curiosity and interest, though we often
fail in attempting to explain the laws by which it is governed.
EMILY.
But since all animals ultimately derive their nourishment from
vegetables, the chemistry of this order of beings must consist merely in
the conversion of vegetable into animal matter.
MRS. B.
Very true; but the manner in which this is effected is, in a great
measure, concealed from our observation. This process is called
animalisation,
277
and is performed by peculiar organs. The difference of the animal and
vegetable kingdoms does not however depend merely on a different
arrangement of combinations. A new principle abounds in the animal
kingdom, which is but rarely and in very small quantities found in
vegetables; this is nitrogen. There is likewise in animal substances a
greater and more constant proportion of phosphoric acid, and other
saline matters. But these are not essential to the formation of animal
matter.
CAROLINE.
Animal compounds contain, then, four fundamental principles; oxygen,
hydrogen, carbon, and nitrogen?
MRS. B.
Yes; and these form the immediate materials of animals, which are
gelatine, albumen, and fibrine.
EMILY.
Are those all? I am surprised that animals should be composed of
fewer kinds of materials than vegetables; for they appear much more
complicated in their organisation.
MRS. B.
Their organisation is certainly more perfect and intricate, and the
ingredients that occasionally
278
enter into their composition are more numerous. But notwithstanding the
wonderful variety observable in the texture of the animal organs, we
find that the original compounds, from which all the varieties of animal
matter are derived, may be reduced to the three heads just mentioned.
Animal substances being the most complicated of all natural compounds,
are most easily susceptible of decomposition, as the scale of
attractions increases in proportion to the number of constituent
principles. Their analysis is, however, both difficult and imperfect;
for as they cannot be examined in their living state, and are liable to
alteration immediately after death, it is probable that, when submitted
to the investigation of a chemist, they are always more or less altered
in their combinations and properties, from what they were, whilst they
made part of the living animal.
EMILY.
The mere diminution of temperature, which they experience by the
privation of animal heat, must, I should suppose, be sufficient to
derange the order of attractions that existed during life.
MRS. B.
That is one of the causes, no doubt: but there are many other
circumstances which prevent us from studying the nature of living animal
substances.
279
We must therefore, in a considerable degree, confine our researches to
the phenomena of these compounds in their inanimate state.
These three kinds of animal matter, gelatine, albumen, and fibrine,
form the basis of all the various parts of the animal system; either
solid, as the skin, flesh, nerves,
membranes, cartilages, and bones; or fluid, as
blood, chyle, milk, mucus, the
gastric and pancreatic juices, bile,
perspiration, saliva, tears, &c.
CAROLINE.
Is it not surprising that so great a variety of substances, and so
different in their nature, should yet all arise from so few materials,
and from the same original elements?
MRS. B.
The difference in the nature of various bodies depends, as I have
often observed to you, rather on their state of combination, than on the
materials of which they are composed. Thus, in considering the chemical
nature of the creation in a general point of view, we observe that it is
throughout composed of a very small number of elements. But when we
divide it into the three kingdoms, we find that, in the mineral, the
combinations seem to result from the union of elements casually brought
together; whilst in the
280
vegetable and animal kingdoms, the attractions are peculiarly and
regularly produced by appropriate organs, whose action depends on the
vital principle. And we may further observe, that by means of certain
spontaneous changes and decompositions, the elements of one kind of
matter become subservient to the reproduction of another; so that the
three kingdoms are intimately connected, and constantly contributing to
the preservation of each other.
EMILY.
There is, however, one very considerable class of elements, which
seems to be confined to the mineral kingdom: I mean metals.
MRS. B.
Not entirely; they are found, though in very minute quantities, both
in the vegetable and animal kingdoms. A small portion of earths and
sulphur enters also into the composition of organised bodies.
Phosphorus, however, is almost entirely confined to the animal kingdom;
and nitrogen, but with few exceptions, is extremely scarce in
vegetables.
Let us now proceed to examine the nature of the three principal
materials of the animal system.
Gelatine, or jelly, is the chief ingredient of skin,
281
and of all the membranous parts of animals. It may be obtained from
these substances, by means of boiling water, under the forms of glue,
size, isinglass, and transparent jelly.
CAROLINE.
But these are of a very different nature; they cannot therefore be
all pure gelatine.
MRS. B.
Not entirely, but very nearly so. Glue is extracted from the skin of
animals. Size is obtained either from skin in its natural state, or from
leather. Isinglass is gelatine procured from a particular species of
fish; it is, you know, of this substance that the finest jelly is made,
and this is done by merely dissolving the isinglass in boiling water,
and allowing the solution to congeal.
EMILY.
The wine, lemon, and spices, are, I suppose, added only to flavour
the jelly?
MRS. B.
Exactly so.
CAROLINE.
But jelly is often made of hartshorn shavings, and of calves’ feet;
do these substances contain gelatine?
282
MRS. B.
Yes. Gelatine may be obtained from almost any animal substance, as it
enters more or less into the composition of all of them. The process for
obtaining it is extremely simple, as it consists merely in boiling the
substance that contains it with water. The gelatine dissolves in water,
and may be attained of any degree of consistence or strength, by
evaporating this solution. Bones in particular produce it very
plentifully, as they consist of phosphat of lime combined or cemented by
gelatine. Horns, which are a species of bone, will yield abundance of
gelatine. The horns of the hart are reckoned to produce gelatine of the
finest quality; they are reduced to the state of shavings in order that
the jelly may be more easily extracted by the water. It is of hartshorn
shavings that the jellies for invalids are usually made, as they are of
very easy digestion.
CAROLINE.
It appears singular that hartshorn, which yields such a powerful
ingredient as ammonia, should at the same time produce so mild and
insipid a substance as jelly?
MRS. B.
And (what is more surprising) it is from the gelatine of bones that
ammonia is produced. You
283
must observe, however, that the processes by which these two substances
are obtained from bones are very different. By the simple action of
water and heat, the gelatine is separated; but in order to procure the
ammonia, or what is commonly called hartshorn, the bones must be
distilled, by which means the gelatine is decomposed, and hydrogen and
nitrogen combined in the form of ammonia. So that the first operation is
a mere separation of ingredients, whilst the second requires a chemical
decomposition.
CAROLINE.
But when jelly is made from hartshorn shavings, what becomes of the
phosphat of lime which constitutes the other part of bones?
MRS. B.
It is easily separated by straining. But the jelly is afterwards more
perfectly purified, and rendered transparent, by adding white of egg,
which being coagulated by heat, rises to the surface along with any
impurities.
EMILY.
I wonder that bones are not used by the common people to make jelly;
a great deal of wholesome nourishment, might, I should
suppose, be procured from them, though the jelly would perhaps
284
not be quite so good as if made from hartshorn shavings?
MRS. B.
There is a prejudice among the poor against a species of food that is
usually thrown to the dogs; and as we cannot expect them to enter into
chemical considerations, it is in some degree excusable. Besides, it
requires a prodigious quantity of fuel to dissolve bones and obtain the
gelatine from them.
The solution of bones in water is greatly promoted by an accumulation
of heat. This may be effected by means of an extremely strong metallic
vessel, called Papin’s digester, in which the bones and water are
enclosed, without any possibility of the steam making its escape.
A heat can thus be applied much superior to that of boiling water;
and bones, by this means, are completely reduced to a pulp. But the
process still consumes too much fuel to be generally adopted among the
lower classes.
CAROLINE.
And why should not a manufacture be established for grinding or
macerating bones, or at least for reducing them to the state of
shavings, when I suppose they would dissolve as readily as hartshorn
shavings?
285
MRS. B.
They could not be collected clean for such a purpose, but they are
not lost, as they are used for making hartshorn and sal ammoniac; and
such is the superior science and industry of this country, that we now
send sal ammoniac to the Levant, though it originally came to us from
Egypt.
EMILY.
When jelly is made of isinglass, does it leave no sediment?
MRS. B.
No; nor does it so much require clarifying, as it consists almost
entirely of pure gelatine,
and any foreign matter that is mixed with
it, is thrown off during the boiling in the form of scum.—These
are processes which you may see performed in great perfection in the
culinary laboratory, by that very able and most useful chemist the
cook.
CAROLINE.
To what an immense variety of purposes chemistry is subservient!
EMILY.
It appears, in that respect, to have an advantage over most other
arts and sciences; for these, very often, have a tendency to confine the
imagination
286
to their own particular object, whilst the pursuit of chemistry is so
extensive and diversified, that it inspires a general curiosity, and a
desire of enquiring into the nature of every object.
CAROLINE.
I suppose that soup is likewise composed of gelatine; for, when cold,
it often assumes the consistence of jelly?
MRS. B.
Not entirely; for though soups generally contain a quantity of
gelatine, the most essential ingredient is a mucous or extractive
matter, a peculiar animal substance, very soluble in water, which
has a strong taste, and is more nourishing than gelatine. The various
kinds of portable soup consist of this extractive matter in a dry state,
which, in order to be made into soup, requires only to be dissolved in
water.
Gelatine, in its solid state, is a semiductile transparent substance,
without either taste or smell.—When exposed to heat, in contact
with air and water, it first swells, then fuses, and finally burns. You
may have seen the first part of this operation performed in the
carpenter’s glue-pot.
CAROLINE.
But you said that gelatine had no smell, and glue has a very
disagreeable one.
287
MRS. B.
Glue is not pure gelatine; as it is not designed for eating, it is
prepared without attending to the state of the ingredients, which are
more or less contaminated by particles that have become putrid.
Gelatine may be precipitated from its solution in water by
alcohol.—We shall try this experiment with a glass of warm
jelly.—You see that the gelatine subsides by the union of the
alcohol and the water.
EMILY.
How is it, then, that jelly is flavoured with wine, without producing
any precipitation?
MRS. B.
Because the alcohol contained in wine is already combined with water,
and other ingredients, and is therefore not at liberty to act upon the
jelly as when in its separate state. Gelatine is soluble both in acids
and in alkalies; the former, you know, are frequently used to season
jellies.
CAROLINE.
Among the combinations of gelatine we must not forget one which you
formerly mentioned; that with tannin, to form leather.
288
MRS. B.
True; but you must observe that leather can be produced only by
gelatine in a membranous state; for though pure gelatine and tannin will
produce a substance chemically similar to leather, yet the texture of
the skin is requisite to make it answer the useful purposes of that
substance.
The next animal substance we are to examine is albumen; this,
although constituting a part of most of the animal compounds, is
frequently found insulated in the animal system; the white of egg, for
instance, consists almost entirely of albumen; the substance that
composes the nerves, the serum, or white part of the blood, and the
curds of milk, are little else than albumen variously modified.
In its most simple state, albumen appears in the form of a
transparent viscous fluid, possessed of no distinct taste or smell; it
coagulates at the low temperature of 165 degrees, and, when once
solidified, it will never return to its fluid state.
Sulphuric acid and alcohol are each of them capable of coagulating
albumen in the same manner as heat, as I am going to show you.
EMILY.
Exactly so.—Pray, Mrs. B., what kind of action
289
is there between albumen and silver? I have sometimes observed,
that if the spoon with which I eat an egg happens to be wetted, it
becomes tarnished.
MRS. B.
It is because the white of egg (and, indeed, albumen in general)
contains a little sulphur, which, at the temperature of an egg just
boiled, will decompose the drop of water that wets the spoon, and
produce sulphurated hydrogen gas, which has the property of tarnishing
silver.
We may now proceed to fibrine. This is an insipid and
inodorous substance, having somewhat the appearance of fine white
threads adhering together; it is the essential constituent of muscles or
flesh, in which it is mixed with and softened by gelatine. It is
insoluble both in water and alcohol, but sulphuric acid converts it into
a substance very analogous to gelatine.
These are the essential and general ingredients of animal matter; but
there are other substances, which, though not peculiar to the animal
system, usually enter into its composition, such as oils, acids,
salts, &c.
Animal oil is the chief constituent of fat; it is contained in
abundance in the cream of milk, whence it is obtained in the form of
butter.
290
EMILY.
Is animal oil the same in its composition as vegetable oils?
MRS. B.
Not the same, but very analogous. The chief difference is that animal
oil contains nitrogen, a principle which seldom enters into the
composition of vegetable oils, and never in so large a proportion.
There are a few animal acids, that is to say, acids peculiar to
animal matter, from which they are almost exclusively obtained.
The animal acids have triple bases of hydrogen, carbon, and nitrogen.
Some of them are found native in animal matter; others are produced
during its decomposition.
Those that we find ready formed are:
The bombic acid, which is obtained from silk-worms.
The formic acid, from ants.
The lactic acid, from the whey of milk.
The sebacic, from oil or fat.
Those produced during the decomposition of animal substances by heat,
are the prussic and zoonic acids. This last is produced by
the roasting of meat, and gives it a brisk flavour.
291
CAROLINE.
The class of animal acids is not very extensive?
MRS. B.
No; nor are they, generally speaking, of great importance. The
prussic acid is, I think, the only one sufficiently
interesting to require any further comment. It can be formed by any
artificial process, without the presence of any animal matter; and it
may likewise be obtained from a variety of vegetables, particularly
those of the narcotic kind, such as poppies, laurel, &c. But it is
commonly obtained from blood, by strongly heating that substance with
caustic potash; the alkali attracts the acid from the blood, and forms
with it a prussiat of potash. From this state of combination the
prussic acid can be obtained pure by means of other substances which
have the power of separating it from the alkali.
EMILY.
But if this acid does not exist ready formed in blood, how can the
alkali attract it from it?
MRS. B.
It is the triple basis only of this acid that exists in the blood;
and this is developed and brought to the state of acid, during the
combustion. The
292
acid therefore is first formed, and it afterwards combines with the
potash.
EMILY.
Now I comprehend it. But how can the prussic acid be artificially
made?
MRS. B.
By passing ammoniacal gas over red-hot charcoal; and hence we learn
that the constituents of this acid are hydrogen, nitrogen, and carbon.
The two first are derived from the volatile alkali, the last from the
combustion of the charcoal.
CAROLINE.
But this does not accord with the system of oxygen being the
principle of acidity.
MRS. B.
The colouring matter of prussian blue is called an acid, because it
unites with alkalies and metals, and not from any other characteristic
properties of acids; perhaps the name is not strictly appropriate. But
this circumstance, together with some others of the same kind, has
induced several chemists to think that oxygen may not be the exclusive
generator of acids. Sir H. Davy, I have already informed you, was
led by his experiments
293
on dry acids to suspect that water might be essential to acidity. And it
is the opinion of some chemists that acidity may possibly depend rather
on the arrangement than on the presence of any particular principles.
But we have not yet done with the prussic acid. It has a strong affinity
for metallic oxyds, and precipitates the solutions of iron in acids of a
blue colour. This is the prussian blue, or prussiat of iron, so much
used in the arts, and with which I think you must be acquainted.
EMILY.
Yes, I am; it is much used in painting, both in oil and in water
colours; but it is not reckoned a permanent oil-colour.
MRS. B.
That defect arises, I believe, in general, from its being badly
prepared, which is the case when the iron is not so fully oxydated as to
form a red oxyd. For a solution of green oxyd of iron (in which the
metal is more slightly oxydated), makes only a pale green, or even a
white precipitate, with prussiat of potash; and this gradually changes
to blue by being exposed to the air, as I can immediately show you.
294
CAROLINE.
It already begins to assume a pale blue colour. But how does the air
produce this change?
MRS. B.
By oxydating the iron more perfectly. If we pour some nitrous acid on
it, the prussian blue colour will be immediately produced, as the acid
will yield its oxygen to the precipitate, and fully saturate it with
this principle, as you shall see.
CAROLINE.
It is very curious to see a colour change so instantaneously.
MRS. B.
Hence you perceive that prussian blue cannot be a permanent colour,
unless prepared with red oxyd of iron, since by exposure to the
atmosphere it gradually darkens, and in a short time is no longer in
harmony with the other colours of the painting.
CAROLINE.
But it can never become darker, by exposure to the atmosphere, than
the true prussian blue, in which the oxyd is perfectly saturated?
MRS. B.
Certainly not. But in painting, the artist not
295
reckoning upon partial alterations in his colours, gives his blue tints
that particular shade which harmonises with the rest of the picture. If,
afterwards, those tints become darker, the harmony of the colouring must
necessarily be destroyed.
CAROLINE.
Pray, of what nature is the paint called carmine?
MRS. B.
It is an animal colour prepared from cochineal, an insect, the
infusion of which produces a very beautiful red.
CAROLINE.
Whilst we are on the subject of colours, I should like to learn
what ivory black is?
MRS. B.
It is a carbonaceous substance obtained by the combustion of ivory.
A more common species of black is obtained from the burning of
bone.
CAROLINE.
But during the combustion of ivory or bone, the carbon, I should
have imagined, must be converted into carbonic acid gas, instead of this
black substance?
296
MRS. B.
In this, as in most combustions, a considerable part of the carbon is
simply volatilised by the heat, and again obtained concrete on cooling.
This colour, therefore, may be called the soot produced by the burning
of ivory or bone.
297

MRS. B.
We have now acquired some idea of the
various materials that compose the animal system; but if you are curious
to know in what manner these substances are formed by the animal organs,
from vegetable, as well as from animal substances, it will be necessary
to have some previous knowledge of the nature and functions of these
organs, without which it is impossible to form any distinct idea of the
process of animalisation and nutrition.
CAROLINE.
I do not exactly understand the meaning of the word
animalisation?
MRS. B.
Animalisation is the process by which the food
298
is assimilated, that is to say, converted into animal matter; and
nutrition is that by which the food thus assimilated is rendered
subservient to the purposes of nourishing and maintaining the animal
system.
EMILY.
This, I am sure, must be the most interesting of all the branches of
chemistry!
CAROLINE.
So I think; particularly as I expect that we shall hear something of
the nature of respiration, and of the circulation of the blood?
MRS. B.
These functions undoubtedly occupy a most important place in the
history of the animal economy.—But I must previously give you a
very short account of the principal organs by which the various
operations of the animal system are performed. These are:
| The | Bones; |
| Muscles, |
| Blood vessels, |
| Lymphatic vessels, |
| Glands, and |
| Nerves. |
299
The bones are the most solid part of the animal frame, and in
a great measure determine its form and dimensions. You recollect,
I suppose, what are the ingredients which enter into their
composition?
CAROLINE.
Yes; phosphat of lime, cemented by gelatine.
MRS. B.
During the earliest period of animal life, they consist almost
entirely of gelatinous membrane having the form of the bones, but of a
loose spongy texture, the cells or cavities of which are destined to be
filled with phosphat of lime; it is the gradual acquisition of this salt
which gives to the bones their subsequent hardness and durability.
Infants first receive it from their mother’s milk, and afterwards derive
it from all animal and from most vegetable food, especially farinaceous
substances, such as wheat-flour, which contain it in sensible
quantities. A portion of the phosphat, after the bones of the
infant have been sufficiently expanded and solidified, is deposited in
the teeth, which consist at first only of a gelatinous membrane or case,
fitted for the reception of this salt; and which, after acquiring
hardness within the gum, gradually protrude from it.
CAROLINE.
How very curious this is; and how ingeniously
300
nature has first provided for the solidification of such bones as are
immediately wanted, and afterwards for the formation of the teeth, which
would not only be useless, but detrimental in infancy!
MRS. B.
In quadrupeds the phosphat of lime is deposited likewise in their
horns, and in the hair or wool with which they are generally
clothed.
In birds it serves also to harden the beaks and the quills of their
feathers.
When animals are arrived at a state of maturity, and their bones have
acquired a sufficient degree of solidity, the phosphat of lime which is
taken with the food is seldom assimilated, excepting when the female
nourishes her young; it is then all secreted into the milk, as a
provision for the tender bones of the nursling.
EMILY.
So that whatever becomes superfluous to one being, is immediately
wanted by another; and the child acquires strength precisely by the
species of nourishment which is no longer necessary to the mother.
Nature is, indeed, an admirable economist!
CAROLINE.
Pray, Mrs. B., does not the disease in the bones
301
of children, called the rickets, proceed from a deficiency of phosphat
of lime?
MRS. B.
I have heard that this disease may arise from two causes; it is
sometimes occasioned by the growth of the muscles being too rapid in
proportion to that of the bones. In this case the weight of the flesh is
greater than the bones can support, and presses upon them so as to
produce a swelling of the joints, which is the great indication of the
rickets. The other cause of this disorder is supposed to be an imperfect
digestion and assimilation of the food, attended with an excess of acid,
which counteracts the formation of phosphat of lime. In both instances,
therefore, care should be taken to alter the child’s diet, not merely by
increasing the quantity of aliment containing phosphat of lime, but also
by avoiding all food that is apt to turn acid on the stomach, and to
produce indigestion. But the best preservative against complaints of
this kind is, no doubt, good nursing: when a child has plenty of air and
exercise, the digestion and assimilation will be properly performed, no
acid will be produced to interrupt these functions, and the muscles and
bones will grow together in just proportions.
302
CAROLINE.
I have often heard the rickets attributed to bad nursing, but I never
could have guessed what connection there was between exercise and the
formation of the bones.
MRS. B.
Exercise is generally beneficial to all the animal functions. If man
is destined to labour for his subsistence, the bread which he earns is
scarcely more essential to his health and preservation than the
exertions by which he obtains it. Those whom the gifts of fortune have
placed above the necessity of bodily labour are compelled to take
exercise in some mode or other, and when they cannot convert it into an
amusement, they must submit to it as a task, or their health will soon
experience the effects of their indolence.
EMILY.
That will never be my case: for exercise, unless it becomes fatigue,
always gives me pleasure; and, so far from being a task, is to me a
source of daily enjoyment. I often think what a blessing it is,
that exercise, which is so conducive to health, should be so delightful;
whilst fatigue, which is rather hurtful, instead of pleasure, occasions
painful sensations. So that fatigue, no doubt, was
303
intended to moderate our bodily exertions, as satiety puts a limit to
our appetites.
MRS. B.
Certainly.—But let us not deviate too far from our
subject.—The bones are connected together by ligaments, which
consist of a white thick flexible substance, adhering to their
extremities, so far as to secure the joints firmly, though without
impeding their motion. And the joints are moreover covered by a solid,
smooth, elastic, white substance, called cartilage, the use of
which is to allow, by its smoothness and elasticity, the bones to slide
easily over one another, so that the joints may perform their office
without difficulty or detriment.
Over the bones the muscles are placed; they consist of bundles
of fibres which terminate in a kind of string, or ligament, by which
they are fastened to the bones. The muscles are the organs of motion; by
their power of dilatation and contraction they put into action the
bones, which act as levers, in all the motions of the body, and form the
solid support of its various parts. The muscles are of various degrees
of strength or consistence in different species of animals. The
mammiferous tribe, or those that suckle their young, seem in this
respect to occupy an intermediate place between birds and cold-blooded
animals, such as reptiles and fishes.
304
EMILY.
The different degrees of firmness and solidity in the muscles of
these several species of animals proceed, I imagine, from the
different nature of the food on which they subsist?
MRS. B.
No; that is not supposed to be the case: for the human species, who
are of the mammiferous tribe, live on more substantial food than birds,
and yet the latter exceed them in muscular strength. We shall hereafter
attempt to account for this difference; but let us now proceed in the
examination of the animal functions.
The next class of organs is that of the vessels of the body,
the office of which is to convey the various fluids throughout the
frame. These vessels are innumerable. The most considerable of them are
those through which the blood circulates, which are of two kinds: the
arteries, which convey it from the heart to the extremities of
the body, and the veins, which bring it back into the heart.
Besides these, there are a numerous set of small transparent vessels,
destined to absorb and convey different fluids into the blood; they are
generally called the absorbent or lymphatic vessels: but
it is to a portion of them only that the function of conveying into the
blood the fluid called lymph is assigned.
EMILY.
Pray what is the nature of that fluid?
305
MRS. B.
The nature and use of the lymph have, I believe, never been perfectly
ascertained; but it is supposed to consist of matter that has been
previously animalised, and which, after answering the purpose for which
it was intended, must, in regular rotation, make way for the fresh
supplies produced by nourishment. The lymphatic vessels pump up this
fluid from every part of the system, and convey it into the veins to be
mixed with the blood which runs through them, and which is commonly
called venous blood.
CAROLINE.
But does it not again enter into the animal system through that
channel?
MRS. B.
Not entirely; for the venous blood does not return into the
circulation until it has undergone a peculiar change, in which it throws
off whatever is become useless.
Another set of absorbent vessels pump up the chyle from the
stomach and intestines, and convey it, after many circumvolutions, into
the great vein near the heart.
EMILY.
Pray what is chyle?
306
MRS. B.
It is the substance into which food is converted by digestion.
CAROLINE.
One set of the absorbent vessels, then, is employed in bringing away
the old materials that are no longer fit for use; whilst the other set
is busy in conveying into the blood the new materials that are to
replace them.
EMILY.
What a great variety of ingredients must enter into the composition
of the blood?
MRS. B.
You must observe that there is also a great variety of substances to
be secreted from it. We may compare the blood to a general receptacle or
storehouse for all kinds of commodities, which are afterwards fashioned,
arranged, and disposed of as circumstances require.
There is another set of absorbent vessels in females which is
destined to secrete milk for the nourishment of the young.
EMILY.
Pray is not milk very analogous in its composition to blood; for,
since the nursling derives its nourishment from that source only, it
must contain
307
every principle which the animal system requires?
MRS. B.
Very true. Milk is found, by its analysis, to contain the principal
materials of animal matter, albumen, oil, and phosphat of lime; so that
the suckling has but little trouble to digest and assimilate this
nourishment. But we shall examine the composition of milk more fully
afterwards.
In many parts of the body numbers of small vessels are collected
together in little bundles called glands, from a Latin word
meaning acorn, on account of the resemblance which some of them bear in
shape to that fruit. The function of the glands is to secrete, or
separate certain matters from the blood.
The secretions are not only mechanical, but chemical separations from
the blood; for the substances thus formed, though contained in the
blood, are not ready combined in that fluid. The secretions are of two
kinds, those which form peculiar animal fluids, as bile, tears, saliva,
&c.; and those which produce the general materials of the animal
system, for the purpose of recruiting and nourishing the several organs
of the body; such as albumen, gelatine, and fibrine; the latter may be
distinguished by the name of nutritive secretions.
308
CAROLINE.
I am quite astonished to hear that all the secretions should be
derived from the blood.
EMILY.
I thought that the bile was produced by the liver?
MRS. B.
So it is; but the liver is nothing more than a very large gland,
which secretes the bile from the blood.
The last of the animal organs which we have mentioned are the
nerves; these are the vehicles of sensation, every other part of
the body being, of itself, totally insensible.
CAROLINE.
They must then be spread through every part of the frame, for we are
every where susceptible of feeling.
EMILY.
Excepting the nails and the hair.
MRS. B.
And those are almost the only parts in which nerves cannot be
discovered. The common source of all the nerves is the brain; thence
they descend, some of them through different holes of the skull, but the
greatest part through the back bone,
309
and extend themselves by innumerable ramifications throughout the whole
body. They spread themselves over the muscles, penetrate the glands,
wind round the vascular system, and even pierce into the interior of the
bones. It is most probably through them that the communication is
carried on between the mind and the other parts of the body; but in what
manner they are acted on by the mind, and made to re-act on the body, is
still a profound secret. Many hypotheses have been formed on this very
obscure subject, but they are all equally improbable, and it would be
useless for us to waste our time in conjectures on an enquiry, which, in
all probability, is beyond the reach of human capacity.
CAROLINE.
But you have not mentioned those particular nerves that form the
senses of hearing, seeing, smelling, and tasting?
MRS. B.
They are considered as being of the same nature as those which are
dispersed over every part of the body, and constitute the general sense
of feeling. The different sensations which they produce arise from their
peculiar situation and connection with the several organs of taste,
smell, and hearing.
310
EMILY.
But these senses appear totally different from that of feeling?
MRS. B.
They are all of them sensations, but variously modified according to
the nature of the different organs in which the nerves are situated.
For, as we have formerly observed, it is by contact only that the nerves
are affected. Thus odoriferous particles must strike upon the nerves of
the nose, in order to excite the sense of smelling; in the same manner
that taste is produced by the particular substance coming in contact
with the nerves of the palate. It is thus also that the sensation of
sound is produced by the concussion of the air striking against the
auditory nerve; and sight is the effect of the light falling upon the
optic nerve. These various senses, therefore, are affected only by the
actual contact of particles of matter, in the same manner as that of
feeling.
The different organs of the animal body, though easily separated and
perfectly distinct, are loosely connected together by a kind of spongy
substance, in texture somewhat resembling net-work, called the cellular
membrane; and the whole is covered by the skin.
The skin, as well as the bark of vegetables, is formed of
three coats. The external one is called the cuticle or
epidermis; the second, which is
311
called the mucous membrane, is of a thin soft texture, and
consists of a mucous substance, which in negroes is black, and is the
cause of their skin appearing of that colour.
CAROLINE.
Is then the external skin of negroes white like ours?
MRS. B.
Yes; but as the cuticle is transparent, as well as porous, the
blackness of the mucous membrane is visible through it. The extremities
of the nerves are spread over this skin, so that the sensation of
feeling is transmitted through the cuticle. The internal covering of the
muscles, which is properly the skin, is the thickest, the toughest, and
most resisting of the whole; it is this membrane which is so essential
in the arts, by forming leather when combined with tannin.
The skin which covers the animal body, as well as those membranes
that form the coats of the vessels, consists almost exclusively of
gelatine; and is capable of being converted into glue, size, or
jelly.
The cavities between the muscles and the skin are usually filled with
fat, which lodges in the cells of the membranous net before mentioned,
and gives to the external form (especially in the
312
human figure) that roundness, smoothness, and softness, so essential to
beauty.
EMILY.
And the skin itself is, I think, a very ornamental part of the
human frame, both from the fineness of its texture, and the variety and
delicacy of its tints.
MRS. B.
This variety and harmonious graduation of colours, proceed, not so
much from the skin itself, as from the internal organs which transmit
their several colours through it, these being only softened and blended
by the colour of the skin, which is uniformly of a yellowish white.
Thus modified, the darkness of the veins appears of a pale blue
colour, and the floridness of the arteries is changed to a delicate
pink. In the most transparent parts, the skin exhibits the bloom of the
rose, whilst where it is more opake its own colour predominates; and at
the joints, where the bones are most prominent, their whiteness is often
discernible. In a word, every part of the human frame seems to
contribute to its external grace; and this not merely by producing a
pleasing variety of tints, but by a peculiar kind of beauty which
belongs to each individual part. Thus it is to the solidity and
arrangement of the bones that the human figure owes the grandeur of its
stature,
313
and its firm and dignified deportment. The muscles delineate the form,
and stamp it with energy and grace; and the soft substance which is
spread over them smooths their ruggedness, and gives to the contours the
gentle undulations of the line of beauty. Every organ of sense is a
peculiar and separate ornament; and the skin, which polishes the
surface, and gives it that charm of colouring so inimitable by art,
finally conspires to render the whole the fairest work of the
creation.
But now that we have seen in what manner the animal frame is formed,
let us observe how it provides for its support, and how the several
organs, which form so complete a whole, are nourished and
maintained.
This will lead us to a more particular explanation of the internal
organs: here we shall not meet with so much apparent beauty, because
these parts were not intended by nature to be exhibited to view; but the
beauty of design, in the internal organisation of the animal frame, is,
if possible, still more remarkable than that of the external parts.
We shall defer this subject till our next interview.
314
CONVERSATION XXV.
ON ANIMALISATION, NUTRITION, AND
RESPIRATION.

MRS. B.
We have now learnt of what materials the
animal system is composed, and have formed some idea of the nature of
its organisation. In order to complete the subject, it remains for us to
examine in what manner it is nourished and supported.
Vegetables, we have observed, obtain their nourishment from various
substances, either in their elementary state, or in a very simple state
of combination; as carbon, water, and salts, which they pump up from the
soil; and carbonic acid and oxygen, which they absorb from the
atmosphere.
Animals, on the contrary, feed on substances of the most complicated
kind; for they derive their sustenance, some from the animal creation,
others from the vegetable kingdom, and some from both.
315
CAROLINE.
And there is one species of animals, which, not satisfied with
enjoying either kind of food in its simple state, has invented the art
of combining them together in a thousand ways, and of rendering even the
mineral kingdom subservient to its refinements.
EMILY.
Nor is this all; for our delicacies are collected from the various
climates of the earth, so that the four quarters of the globe are often
obliged to contribute to the preparation of our simplest dishes.
CAROLINE.
But the very complicated substances which constitute the nourishment
of animals, do not, I suppose, enter into their system in their
actual state of combination?
MRS. B.
So far from it, that they not only undergo a new arrangement of their
parts, but a selection is made of such as are most proper for the
nourishment of the body, and those only enter into the system, and are
animalised.
EMILY.
And by what organs is this process performed?
MRS. B.
Chiefly by the stomach, which is the organ of
316
digestion, and the prime regulator of the animal frame.
Digestion is the first step towards nutrition. It consists in
reducing into one homogeneous mass the various substances that are taken
as nourishment; it is performed by first chewing and mixing the solid
aliment with the saliva, which reduces it to a soft mass, in which state
it is conveyed into the stomach, where it is more completely dissolved
by the gastric juice.
This fluid (which is secreted into the stomach by appropriate glands)
is so powerful a solvent that scarcely any substances will resist its
action.
EMILY.
The coats of the stomach, however, cannot be attacked by it,
otherwise we should be in danger of having them destroyed when the
stomach was empty.
MRS. B.
They are probably not subject to its action; as long, at least, as
life continues. But it appears, that when the gastric juice has no
foreign substance to act upon, it is capable of occasioning a degree of
irritation in the coats of the stomach, which produces the sensation of
hunger. The gastric juice, together with the heat and muscular action of
the stomach, converts the aliment into an uniform pulpy mass called
chyme. This passes
317
into the intestines, where it meets with the bile and some other fluids,
by the agency of which, and by the operation of other causes hitherto
unknown, the chyme is changed into chyle, a much thinner substance,
somewhat resembling milk, which is pumped by immense numbers of small
absorbent vessels spread over the internal surface of the intestines.
These, after many circumvolutions, gradually meet and unite into large
branches, till they at length collect the chyle into one vessel, which
pours its contents into the great vein near the heart, by which means
the food, thus prepared, enters into the circulation.
CAROLINE.
But I do not yet clearly understand how the blood, thus formed,
nourishes the body and supplies all the secretions?
MRS. B.
Before this can be explained to you, you must first allow me to
complete the formation of the blood. The chyle may, indeed, be
considered as forming the chief ingredient of blood; but this fluid is
not perfect until it has passed through the lungs, and undergone
(together with the blood that has already circulated) certain necessary
changes that are effected by RESPIRATION.
318
CAROLINE.
I am very glad that you are going to explain the nature of
respiration: I have often longed to understand it, for though we
talk incessantly of breathing, I never knew precisely what
purpose it answered.
MRS. B.
It is indeed one of the most interesting processes imaginable; but,
in order to understand this function well, it will be necessary to enter
into some previous explanations. Tell me, Emily,—what do you
understand by respiration?
EMILY.
Respiration, I conceive, consists simply in alternately
inspiring air into the lungs, and expiring it from
them.
MRS. B.
Your answer will do very well as a general definition. But, in order
to form a tolerably clear notion of the various phenomena of
respiration, there are many circumstances to be taken into
consideration.
In the first place, there are two things to be distinguished in
respiration, the mechanical and the chemical part of the
process.
The mechanism of breathing depends on the alternate expansions and
contractions of the chest, in which the lungs are contained. When the
319
chest dilates, the cavity is enlarged, and the air rushes in at the
mouth, to fill up the vacuum formed by this dilatation; when it
contracts, the cavity is diminished, and the air forced out again.
CAROLINE.
I thought that it was the lungs that contracted and expanded in
breathing?
MRS. B.
They do likewise; but their action is only the consequence of that of
the chest. The lungs, together with the heart and largest blood vessels,
in a manner fill up the cavity of the chest; they could not, therefore,
dilate if the chest did not previously expand; and, on the other hand,
when the chest contracts, it compresses the lungs and forces the air out
of them.
CAROLINE.
The lungs, then, are like bellows, and the chest is the power that
works them.
MRS. B.
Precisely so. Here is a curious little figure (Plate XV. Fig. 5.), that will assist me in
explaining the mechanism of breathing.
Plate XV.
Vol. II. p. 250
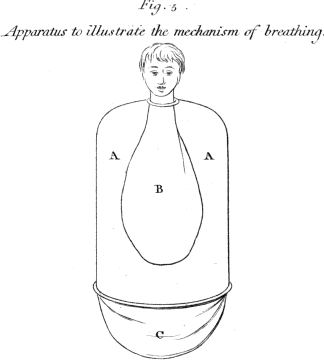
Fig. 5. A.A Glass Bell.
B Bladder representing the lungs.
C Bladder representing the Diaphragm.
Larger view
(complete Plate)
CAROLINE.
What a droll figure! a little head fixed upon a glass bell, with a
bladder tied over the bottom of it!
320
MRS. B.
You must observe that there is another bladder within the glass, the
neck of which communicates with the mouth of the figure—this
represents the lungs contained within the chest; the other bladder,
which you see is tied loose, represents a muscular membrane, called the
diaphragm, which separates the chest from the lower part of the
body. By the chest, therefore, I mean that large cavity in the
upper part of the body contained within the ribs, the neck, and the
diaphragm; this membrane is muscular, and capable of contraction and
dilatation. The contraction may be imitated by drawing the bladder tight
over the bottom of the receiver, when the air in the bladder, which
represents the lungs, will be forced out through the mouth of the
figure—
EMILY.
See, Caroline, how it blows the flame of the candle in breathing!
MRS. B.
By letting the bladder loose again, we imitate the dilatation of the
diaphragm, and the cavity of the chest being enlarged, the lungs expand,
and the air rushes in to fill them.
EMILY.
This figure, I think, gives a very clear idea of the process of
breathing.
321
MRS. B.
It illustrates tolerably well the action of the lungs and diaphragm;
but those are not the only powers that are concerned in enlarging or
diminishing the cavity of the chest; the ribs are also possessed of a
muscular motion for the same purpose; they are alternately drawn in,
edgeways, to assist the contraction, and stretched out, like the hoops
of a barrel, to contribute to the dilatation of the chest.
EMILY.
I always supposed that the elevation and depression of the ribs were
the consequence, not the cause of breathing.
MRS. B.
It is exactly the reverse. The muscular action of the diaphragm, together with
that of the ribs, are the causes of the contraction and expansion
of the chest; and the air rushing into, and being expelled from the
lungs, are only consequences of those actions.
CAROLINE.
I confess that I thought the act of breathing began by opening the
mouth for the air to rush in, and that it was the air alone, which, by
alternately rushing in and out, occasioned the dilatations and
contractions of the lungs and chest.
322
MRS. B.
Try the experiment of merely opening your mouth; the air will not
rush in, till by an interior muscular action you produce a
vacuum—yes, just so, your diaphragm is now dilated, and the ribs
expanded. But you will not be able to keep them long in that state. Your
lungs and chest are already resuming their former state, and expelling
the air with which they had just been filled. This mechanism goes on
more or less rapidly, but, in general, a person at rest and in
health will breathe between fifteen and twenty-five times in a
minute.
We may now proceed to the chemical effects of respiration; but, for
this purpose, it is necessary that you should previously have some
notion of the circulation of the blood. Tell me, Caroline, what
do you understand by the circulation of the blood?
CAROLINE.
I am delighted that you come to that subject, for it is one that has
long excited my curiosity. But I cannot conceive how it is connected
with respiration. The idea I have of the circulation is, that the blood
runs from the heart through the veins all over the body, and back again
to the heart.
MRS. B.
I could hardly have expected a better definition
323
from you; it is, however, not quite correct, for you do not distinguish
the arteries from the veins, which, as we have already
observed, are two distinct sets of vessels, each having its own peculiar
functions. The arteries convey the blood from the heart to the
extremities of the body; and the veins bring it back into the heart.
This sketch will give you an idea of the manner in which some of the
principal veins and arteries of the human body branch out of the heart,
which may be considered as a common centre to both sets of vessels. The
heart is a kind of strong elastic bag, or muscular cavity, which
possesses a power of dilating and contracting itself, for the purposes
of alternately receiving and expelling the blood, in order to carry on
the process of circulation.
EMILY.
Why are the arteries in this drawing painted red, and the veins
purple?
MRS. B.
It is to point out the difference of the colour of the blood in these
two sets of vessels.
CAROLINE.
But if it is the same blood that flows from the arteries into the
veins, how can its colour be changed?
324
MRS. B.
This change arises from various circumstances. In the first place,
during its passage through the arteries, the blood undergoes a
considerable alteration, some of its constituent parts being gradually
separated from it for the purpose of nourishing the body, and of
supplying the various secretions. The consequence of this is, that the
florid arterial colour of the blood changes by degrees to a deep purple,
which is its constant colour in the veins. On the other hand, the blood
is recruited during its return through the veins by the fresh chyle, or
imperfect blood, which has been produced by food; and it receives also
lymph from the absorbent vessels, as we have before mentioned. In
consequence of these several changes, the blood returns to the heart in
a state very different from that in which it left it. It is loaded with
a greater proportion of hydrogen and carbon, and is no longer fit for
the nourishment of the body, or other purposes of circulation.
EMILY.
And in this state does it mix in the heart with the pure florid blood
that runs into the arteries?
MRS. B.
No. The heart is divided into two cavities or compartitions, called
the right and left ventricles.
325
The left ventricle is the receptacle for the pure arterial blood
previous to its circulation; whilst the venous, or impure blood, which
returns to the heart after having circulated, is received into the right
ventricle, previous to its purification, which I shall presently
explain.
CAROLINE.
For my part, I always thought that the same blood circulated again
and again through the body, without undergoing any change.
MRS. B.
Yet you must have supposed that the blood circulated for some
purpose?
CAROLINE.
I knew that it was indispensable to life; but had no idea of its real
functions.
MRS. B.
But now that you understand that the blood conveys nourishment to
every part of the body, and supplies the various secretions, you must be
sensible that it cannot constantly answer these objects without being
proportionally renovated and purified.
CAROLINE.
But does not the chyle answer this purpose?
326
MRS. B.
Only in part. It renovates the nutritive principles of the blood, but
does not relieve it from the superabundance of water and carbon with
which it is encumbered.
EMILY.
How, then, is this effected?
MRS. B.
By RESPIRATION. This is one of the
grand mysteries which modern chemistry has disclosed. When the venous
blood enters the right ventricle of the heart, it contracts by its
muscular power, and throws the blood through a large vessel into the
lungs, which are contiguous, and through which it circulates by millions
of small ramifications. Here it comes in contact with the air which we
breathe. The action of the air on the blood in the lungs is, indeed,
concealed, from our immediate observation; but we are able to form a
tolerably accurate judgment of it from the changes which it effects not
only in the blood, but also on the air expired.
The air, after passing through the lungs, is found to contain all the
nitrogen inspired, but to have lost part of its oxygen, and to have
acquired a portion of watery vapour and of carbonic acid gas. Hence it
is inferred, that when the air comes
327
in contact with the venous blood in the lungs, the oxygen attracts from
it the superabundant quantity of carbon with which it has impregnated
itself during the circulation, and converts it into carbonic acid. This
gaseous acid, together with the redundant moisture from the lungs*, being
then expired, the blood is restored to its former purity, that is, to
the state of arterial blood, and is thus again enabled to perform its
various functions.
CAROLINE.
This is truly wonderful! Of all that we have yet learned, I do
not recollect any thing that has appeared to me so curious and
interesting. I almost believe that I should like to study anatomy
now, though I have hitherto had so disgusting an idea of it. Pray, to
whom are we indebted for these beautiful discoveries?
MRS. B.
Priestley and Crawford, in this country, and Lavoisier, in France,
are the principal inventors of the theory of respiration. Of late years
the subject has been farther illustrated and simplified by the accurate
experiments of Messrs. Allen and Pepys. But the still more important and
more
328
admirable discovery of the circulation of the blood was made long before
by our immortal countryman Harvey.
EMILY.
Indeed I never heard any thing that delighted me so much as this
theory of respiration. But I hope, Mrs. B., that you will enter a
little more into particulars before you dismiss so interesting a
subject. We left the blood in the lungs to undergo the salutary change:
but how does it thence spread to all the parts of the body?
MRS. B.
After circulating through the lungs, the blood is collected into four
large vessels, by which it is conveyed into the left ventricle of the
heart, whence it is propelled to all the different parts of the body by
a large artery, which gradually ramifies into millions of small arteries
through the whole frame. From the extremities of these little
ramifications the blood is transmitted to the veins, which bring it back
to the heart and lungs, to go round again and again in the manner we
have just described. You see, therefore, that the blood actually
undergoes two circulations; the one, through the lungs, by which it is
converted into pure arterial blood; the other, or general circulation,
by which nourishment is conveyed to every part of the
329
body; and these are both equally indispensable to the support of animal
life.
EMILY.
But whence proceeds the carbon with which the blood is impregnated
when it comes into the lungs?
MRS. B.
Carbon exists in a greater proportion in blood than in organised
animal matter. The blood, therefore, after supplying its various
secretions, becomes loaded with an excess of carbon, which is carried
off by respiration; and the formation of new chyle from the food affords
a constant supply of carbonaceous matter.
CAROLINE.
I wonder what quantity of carbon may be expelled from the blood by
respiration in the course of 24 hours?
MRS. B.
It appears by the experiments of Messrs. Allen and Pepys that about
40,000 cubic inches of carbonic acid gas are emitted from the lungs of a
healthy person, daily; which is equivalent to eleven ounces of
solid carbon every 24 hours.
EMILY.
What an immense quantity! And pray how
330
much of carbonic acid gas do we expel from our lungs at each
expiration?
MRS. B.
The quantity of air which we take into our lungs at each inspiration,
is about 40 cubic inches, which contain a little less than 10 cubic
inches of oxygen; and of those 10 inches, one-eighth is converted into
carbonic acid gas on passing once through the lungs*, a change which is
sufficient to prevent air which has only been breathed once from
suffering a taper to burn in it.
CAROLINE.
Pray, how does the air come in contact with the blood in the
lungs?
MRS. B.
I cannot answer this question without entering into an explanation of
the nature and structure of the lungs. You recollect that the venous
blood, on being expelled from the right ventricle, enters the lungs to
go through what we may call the lesser circulation; the large trunk or
vessel that conveys it branches out, at its entrance into the lungs,
into an infinite number of very fine ramifications.
331
The windpipe, which conveys the air from the mouth into the lungs,
likewise spreads out into a corresponding number of air vessels, which
follow the same course as the blood vessels, forming millions of very
minute air-cells. These two sets of vessels are so interwoven as to form
a sort of net-work, connected into a kind of spongy mass, in which every
particle of blood must necessarily come in contact with a particle of
air.
CAROLINE.
But since the blood and the air are contained in different vessels,
how can they come into contact?
MRS. B.
They act on each other through the membrane which forms the coats of
these vessels; for although this membrane prevents the blood and the air
from mixing together in the lungs, yet it is no impediment to their
chemical action on each other.
EMILY.
Are the lungs composed entirely of blood vessels and air vessels?
MRS. B.
I believe they are, with the addition only of nerves and of a small
quantity of the cellular substance before mentioned, which connects the
whole into an uniform mass.
332
EMILY.
Pray, why are the lungs always spoken of in the plural number? Are
there more than one?
MRS. B.
Yes; for though they form but one organ, they really consist of two
compartments called lobes, which are enclosed in separate membranes or
bags, each occupying one side of the chest, and being in close contact
with each other, but without communicating together. This is a beautiful
provision of nature, in consequence of which, if one of the lobes be
wounded, the other performs the whole process of respiration till the
first is healed.
The blood, thus completed, by the process of respiration, forms the
most complex of all animal compounds, since it contains not only the
numerous materials necessary to form the various secretions, as saliva,
tears, &c. but likewise all those that are required to nourish the
several parts of the body, as the muscles, bones, nerves,
glands, &c.
EMILY.
There seems to be a singular analogy between the blood of animals and
the sap of vegetables; for each of these fluids contains the several
materials destined for the nutrition of the numerous class of bodies to
which they respectively belong.
333
MRS. B.
Nor is the production of these fluids in the animal and vegetable
systems entirely different; for the absorbent vessels, which pump up the
chyle from the stomach and intestines, may be compared to the absorbents
of the roots of plants, which suck up the nourishment from the soil. And
the analogy between the sap and the blood may be still further traced,
if we follow the latter in the course of its circulation; for, in the
living animal, we find every where organs which are possessed of a power
to secrete from the blood and appropriate to themselves the ingredients
requisite for their support.
CAROLINE.
But whence do these organs derive their respective powers?
MRS. B.
From a peculiar organisation, the secret of which no one has yet been
able to unfold. But it must be ultimately by means of the vital
principle that both their mechanical and chemical powers are brought
into action.
I cannot dismiss the subject of circulation without mentioning
perspiration, a secretion which is immediately connected
with it, and acts a most important part in the animal economy.
334
CAROLINE.
Is not this secretion likewise made by appropriate glands?
MRS. B.
No; it is performed by the extremities of the arteries, which
penetrate through the skin and terminate under the cuticle, through the
pores of which the perspiration issues. When this fluid is not secreted
in excess, it is insensible, because it is dissolved by the air
as it exudes from the pores; but when it is secreted faster than it can
be dissolved, it becomes sensible, as it assumes its liquid
state.
EMILY.
This secretion bears a striking resemblance to the transpiration of
the sap of plants. They both consist of the most fluid part, and both
exude from the surface by the extremities of the vessels through which
they circulate.
MRS. B.
And the analogy does not stop there; for, since it has been
ascertained that the sap returns into the roots of the plants, the
resemblance between the animal and vegetable circulation is become still
more obvious. The latter, however, is far from being complete, since, as
we observed before, it consists only in a rising and descending of
335
the sap, whilst in animals the blood actually circulates through
every part of the system.
We have now, I think, traced the process of nutrition, from the
introduction of the food into the stomach to its finally becoming a
constituent part of the animal frame. This will, therefore, be a fit
period to conclude our present conversation. What further remarks we
have to make on the animal economy shall be reserved for our next
interview.
336
CONVERSATION XXVI.
ON ANIMAL HEAT; AND ON VARIOUS ANIMAL
PRODUCTS.

EMILY.
Since our last interview, I have been
thinking much of the theory of respiration; and I cannot help being
struck with the resemblance which it appears to bear to the process of
combustion. For in respiration, as in most cases of combustion, the air
suffers a change, and a portion of its oxygen combines with carbon,
producing carbonic acid gas.
MRS. B.
I am much pleased that this idea has occurred to you: these two
processes appear so very analogous, that it has been supposed that a
kind of combustion actually takes place in the lungs; not
337
of the blood, but of the superfluous carbon which the oxygen attracts
from it.
CAROLINE.
A combustion in our lungs! that is a curious idea indeed! But,
Mrs. B., how can you call the action of the air on the blood in the
lungs combustion, when neither light nor heat are produced
by it?
EMILY.
I was going to make the same objection.—Yet I do not conceive
how the oxygen can combine with the carbon, and produce carbonic acid,
without disengaging heat?
MRS. B.
The fact is, that heat is disengaged.* Whether any light be evolved,
I cannot pretend to determine; but that heat is produced in
considerable and very sensible quantities is certain, and this is the
principal, if not the only source of ANIMAL
HEAT.
EMILY.
How wonderful! that the very process which purifies and elaborates
the blood, should afford an inexhaustible supply of internal heat?
338
MRS. B.
This is the theory of animal heat in its original simplicity, such
nearly as it was first proposed by Black and Lavoisier. It was equally
clear and ingenious; and was at first generally adopted. But it was
objected, on second consideration, that if the whole of the animal heat
was evolved in the lungs, it would necessarily be much less in the
extremities of the body than immediately at its source; which is not
found to be the case. This objection, however, which was by no means
frivolous, is now satisfactorily removed by the following
consideration:—Venous blood has been found by experiment to have
less capacity for heat than arterial blood; whence it follows
that the blood, in gradually passing from the arterial to the venous
state, during the circulation, parts with a portion of caloric, by means
of which heat is diffused through every part of the body.
EMILY.
More and more admirable!
CAROLINE.
The cause of animal heat was always a perfect mystery to me, and I am
delighted with its explanation.—But pray, Mrs. B., can you
tell me what is the reason of the increase of heat that takes place in a
fever?
339
EMILY.
Is it not because we then breathe quicker, and therefore more heat is
disengaged in the system?
MRS. B.
That may be one reason: but I should think that the principal cause
of the heat experienced in fevers, is, that there is no vent for the
caloric which is generated in the body. One of the most considerable
secretions is the insensible perspiration; this is constantly carrying
off caloric in a latent state; but during the hot stage of a fever, the
pores are so contracted, that all perspiration ceases, and the
accumulation of caloric in the body occasions those burning sensations
which are so painful.
EMILY.
This is, no doubt, the reason why the perspiration that often
succeeds the hot stage of a fever affords so much relief. If I had known
this theory of animal heat when I had a fever last summer, I think
I should have found some amusement in watching the chemical processes
that were going on within me.
CAROLINE.
But exercise likewise produces animal heat, and that must be quite in
a different manner.
340
MRS. B.
Not so much so as you think; for the more exercise you take, the more
the body is stimulated, and requires recruiting. For this purpose the
circulation of the blood is quickened, the breath proportionably
accelerated, and consequently a greater quantity of caloric evolved.
CAROLINE.
True; after running very fast, I gasp for breath, my respiration is
quick and hard, and it is just then that I begin to feel hot.
EMILY.
It would seem, then, that violent exercise should produce fever.
MRS. B.
Not if the person is in a good state of health; for the additional
caloric is then carried off by the perspiration which succeeds.
EMILY.
What admirable resources nature has provided for us! By the
production of animal heat she has enabled us to keep up the temperature
of our bodies above that of inanimate objects; and whenever this source
becomes too abundant, the excess is carried off by perspiration.
341
MRS. B.
It is by the same law of nature that we are enabled, in all climates,
and in all seasons, to preserve our bodies of an equal temperature, or
at least very nearly so.
CAROLINE.
You cannot mean to say that our bodies are of the same temperature in
summer, and in winter, in England, and in the West-Indies.
MRS. B.
Yes, I do; at least if you speak of the temperature of the blood, and
the internal parts of the body; for those parts that are immediately in
contact with the atmosphere, such as the hands and face, will
occasionally get warmer, or colder, than the internal or more sheltered
parts. But if you put the bulb of a thermometer in your mouth, which is
the best way of ascertaining the real temperature of your body, you will
scarcely perceive any difference in its indication, whatever may be the
difference of temperature of the atmosphere.
CAROLINE.
And when I feel overcome by heat, I am really not hotter than when I
am shivering with cold?
342
MRS. B.
When a person in health feels very hot, whether from internal heat,
from violent exercise, or from the temperature of the atmosphere, his
body is certainly a little warmer than when he feels very cold; but this
difference is much smaller than our sensations would make us believe;
and the natural standard is soon restored by rest and by perspiration.
It is chiefly the external parts that are warmer, and I am sure that you
will be surprised to hear that the internal temperature of the body
scarcely ever descends below ninety-five or ninety-six degrees, and
seldom attains one hundred and four or one hundred and five degrees,
even in the most violent fevers.
EMILY.
The greater quantity of caloric, therefore, that we receive from the
atmosphere in summer, cannot raise the temperature of our bodies beyond
certain limits, as it does that of inanimate bodies, because an excess
of caloric is carried off by perspiration.
CAROLINE.
But the temperature of the atmosphere, and consequently that of
inanimate bodies, is surely never so high as that of animal heat?
343
MRS. B.
I beg your pardon. Frequently in the East and West Indies, and
sometimes in the southern parts of Europe, the atmosphere is above
ninety-eight degrees, which is the common temperature of animal heat.
Indeed, even in this country, it occasionally happens that the sun’s
rays, setting full on an object, elevate its temperature above that
point.
In illustration of the power which our bodies have to resist the
effects of external heat, Sir Charles Blagden, with some other
gentlemen, made several very curious experiments. He remained for some
time in an oven heated to a temperature not much inferior to that of
boiling water, without suffering any other inconvenience than a profuse
perspiration, which he supported by drinking plentifully.
EMILY.
He could scarcely consider the perspiration as an inconvenience,
since it saved him from being baked by giving vent to the excess of
caloric.
CAROLINE.
I always thought, I confess, that it was from the heat of the
perspiration that we suffered in summer.
344
MRS. B.
You now find that you are quite mistaken. Whenever evaporation takes
place, cold, you know, is produced in consequence of a quantity of
caloric being carried off in a latent state; this is the case with
perspiration, and it is in this way that it affords relief. It is on
that account also that we are so apt to catch cold, when in a
state of profuse perspiration. It is for the same reason that tea is
often refreshing in summer, though it appears to heat you at the moment
you drink it.
EMILY.
And in winter, on the contrary, tea is pleasant on account of its
heat.
MRS. B.
Yes; for we have then rather to guard against a deficiency than an
excess of caloric, and you do not find that tea will excite perspiration
in winter, unless after dancing, or any other violent exercise.
CAROLINE.
What is the reason that it is dangerous to eat ice after dancing, or
to drink any thing cold when one is very hot?
345
MRS. B.
Because the loss of heat arising from the perspiration, conjointly
with the chill occasioned by the cold draught, produce more cold than
can be borne with safety, unless you continue to use the same exercise
after drinking that you did before; for the heat occasioned by the
exercise will counteract the effects of the cold drink, and the danger
will be removed. You may, however, contrary to the common notion,
consider it as a rule, that cold liquids may, at all times, be drunk
with perfect safety, however hot you may feel, provided you are not at
the moment in a state of great perspiration, and on condition that you
keep yourself in gentle exercise afterwards.
EMILY.
But since we are furnished with such resources against the extremes
of heat or cold, I should have thought that all climates would have
been equally wholesome.
MRS. B.
That is true, in a certain degree, with regard to those who have been
accustomed to them from birth; for we find that the natives of those
climates, which we consider as most deleterious, are as healthy as
ourselves; and if such climates are unwholesome to those who are
habituated to a
346
more moderate temperature, it is because the animal economy does not
easily accustom itself to considerable changes.
CAROLINE.
But pray, Mrs. B., if the circulation preserves the body of an
uniform temperature, how does it happen that animals are sometimes
frozen?
MRS. B.
Because, if more heat be carried off by the atmosphere than the
circulation can supply, the cold will finally prevail, the heart will
cease to beat, and the animal will be frozen. And, likewise, if the body
remained long exposed to a degree of heat, greater than the perspiration
could carry off, it would at last lose the power of resisting its
destructive influence.
CAROLINE.
Fish, I suppose, have no animal heat, but only partake of the
temperature of the water in which they live?
EMILY.
And their coldness, no doubt, proceeds from their not breathing?
MRS. B.
All kinds of fish breathe more or less, though
347
in a much smaller degree than land animals. Nor are they entirely
destitute of animal heat, though, for the same reason, they are much
colder than other creatures. They have comparatively but a very small
quantity of blood, therefore but very little oxygen is required, and a
proportionally small quantity of animal heat is generated.
CAROLINE.
But how can fish breathe under water?
MRS. B.
They breathe by means of the air which is dissolved in the water, and
if you put them into water deprived of air by boiling, they are soon
suffocated.
If a fish is confined in a vessel of water closed from the air, it
soon dies; and any fish put in afterwards would be killed immediately,
as all the air had been previously consumed.
CAROLINE.
Are there any species of animals that breathe more than
we do?
MRS. B.
Yes; birds, of all animals, breathe the greatest quantity of air in
proportion to their size; and it
348
is to this that they are supposed to owe the peculiar firmness and
strength of their muscles, by which they are enabled to support the
violent exertion of flying.
This difference between birds and fish, which may be considered as
the two extremes of the scale of muscular strength, is well worth
observing. Birds residing constantly in the atmosphere, surrounded by
oxygen, and respiring it in greater proportions than any other species
of animals, are endowed with a superior degree of muscular strength,
whilst the muscles of fish, on the contrary, are flaccid and oily; these
animals are comparatively feeble in their motions, and their temperature
is scarcely above that of the water in which they live. This is, in all
probability, owing to their imperfect respiration; the quantity of
hydrogen and carbon, that is in consequence accumulated in their bodies,
forms the oil which is so strongly characteristic of that species of
animals, and which relaxes and softens the small quantity of fibrine
which their muscles contain.
CAROLINE.
But, Mrs. B., there are some species of birds that frequent both
elements, as, for instance, ducks and other water fowl. Of what nature
is the flesh of these?
349
MRS. B.
Such birds, in general, make but little use of their wings; if they
fly, it is but feebly, and only to a short distance. Their flesh, too,
partakes of the oily nature, and even in taste sometimes resembles that
of fish. This is the case not only with the various kinds of water
fowls, but with all other amphibious animals, as the otter, the
crocodile, the lizard, &c.
CAROLINE.
And what is the reason that reptiles are so deficient in muscular
strength?
MRS. B.
It is because they usually live under ground, and seldom come into
the atmosphere. They have imperfect, and sometimes no discernible organs
of respiration; they partake therefore of the soft oily nature of fish;
indeed, many of them are amphibious, as frogs, toads, and snakes, and
very few of them find any difficulty in remaining a length of time under
water. Whilst, on the contrary, the insect tribe, that are so strong in
proportion to their size, and alert in their motions, partake of the
nature of birds, air being their peculiar element, and their organs of
respiration being comparatively larger than in other classes of
animals.
350
I have now given you a short account of the principal animal
functions. However interesting the subject may appear to you,
a fuller investigation of it would, I fear, lead us too far
from our object.
EMILY.
Yet I shall not quit it without much regret; for of all the branches
of chemistry, it is certainly the most curious and most interesting.
CAROLINE.
But, Mrs. B., I must remind you that you promised to give us
some account of the nature of milk.
MRS. B.
True. There are several other animal productions that deserve
likewise to be mentioned. We shall begin with milk, which is certainly
the most important and the most interesting of all the animal
secretions.
Milk, like all other animal substances, ultimately yields by analysis
oxygen, hydrogen, carbon, and nitrogen. These are combined in it under
the forms of albumen, gelatine, oil, and water. But milk contains,
besides, a considerable portion of phosphat of lime, the purposes
of which I have already pointed out.
351
CAROLINE.
Yes; it is this salt which serves to nourish the tender bones of the
suckling.
MRS. B.
To reduce milk to its elements, would be a very complicated, as well
as useless operation; but this fluid, without any chemical assistance,
may be decomposed into three parts, cream, curds, and
whey. These constituents of milk have but a very slight affinity
for each other, and you find accordingly that cream separates from milk
by mere standing. It consists chiefly of oil, which being lighter than
the other parts of the milk, gradually rises to the surface. It is of
this, you know, that butter is made, which is nothing more than
oxygenated cream.
CAROLINE.
Butter, then, is somewhat analogous to the waxy substance formed by
the oxygenation of vegetable oils.
MRS. B.
Very much so.
EMILY.
But is the cream oxygenated by churning?
MRS. B.
Its oxygenation commences previous to churning,
352
merely by standing exposed to the atmosphere, from which it absorbs
oxygen. The process is afterwards completed by churning; the violent
motion which this operation occasions brings every particle of cream in
contact with the atmosphere, and thus facilitates its oxygenation.
CAROLINE.
But the effect of churning, I have often observed in the dairy, is to
separate the cream into two substances, butter and butter-milk.
MRS. B.
That is to say, in proportion as the oily particles of the cream
become oxygenated, they separate from the other constituent parts of the
cream in the form of butter. So by churning you produce, on the one
hand, butter, or oxygenated oil; and, on the other, butter-milk, or
cream deprived of oil. But if you make butter by churning new milk
instead of cream, the butter-milk will then be exactly similar in its
properties to creamed or skimmed milk.
CAROLINE.
Yet butter-milk is very different from common skimmed milk.
MRS. B.
Because you know it is customary, in order to
353
save time and labour, to make butter from cream alone. In this case,
therefore, the butter-milk is deprived of the creamed milk, which
contains both the curd and whey. Besides, in consequence of the milk
remaining exposed to the atmosphere during the separation of the cream,
the latter becomes more or less acid, as well as the butter-milk which
it yields in churning.
EMILY.
Why should not the butter be equally acidified by oxygenation?
MRS. B.
Animal oil is not so easily acidified as the other ingredients of
milk. Butter, therefore, though usually made of sour cream, is not sour
itself, because the oily part of the cream had not been acidified.
Butter, however, is susceptible of becoming acid by an excess of oxygen;
it is then said to be rancid, and produces the sebacic acid, the same as
that which is obtained from fat.
EMILY.
If that be the case, might not rancid butter be sweetened by mixing
with it some substance that would take the acid from it?
MRS. B.
This idea has been suggested by Sir H. Davy,
354
who supposes, that if rancid butter were well washed in an alkaline
solution, the alkali would separate the acid from the butter.
CAROLINE.
You said just now that creamed milk consisted of curd and whey. Pray
how are these separated?
MRS. B.
They may be separated by standing for a certain length of time
exposed to the atmosphere; but this decomposition may be almost
instantaneously effected by the chemical agency of a variety of
substances. Alkalies, rennet*, and indeed almost all animal substances,
decompose milk by combining with the curds.
Acids and spirituous liquors, on the other hand, produce a
decomposition by combining with the whey. In order, therefore, to obtain
the whey pure, rennet, or alkaline substances, must be used to attract
the curds from it.
But if it be wished to obtain the curds pure, the whey must be
separated by acids, wine, or other spirituous liquors.
355
EMILY.
This is a very useful piece of information; for I find white-wine
whey, which I sometimes take when I have a cold, extremely heating; now,
if the whey were separated by means of an alkali instead of wine, it
would not produce that effect.
MRS. B.
Perhaps not. But I would strenuously advise you not to place too much
reliance on your slight chemical knowledge in medical matters. I do
not know why whey is not separated from curd by rennet, or by an alkali,
for the purpose which you mention; but I strongly suspect that there
must be some good reason why the preparation by means of wine is
generally preferred. I can, however, safely point out to you a
method of obtaining whey without either alkali, rennet, or wine; it is
by substituting lemon juice, a very small quantity of which will
separate it from the curds.
Whey, as an article of diet, is very wholesome, being remarkable
light of digestion. But its effect, taken medicinally, is chiefly,
I believe, to excite perspiration, by being drunk warm on going to
bed.
From whey a substance may be obtained in crystals by evaporation,
called sugar of milk. This substance is sweet to the taste, and
in its composition is so analogous to common sugar,
356
that it is susceptible of undergoing the vinous fermentation.
CAROLINE.
Why then is not wine, or alcohol, made from whey?
MRS. B.
The quantity of sugar contained in milk is so trifling, that it can
hardly answer that purpose. I have heard of only one instance of
its being used for the production of a spirituous liquor, and this is by
the Tartan
Arabs; their abundance of horses, as well as their scarcity of fruits,
has introduced the fermentation of mares’ milk, by which they produce a
liquor called koumiss. Whey is likewise susceptible of being
acidified by combining with oxygen from the atmosphere. It then produces
the lactic acid, which you may recollect is mentioned amongst the
animal acids, as the acid of milk.
Let us now see what are the properties of curds.
EMILY.
I know that they are made into cheese; but I have heard that for that
purpose they are separated from the whey by rennet, and yet this you
have just told us is not the method of obtaining pure curds?
MRS. B.
Nor are pure curds so well adapted for the formation
357
of cheese. For the nature and flavour of the cheese depend, in a great
measure, upon the cream or oily matter which is left in the curds; so
that if every particle of cream be removed from the curds, the cheese is
scarcely eatable. Rich cheeses, such as cream and Stilton cheeses,
derive their excellence from the quantity, as well as the quality, of
the cream that enters into their composition.
CAROLINE.
I had no idea that milk was such an interesting compound. In many
respects there appears to me to be a very striking analogy between milk
and the contents of an egg, both in respect to their nature and their
use. They are, each of them, composed of the various substances
necessary for the nourishment of the young animal, and equally destined
for that purpose.
MRS. B.
There is, however, a very essential difference. The young animal is
formed, as well as nourished, by the contents of the egg-shell; whilst
milk serves as nutriment to the suckling, only after it is born.
There are several peculiar animal substances which do not enter into the
general enumeration of animal compounds, and which, however, deserve to
be mentioned.
358
Spermaceti is of this class; it is a kind of oily substance
obtained from the head of the whale, which, however, must undergo a
certain preparation before it is in a fit state to be made into candles.
It is not much more combustible than tallow, but it is pleasanter to
burn, as it is less fusible and less greasy.
Ambergris is another peculiar substance derived from a species
of whale. It is, however, seldom obtained from the animal itself, but is
generally found floating on the surface of the sea.
Wax, you know, is a concrete oil, the peculiar product of the
bee, part of the constituents of which may probably be derived from
flowers, but so prepared by the organs of the bee, and so mixed with its
own substance, as to be decidedly an animal product. Bees’ wax is
naturally of a yellow colour, but it is bleached by long exposure to the
atmosphere, or may be instantaneously whitened by the oxy-muriatic acid.
The combustion of wax is far more perfect than that of tallow, and
consequently produces a greater quantity of light and heat.
Lac is a substance very similar to wax in the manner of its
formation; it is the product of an insect, which collects its
ingredients from flowers, apparently for the purpose of protecting its
eggs from injury. It is formed into cells, fabricated with as much skill
as those of the honey-comb,
359
but differently arranged. The principal use of lac is in the manufacture
of sealing-wax, and in making varnishes and lacquers.
Musk, civet, and castor, are other particular
productions, from different species of quadrupeds. The two first are
very powerful perfumes; the latter has a nauseous smell and taste, and
is only used medicinally.
CAROLINE.
Is it from this substance that castor oil is obtained?
MRS. B.
No. Far from it, for castor oil is a vegetable oil, expressed from
the seeds of a particular plant; and has not the least resemblance to
the medicinal substance obtained from the castor.
Silk is a peculiar secretion of the silk-worm, with which it
builds its nest or cocoon. This insect was originally brought to Europe
from China. Silk, in its chemical nature, is very similar to the hair
and wool of animals; whilst in the insect it is a fluid, which is
coagulated, apparently by uniting with oxygen, as soon as it comes in
contact with the air. The moth of the silk-worm ejects a liquor which
appears to contain a particular acid, called bombic, the
properties of which are but very little known.
360
EMILY.
Before we conclude the subject of the animal economy, shall we not
learn by what steps dead animals return to their elementary state?
MRS. B.
Animal matter, although the most complicated of all natural
substances, returns to its elementary state by one single spontaneous
process, the putrid fermentation. By this, the albumen, fibrine,
&c. are slowly reduced to the state of oxygen, hydrogen, nitrogen,
and carbon; and thus the circle of changes through which these
principles have passed is finally completed. They first quitted their
elementary form, or their combination with unorganised matter, to enter
into the vegetable system. Hence they were transmitted to the animal
kingdom; and from this they return, again to their primitive simplicity,
soon to re-enter the sphere of organised existence.
When all the circumstances necessary to produce fermentation do not
take place, animal, like vegetable matter, is liable to a partial or
imperfect decomposition, which converts it into a combustible substance
very like spermaceti. I dare say that Caroline, who is so fond of
analogies, will consider this as a kind of animal bitumen.
361
CAROLINE.
And why should I not, since the processes which produce these
substances are so similar?
MRS. B.
There is, however, one considerable difference; the state of bitumen
seems permanent, whilst that of animal substances, thus imperfectly
decomposed, is only transient; and unless precautions be taken to
preserve them in that state, a total dissolution infallibly ensues.
This circumstance, of the occasional conversion of animal matter into a
kind of spermaceti, is of late discovery. A manufacture has in
consequence been established near Bristol, in which, by exposing the
carcases of horses and other animals for a length of time under water,
the muscular parts are converted into this spermaceti-like substance.
The bones afterwards undergo a different process to produce hartshorn,
or, more properly, ammonia, and phosphorus; and the skin is prepared for
leather.
Thus art contrives to enlarge the sphere of useful purposes, for
which the elements were intended by nature; and the productions of the
several kingdoms are frequently arrested in their course, and variously
modified, by human skill, which compels them to contribute, under new
forms, to the necessities or luxuries of man.
But all that we enjoy, whether produced by the
362
spontaneous operations of nature, or the ingenious efforts of art,
proceed alike from the goodness of Providence.—To God alone man owes the admirable faculties which
enable him to improve and modify the productions of nature, no less than
those productions themselves. In contemplating the works of the
creation, or studying the inventions of art, let us, therefore, never forget the Divine Source
from which they proceed; and thus every acquisition of knowledge will
prove a lesson of piety and virtue.
END.
Printed by A. Strahan,
Printers-Street, London.
_Diagram from Conversation XIII_
Original Compound
Sulphat of Soda.
Soda 8 Sulphuric Acid
|
| Quies-
|
| cent
|
Result Result
Nitrat Divellent Attractions 6} 13 Sulphat
of Soda of Lime
|
|
| Attrac-
|
| tions
|
Nitric Acid 4 Lime
--
12
Original Compound
Nitrat of Lime.
363
Index links lead only to the top of the page, not to the exact item
referenced. Some browsers may not display page numbers correctly. If a
link seems to lead to the wrong page, the link is right and the visible
page number is wrong.
A
B
C
D
E
F
G
H
IJ
K
L
M
N
O
P
Q
R
S
T
V
U
W
Y
Z
U, V are alphabetized as shown. J is not separated from I.
|
A
Absorbent vessels, ii. 304
Absorption of caloric, i. 59.
66
Acetic acid, ii. 75.
197
Acetous fermentation, ii.
232
—— acid, ii. 193.
232
Acidulous gaseous mineral waters, ii. 129
—— salts, ii.
200
Acids, i. 262. ii. 69
Aeriform, i. 36
Affinity, i. 19. ii. 1
Agate, ii. 51
Agriculture, ii. 252
Air, i. 182. ii. 262
Albumen, ii. 277.
288
Alburnum, ii. 267
Alchemists, i. 4
Alcohol, or spirit of wine, ii. 215.
222
Alembic, i. 258
Alkalies, ii. 19
Alkaline earths, ii. 50.
58
Alloys, i. 344
Alum, or sulphat of alumine, ii. 55.
95
Alumine, ii. 54
Alumium, i. 13
Amalgam, i. 347
Ambergris, ii. 358
Amethyst, ii. 58
Amianthus, ii. 66
Ammonia, or volatile alkali, i. 363. ii. 20.
35
Ammoniacal gas, ii. 36
Ammonium, i. 13
Analysis, i. 287
—— of vegetables, ii.
165
Animals, ii. 276
Animal acids, ii. 75.
290
—— colours, ii.
292
—— heat, ii. 337
—— oil, ii. 178.
283
Animalization, ii. 276.
297.
315
Antidotes, ii. 41.
87
Antimony, i. 14
Aqua fortis, ii. 105
—— regia, i. 340.
ii. 144
Arrack,
ii. 220
Argand’s Lamp, i. 208
Arsenic, i. 14.
340.
348
Arteries, ii. 304.
323
Arterial blood, ii. 305.
326.
338
Asphaltum, ii. 240
Assafœtida, ii. 188
Assimilation, ii. 298
Astringent principle, ii.
198
Atmosphere, i. 90.
181. ii. 262
Atmospherical air, i. 182
Attraction of aggregation, or cohesion, i. 16. ii. 2
364
—— of composition, i. 16. ii. 1
Azot, or nitrogen, i. 182,
ii. 100
Azotic gas, i. 182
B
Index top
Balsams, ii. 165.
188
Balloons, i. 245
Bark, ii. 193.
265
Barytes, ii. 44.
58.
61
Bases of acids, i. 263.
ii. 69
—— gases, i. 183
—— salts, ii. 5
Beer, ii. 212.
220
Benzoic acid, ii. 74.
197
Bile, ii. 308
Birds, ii. 347
Bismuth, i. 14
Bitumens, ii. 239
Black lead, or plumbago, i. 304
Bleaching, i. 32. ii. 89.
140.
Blow-pipe, i. 324. ii. 226
Blood, ii. 306.
317
Blood-vessels, ii. 298
Boiling water, i. 93
Bombic acid, ii. 75.
290
Bones, ii. 298, 299
Boracic acid, i. 365. ii. 131
Boracium, i. 13. ii. 132
Borat of soda, ii. 133
Brandy, ii. 218
Brass, i. 344
Bread, ii. 233
Bricks, ii. 56
Brittle-metals, i. 14
Bronze, i. 341
Butter, ii. 351
Butter-milk, ii. 352
C
Index top
Calcareous earths, ii. 65
—— stones, ii.
123
Calcium, i. 13
Caloric, i. 12.
33
——, absorption of, i. 66
——, conductors of, i. 70
——, combined, i.
122
——, expansive power of i. 35
——, equilibrium of, i. 50
——, reflexion of, i. 54.
67
——, radiation of, i. 52.
61
——, solvent power of, i. 96.
102
——, capacity for, i. 124
Calorimeter, i. 156
Calx, i. 183
Camphor, ii. 165.
185
Camphoric acid, ii. 74.
197
Caoutchouc, ii. 165.
189
Carbonats, ii. 25.
129
Carbonat of ammonia, ii. 41
—— lead, i. 320
—— lime, ii. 59.
130
—— magnesia, ii.
67
—— potash, ii. 25
Carbonated hydrogen gas, i. 302
Carbon, i. 282. ii. 329
Carbonic acid, i. 290.
359. ii. 327
Carburet of iron, i. 304.
342
Carmine, ii. 295
Cartilage, ii. 303
Castor, ii. 359
Cellular membrane, ii. 311
Caustics, i. 349
Chalk, ii. 62.
123
Charcoal, i. 282
Cheese, ii. 356
Chemical attraction, i. 15.
ii. 9
Chemistry, i. 3
Chest, ii. 318
China, ii. 54
Chlorine, i. 214
Chrome, i. 14.
340
Chyle, ii. 305.
317
Chyme, ii. 316
365
Citric acid, ii. 74.
197
Circulation of the blood, ii. 322
Civet, ii. 359
Clay, i. 48. ii. 55
Coke, ii. 241
Coal, ii. 240.
252
Cobalt, i. 14
Cochineal, ii. 295
Cold, i. 50.
58
—— from evaporation, i. 102.
113.
150
Colours of metallic oxyds, i. 319
Columbium, i. 14.
340.
348
Combined caloric, i. 122
Combustion, i. 190
——, volatile products of, i. 207
——, fixed products of, i. 207
——, of alcohol, ii.
225
——, of ammoniacal gas, ii. 42
——, of boracium, ii.
133
——, by oxymuriatic acid or chlorine, ii. 142
——, of carbon, i. 289
——, of coals, i. 207.
297
——, of charcoal by nitric acid, ii. 102
——, of candles, i. 236.
309. ii. 179
——, of diamonds, i. 292
——, of ether, ii.
230
——, of hydrogen, i. 229.
——, of iron, i. 200.
322
——, of metals, i. 321
——, of oils, i. 208. ii. 178.
309
——, of oil of turpentine by nitrous acid, ii. 6
——, of phosphorus, i. 272
——, of sulphur, i. 261
—— of potassium, i. 358. ii. 132.
138, 139
Compound bodies, i. 9. ii. 14
—— or neutral salts i. 333. ii. 4
Conductors of heat, i. 71
——, solids, i. 73
——, fluids, i. 78
——, Count Rumford’s theory, i. 79
Constituent parts, i. 9
Copper, i. 14.
331
Copal, ii. 187.
224
Cortical layers, ii. 265.
267
Cotyledons, or lobes, ii. 256
Cream, ii. 351
Cream of tartar, or tartrit of potash, ii. 200.
222
Cryophorus, i. 154
Crystallisation, i. 338. ii. 47
Cucurbit, i. 258
Culinary heat, i. 88
Curd, ii. 351.
354
Cuticle, or epidermis, ii.
310
D
Index top
Decomposition, i. 8.
20
—— of atmospherical air, i. 181.
209
—— of water by the Voltaic battery, i. 220
—— of salts by the Voltaic battery, ii. 14
—— of water by metals, i. 225.
334
—— —— by carbon, i. 301
—— of vegetables, ii. 202
—— of potash, i. 356
—— of soda, i. 56
—— of ammonia,
i. 363.
ii. 37
366
—— of the boracic acid, ii. 132
—— of the fluoric acid, ii. 136
—— of the muriatic acid, ii. 139
Deflagration, ii. 118
Definite proportions, ii. 13
Deliquescence, ii. 95
Detonation, i. 219. ii. 116
Dew, i. 105
Diamond, i. 285
Diaphragm, ii. 320
Digestion, ii. 316
Dissolution of metals, i. 165.
316.
333
Distillation, i. 259. ii. 218
—— of red wine, ii.
218
Divellent forces, ii. 12
Division, i. 7
Drying oils, ii. 181
Dying, ii. 191
E
Index top
Earths, ii. 44
Earthen-ware, ii. 53.
57
Effervescence, i. 298
Efflorescence, ii. 94
Elastic fluids, i. 37
Electricity, i. 12.
25.
160.
220. ii. 139
Electric machine, i. 169
Elective attractions, ii. 9
Elementary bodies, i. 8.
12
Elixirs, tinctures, or quintessences, ii. 225
Enamel, ii. 57
Epidermis of vegetables, ii.
269
—— of animals, ii.
310
Epsom salts, ii. 63.
95
Equilibrium of caloric, i. 50
Essences, i. 307. ii. 183.
224
Essential, or volatile oils, i. 307. ii. 183
Ether, i. 111. ii. 229
Evaporation, i. 103
Evergreens, ii. 274
Eudiometer, i. 276
Expansion of caloric, i. 36
Extractive colouring matter, ii. 165.
190
F
Index top
Falling stones, i. 319
Fat, i. 306. ii. 311
Feathers, ii. 300
Fecula, ii. 176
Fermentation, ii. 205
Fibrine, ii. 277.
289
Fire, i. 7.
27
Fish, ii. 346
Fixed air, or carbonic acid, i. 290. ii. 125
—— alkalies, ii.
20
—— oils, i. 307.
ii. 165.
177
—— products of combustion, i. 207
Flame, i. 237
Flint, ii. 30.
51
Flower or blossom, ii. 271
Fluoric acid, ii. 54.
134
Fluorium, or Fluorine, i. 12. ii. 136
Formic acid, ii. 290
Fossil wood, ii. 242
Frankincense,
ii. 187
Free or radiant caloric, or heat of temperature, i. 33
Freezing mixtures, i. 142
—— by evaporation, i. 104.
150, &c.
Frost, i. 94
Fruit, ii. 271
Fuller’s earth, ii. 55
Furnace, i. 304
367
G
Index top
Galls, ii. 199
Gallat of iron, ii. 98
Gallic acid, ii. 74.
197, 198
Galvanism, i. 163
Gas, i. 182
Gas-lights, i. 240
Gaseous oxyd of carbon, i. 296
—— nitrogen, ii.
111
Gastric juice, ii. 316
Gelatine, or jelly, ii. 277.
280
Germination, ii. 256
Gin, ii. 221
Glands, ii. 298.
307
Glass, ii. 30
Glauber’s salts, or sulphat of soda, ii. 92
Glazing, ii. 57
Glucium, i. 13
Glue, ii. 281.
287
Gluten, ii. 165.
177
Gold, i. 14.
323
Gum, ii. 170
—— arabic, ii.
170
—— elastic, or caoutchouc, ii. 189
—— resins, ii. 165.
188
Gunpowder, ii. 116
Gypsum, or plaister of Paris, or sulphat of lime, ii. 95
H
Index top
Hair, ii. 300
Harrogate water, i. 268.
341
Hartshorn, ii. 35.
39.
281.
285
Heart, ii. 323
—— wood, ii. 268
Heat, i. 26.
33
—— of capacity, i. 127.
135
—— of temperature, i. 33
Honey, ii. 175
Horns, ii. 282.
300
Hydro-carbonat, i. 241.
303
Hydrogen, i. 214
—— gas, i. 215
IJ
Index top
Jasper, ii. 51
Ice, i. 138
Jelly, ii. 281
Jet, ii. 240
Ignes fatui, i. 277
Ignition, i. 119
Imponderable agents, i. 12
Inflammable air, i. 215
Ink, ii. 98.
199
Insects, ii. 349
Integrant pans, i. 9
Iridium, i. 14
Iron, i. 14.
319.
328
Isinglass, ii. 194.
285
Ivory black, ii. 295
Iodine, i. 214. ii. 157
K
Index top
Kali, ii. 34
Koumiss, ii. 356
|
L
Lac, ii. 358
Lactic acid, ii. 75.
290.
356
Lakes, colours, ii. 190
Latent heat, i. 133
Lavender water, ii. 184.
224
Lead, i. 14.
318.
330
Leather, ii. 193.
287
Leaves, ii. 260
Life, ii. 159.
168
Ligaments, ii. 303
Light, i. 12.
26. ii. 261
Lightning, i. 248
Lime, ii. 59
—— water, ii. 61
368
Limestone, ii. 60
Linseed oil, ii. 178
Liqueurs, ii. 224
Liver, ii. 308
Lobes, ii. 256.
332
Lunar caustic, or nitrat of silver, i. 350. ii. 119
Lungs, ii. 319.
330
Lymph, ii. 304
Lymphatic vessels, ii. 304
M
Index top
Magnesia, ii. 44.
66
Magnium, i. 13
Malic acid, ii. 74.
197
Malt, ii. 211
Malleable metals, i. 14
Manganese, i. 14.
317
Manna, ii. 176
Manure, ii. 247
Marble, ii. 123
Marine acid, or muriatic acid, ii. 136
Mastic, ii. 187.
224
Materials of animals, ii. 277
—— of vegetables, ii.
165
Mercury, i. 14.
346
——, new mode of freezing, i. 155.
347
Metallic acids, i. 340
—— oxyds, i. 316
Metals, i. 12.
314
Meteoric stones, i. 342
Mica, ii. 66
Milk, ii. 299.
306.
350
Minerals, i. 315. ii. 44.
158
Mineral waters, i. 296. ii. 129
—— acids, ii. 73
Miner’s lamp, i. 249
Mixture, i. 99
Molybdena, i. 14.
340
Mordant, ii. 165.
192
Mortar, ii. 53.
65
Mucilage, ii. 170
Mucous acid, ii. 74.
171.
197
—— membrane, ii.
311
Muriatic acid, or marine acid, ii. 136
Muriats, ii. 151
Muriat of ammonia, ii. 35.
152
—— lime, i. 100
—— soda, or common salt, ii. 136.
151
—— potash, ii.
138
Muriatium, i. 13
Muscles of animals, ii. 298.
303
Musk, ii. 359
Myrrh, ii. 188
N.
Index top
Naphtha, i. 357. ii. 240
Negative electricity, i. 25.
161.
185
Nerves, ii. 279.
298.
308
Neutral, or compound salts, i. 333. ii. 4.
22.
69
Nickel, i. 13.
343
Nitre, or nitrat of potash, or saltpetre, ii. 32.
104.
116
Nitric acid, ii. 100
Nitrogen, or azot, i. 181.
ii. 100
—— gas, i. 182.
211
Nitro-muriatic acid, or aqua regia, ii. 144
Nitrous acid gas, ii. 101.
106
—— air, or nitrit oxyd gas, ii. 107
Nitrats, ii. 116
Nitrat of copper, ii. 5
—— ammonia, ii. 113.
118
—— potash, or nitre, or saltpetre, ii. 32.
104.
116
—— silver, or lunar caustic, ii. 19
Nomenclature of acids, i. 264. ii. 69
—— compound salts, ii. 4.
22
369
—— other binary compounds, i. 278
Nut-galls, ii. 98.
199
Nut-oil, ii. 178
Nutrition, ii. 297
O
Index top
Ochres, i. 320
Oils, i. 285. ii. 306
Oil of amber, ii. 241
—— vitriol, or sulphuric acid, ii. 80
Olive oil, ii. 178
Ores, i. 315
Organized bodies, ii. 159
Organs of animals, ii. 290.
310
—— vegetables, ii.
159.
265.
271
Osmium, i. 14.
348
Oxalic acid, ii. 74.
197
Oxyds, i. 198
Oxyd of manganese, i. 117.
317
—— iron, i. 204.
319
—— lead, i. 319
—— sulphur, ii.
91
Oxydation, or oxygenation, i. 196
Oxygen, i. 11.
181.
201.
211
—— gas, or vital air, i. 182.
201
Oxy-muriatic acid, ii. 140
Oxy-muriats, ii. 153
Oxy-muriat of potash, ii. 155
P
Index top
Palladium, i. 13.
348
Papin’s digester, i. 120.
ii. 284
Parenchyma, ii. 256.
266
Particles, i. 16
Pearlash, ii. 24
Peat, ii. 242
Peculiar juice of plants, ii. 268
Perfect metals, i. 14.
324
Perfumes, i. 308. ii. 183
Perspiration, ii. 333.
329
Petrification, ii. 237
Pewter, i. 344
Pharmacy, i. 14
Phosphat of lime, ii. 99.
299
Phosphorated hydrogen gas, i. 277
Phosphorescence, i. 29
Phosphoric acid, i. 273. ii. 99
Phosphorous acid, i. 274.
ii. 99
Phosphorus, i. 270
Phosphoret of lime, i. 278.
341
—— sulphur, i. 279.
341
Pitch, ii. 187
Plaster, ii. 65
Platina, i. 14.
323
Plating, i. 345
Plumbago, or black lead, i. 304
Plumula, ii. 257
Porcelain, ii. 56
Positive electricity, i. 25.
161.
185
Potassium, i. 13.
357. ii. 15
Pottery, ii. 56
Potash, i. 356. ii. 22
Precipitate, i. 22
Pressure of the atmosphere, i. 112.
116
Printer’s ink, ii. 144
Prussiat of iron, or prussian blue, ii. 291
—— potash, ii.
291
Prussic acid, ii. 75.
290
Putrid fermentation, ii. 235.
360
Pyrites, i. 341. ii. 97
Pyrometer, i. 38.
42
Q
Index top
Quick lime, ii. 59
Quiescent forces, ii. 12
370
R
Index top
Radiation of caloric, i. 52
——, Prevost’s theory, i. 52
——, Pictet’s explanations, i. 54
——, Leslie’s illustrations, i. 61
Radicals, ii. 5.
69
Radicle; or root, ii. 257
Rain, i. 104
Rancidity, ii. 182
Rectification, ii. 223
Reflexion of caloric, i. 54.
64
Reptiles, ii. 349
Resins, ii. 165, 186. 266
Respiration, ii. 317.
326
Reviving of metals, i. 327
Rhodium, i. 14.
348
Roasting metals, i. 316
Rock crystal, ii. 61
Ruby, ii. 53
Rum, ii. 219
Rust, i. 318.
328
S
Index top
Saccharine fermentation, ii.
208
Sal ammoniac, or muriat of ammonia, ii. 35
—— polychrest, or sulphat of potash, ii. 91
—— volatile, or carbonat of ammonia, ii. 41
Salifiable bases, ii. 5
Salifying principles, ii. 5
Saltpetre, or nitre, or nitrat of potash, ii. 32.
104.
116
Salt, ii. 91
Sand, ii. 30.
51
Sandstone, ii. 51
Sap of plants, ii. 165.
260.
262.
270.
272
Sapphire, ii. 58
Saturation, i. 101.
Sapphire, ii. 58
Saturation, i. 101
Seas, temperature of, i. 33.
Sebacic acid, ii. 75.
182.
290.
353
Secretions, ii. 307
Seeds of plants, ii. 210.
271
Seltzer water, i. 289. ii. 63.
129
Senses, ii. 310
Silex, or silica, ii. 30.
51
Silicium, i. 13.
Silk, ii. 359
Silver, i. 321
Simple bodies, i. 10.
12
Size, ii. 281
Skin, ii. 279.
310.
193
Slakeing of lime, i. 147.
ii. 56
Slate, ii. 51.
66
Smelting metals, i. 316
Smoke i. 208
Soap, ii. 24
Soda, i. 363. ii. 33
—— water, i. 299
Sodium, i. 13.
363
Soils, i. 42. ii. 245
Soldering, i. 345
Solubility, ii. 92
Solution, i. 96
—— by the air, i. 102
—— of potash, ii.
28
Specific heat, i. 126
Spermaceti, ii. 358
Spirits, ii. 313
Steam, i. 140.
182
Steel, i. 305
Stomach, ii. 315
Stones, ii. 46
Stucco, ii. 65
Strontites, ii. 44.
68
Strontium, i. 13
Suberic acid, ii. 74.
197
Sublimation, i. 257
Succin, or yellow amber, ii. 241
Succinic acid, ii. 74.
197.
241
Sugar, ii. 165.
174.
208
—— of milk, ii.
355
371
Sulphats, ii. 5.
91
Super oxygenated sulphuric acid, ii. 70.
Sulphat of alumine, or alum, ii. 54.
95
—— barytes, ii.
58
—— iron, ii. 96
—— lime, or gypsum, or
plaster of Paris, ii. 95
—— magnesia, or Epsom salt, ii. 67.
95
—— potash, or sal polychrest, ii. 91
—— soda, or Glauber’s salts, ii. 92
Sulphur, i. 256
—— flowers of, i. 257
Sulphurated hydrogen gas, i. 165.
268
Sulphurets, i. 341
Sulphurous acid, i. 254. ii. 88
Sulphuric acid, i. 74. ii. 265
Sympathetic ink, i. 354
Synthesis, i. 287
T
Index top
Tan, ii. 192
Tannin, ii. 165.
192
Tar, ii. 187
Tartarous acid, ii. 74.
197
Tartrit of potash, ii. 222
Teeth, ii. 300
Tellurium, i. 14
Temperature, i. 33
Thaw, i. 158
Thermometers, i. 40
——, Fahrenheit’s, i.
42
——, Reaumur’s, i.
42
——, Centigrade,
i. 43
——, air, i. 44
——, differential,
i. 46
Thunder, i. 248
Tin, i. 14.
344
Titanium, i. 14.
348
Turf, ii. 242
Turpentine, ii. 187
Transpiration of plants, ii. 260
Tungsten, i. 14.
340
V
Index top
Vapour, i. 36.
49.
93.
182
Vaporisation, i. 103
Varnishes, ii. 187
Vegetables, ii. 158
Vegetable acid, i. 310. ii. 74.
197
—— colours, ii.
190
—— heat, ii. 272
—— oils, ii. 177
Veins, ii. 304.
323.
Venous blood, ii. 305.
326.
338
Ventricles, ii. 324
Verdigris, i. 352
Vessels, ii. 304
Vinegar, ii. 232
Vinous fermentation, ii. 212
Vital air, or oxygen gas, i. 182
Vitriol, or sulphat of iron, ii. 81
Volatile oils, i. 307. ii. 165.
183.
224.
269
—— products of combustion, i. 207
—— alkali, i. 363.
ii. 20.
35
Voltaic battery, i. 164.
220.
356. ii. 15
U
Index top
Uranium, i. 14
W
Index top
Water, i. 215. ii. 262
372
——, decomposition of, by electricity, i. 200.
225
——, condensation of, i. 32
—— of the sea, i. 86
——, boiling, i.
93
——, solution by, i. 96
—— of crystallisation, i. 339
Wax, i. 309. ii. 180.
358
Whey, ii. 351
Wine, ii. 212
Wood, ii. 267
Woody fibre, ii. 156.
196.
267
Wool, ii. 300
Y
Index top
Yeast, ii. 234.
Yttria, ii. 44.
Yttrium, i. 13.
Z
Index top
Zinc, ii. 14.
344
Zirconia,
ii. 44
Zirconium,
i. 14.
Zoonic acid, ii. 75.
220
|
Terminology
Many scientific terms used in this book are different from today’s
standard terminology. The following is not meant as a comprehensive
list.
oxy-muriatic acid |
chlorine
proposed as an element in 1815: see Conversation XIX |
“columbium or tantalium” |
niobium and tantalum
the two elements always occur together, and were not recognized as
separate until much later in the 19th century
|
phosphat of lime |
calcium diphosphate or calcium
the element calcium was isolated in 1808, but is named only once in
this 1817 edition
|
glucium |
beryllium
Humphry Davy’s name for the element |
muriatic acid |
hydrochloric acid
but still called “muriatic acid” for some commercial uses
|
muriat of lime |
calcium chloride |
oxymuriate of potash |
potassium chlorate |
muriat of soda |
sodium chloride (table salt) |
carbonic acid |
carbon dioxide |
Note also:
simple body, fundamental principle |
element |
fecula |
starch (usually spelled “fæcula”) |
spirit of wine |
alcohol |
philosopher |
scientist |
arts |
industry, manufacture, crafts etc. (seldom “fine arts”) |
Some essential concepts relating to living
things—photosynthesis, microorganisms, the cell,
proteins—are either unknown or not mentioned. The atom theory had
been proposed, but not by Humphry Davy; it is not mentioned in this
book.
The word “explode” is used at least once in its orginal, figurative
sense (“a word that should be exploded in chemistry”) but far more
often in its later, concrete one. The word “explosion” is always used
concretely (“an explosion, or a detonation as chemists commonly
call it”).
Calculated Values:
“the point of zero, or the absolute privation of heat, must
consequently be 1260 degrees below 32 degrees”
-1228° F. The calculation is based on wrong premises; the correct figure
is about -460° F or -273° C.
“Mercury congeals only at seventy-two degrees below the freezing
point.”
-40° F, which is also -40° C. This figure is correct, though
approximate.
“The proportion stated by Sir H. Davy, in his Chemical Researches, is
as 1 to 2.389.”
[ammonia] “consisted of about one part of hydrogen to four parts of
nitrogen.... and from the latest and most accurate experiments, the
proportions appear to be, one volume of nitrogen gas to three of
hydrogen gas”
These and similar calculations involving relative weight and volume make
more sense when one knows the elements’ atomic weights. For nitric acid,
HNO3, the figures are 1:14:48, giving a proportion closer to
1:3.5. For ammonia, NH3 (not 4), the figures are 14:3.
The first proportion was printed “2,389”. No other decimal numbers
occur in the text, but a comma appears once as a thousands
separator.
“The oxalic acid, distilled from sorrel, is the highest term
of
vegetable acidification; for, if more oxygen be added to it, it loses
its vegetable nature, and is resolved into carbonic acid and water;”
Oxalic acid = H2C2O4; carbonic acid
(carbon dioxide) = CO2.
H2C2O4 + O becomes H2O +
CO2 + CO2.
Chapter Numbering
The 3rd and 4th editions used the same Conversation (chapter)
numbering. The apparent disappearance of XI and XII is the result of
changes between the 4th and 5th (present text) editions:
|
Volume I: On Simple Bodies |
4th
edn. |
5th
edn. |
|
I.
II.
III. |
I.
II.
III. |
No change |
| IV. |
IV. |
4th: On Specific Heat, Latent Heat, and Chemical Heat.
5th: On Combined Caloric, Comprehending Specific Heat and
Latent Heat.
|
|
V. |
On The Chemical Agencies Of Electricity.
Chapter added in 5th edition |
| V. |
VI. |
On Oxygen And Nitrogen. |
| VI. |
VII. |
On Hydrogen.
5th: new sections on Gas lights and Miner’s Lamp |
| VII. |
VIII. |
On Sulphur And Phosphorus.
5th: new section on Decomposition of Sulphur
|
| VIII. |
IX. |
4th: On Carbone.
5th: On Carbon.
|
| IX. |
X. |
On Metals. |
| X. |
|
On Alkalies. |
| XI. |
|
On Earths.
Conversations X, XI were moved to Volume II as XIV, XV.
|
|
Volume II: On Compound Bodies
|
4th
edn. |
5th
edn. |
|
| XII. |
XIII. |
On The Attraction Of Composition. |
|
XIV. |
On Alkalies. |
|
XV. |
On Earths.
Conversations XIV, XV were previously X, XI in Volume I.
|
| XIII. |
XVI. |
4th: On Compound Bodies.
5th: On Acids.
Most of XIII, On Compound Bodies, became XVI, On Acids. Some
introductory material was moved to XIV, On Alkalies.
|
| XIV. |
XVII. |
4th: On The Combinations of Oxygen with Sulphur and with
Phosphorus; and of the Sulphats And Phosphats.
5th: Of the Sulphuric and Phosphoric Acids: or, The
Combinations of ....
|
| XV. |
XVIII. |
4th: On The Combination of Oxygen With Nitrogen and with
Carbone; and of The Nitrats And Carbonats.
5th: Of The Nitric And Carbonic Acids: Or The Combination
...
|
| XVI. |
XIX. |
4th: On Muriatic And Oxygenated Muriatic Acids; and on
Muriats.
5th: On The Boracic, Fluoric, Muriatic, and Oxygenated
Muriatic Acids; and on Muriats.
|
| XVII. |
XX. |
On The Nature And Composition Of Vegetables. |
|
Remainder of book: number in 4th edn. + 3 = number in 5th edn.
|
Plates
Most Plates include the following text, engraved in smaller
print:
Drawn by the Author / Engraved by Lowry / Published by Longman &
Co. Octr. 2nd. 1809.
This date corresponds to the 3rd edition. Plates V, X and
XIII—each containing material new to the 5th edition—read
only “Lowry sculp.”
Inconsistencies and variant spellings
None of these lists are meant to be inclusive. They are typical of
variations that were not marked as errors.
Standard spellings throughout the book:
bason, judgment, embrio, volcanos (plural), potatoe (singular)
Inconsistencies:
capitalization of “Fig.” or “fig.”
hyphenization of words such as “oxy-muriatic”
“glauber salt” and “Glauber’s salt” both occur
Variant forms:
opake, opaque
aëriform, aeriform
gasses, gases
phosphoret, phosphuret (but always carburet)
Libya, Lybia
dy(e)ing (from “dye”)
nap(h)tha
pla(i)ster
slak(e)ing
earthen-ware, earthen ware
“sulphurous”, “naphtha” are used in the Contents and the Index;
“sulphureous”, “naptha” in the body text
forms in “-xion” (such as “connexion”) appear only in the Contents
and the Index
Volume I has more archaic forms than Volume II:
“shew”, “inclose” are sometimes used instead of “show”, “enclose”
“carbone” with final “e” appears in one Plate caption. (In the same
plate’s header, the “e” appears to have been removed by the
engraver.)
“develope(ment)” is more common in Volume I, “develop(ment)” in
Volume II
“-ize” and “-yze” forms (for later “-ise” and “-yse”) are common in
Volume I, rare in Volume II except in the Index
The “Dr. Marcet” mentioned in a few footnotes and figure captions is the
author’s husband. Humphry Davy (“Sir H. Davy”) was knighted in 1812,
between the 3rd and 4th editions of the book.
Reminder
DO NOT TRY THIS AT HOME.
End of Project Gutenberg's Conversations on Chemistry, V. 1-2, by Jane Marcet
*** END OF THIS PROJECT GUTENBERG EBOOK CONVERSATIONS ON CHEMISTRY, V. 1-2 ***
***** This file should be named 26908-h.htm or 26908-h.zip *****
This and all associated files of various formats will be found in:
http://www.gutenberg.org/2/6/9/0/26908/
Produced by Louise Hope
Updated editions will replace the previous one--the old editions
will be renamed.
Creating the works from public domain print editions means that no
one owns a United States copyright in these works, so the Foundation
(and you!) can copy and distribute it in the United States without
permission and without paying copyright royalties. Special rules,
set forth in the General Terms of Use part of this license, apply to
copying and distributing Project Gutenberg-tm electronic works to
protect the PROJECT GUTENBERG-tm concept and trademark. Project
Gutenberg is a registered trademark, and may not be used if you
charge for the eBooks, unless you receive specific permission. If you
do not charge anything for copies of this eBook, complying with the
rules is very easy. You may use this eBook for nearly any purpose
such as creation of derivative works, reports, performances and
research. They may be modified and printed and given away--you may do
practically ANYTHING with public domain eBooks. Redistribution is
subject to the trademark license, especially commercial
redistribution.
*** START: FULL LICENSE ***
THE FULL PROJECT GUTENBERG LICENSE
PLEASE READ THIS BEFORE YOU DISTRIBUTE OR USE THIS WORK
To protect the Project Gutenberg-tm mission of promoting the free
distribution of electronic works, by using or distributing this work
(or any other work associated in any way with the phrase "Project
Gutenberg"), you agree to comply with all the terms of the Full Project
Gutenberg-tm License (available with this file or online at
http://gutenberg.org/license).
Section 1. General Terms of Use and Redistributing Project Gutenberg-tm
electronic works
1.A. By reading or using any part of this Project Gutenberg-tm
electronic work, you indicate that you have read, understand, agree to
and accept all the terms of this license and intellectual property
(trademark/copyright) agreement. If you do not agree to abide by all
the terms of this agreement, you must cease using and return or destroy
all copies of Project Gutenberg-tm electronic works in your possession.
If you paid a fee for obtaining a copy of or access to a Project
Gutenberg-tm electronic work and you do not agree to be bound by the
terms of this agreement, you may obtain a refund from the person or
entity to whom you paid the fee as set forth in paragraph 1.E.8.
1.B. "Project Gutenberg" is a registered trademark. It may only be
used on or associated in any way with an electronic work by people who
agree to be bound by the terms of this agreement. There are a few
things that you can do with most Project Gutenberg-tm electronic works
even without complying with the full terms of this agreement. See
paragraph 1.C below. There are a lot of things you can do with Project
Gutenberg-tm electronic works if you follow the terms of this agreement
and help preserve free future access to Project Gutenberg-tm electronic
works. See paragraph 1.E below.
1.C. The Project Gutenberg Literary Archive Foundation ("the Foundation"
or PGLAF), owns a compilation copyright in the collection of Project
Gutenberg-tm electronic works. Nearly all the individual works in the
collection are in the public domain in the United States. If an
individual work is in the public domain in the United States and you are
located in the United States, we do not claim a right to prevent you from
copying, distributing, performing, displaying or creating derivative
works based on the work as long as all references to Project Gutenberg
are removed. Of course, we hope that you will support the Project
Gutenberg-tm mission of promoting free access to electronic works by
freely sharing Project Gutenberg-tm works in compliance with the terms of
this agreement for keeping the Project Gutenberg-tm name associated with
the work. You can easily comply with the terms of this agreement by
keeping this work in the same format with its attached full Project
Gutenberg-tm License when you share it without charge with others.
1.D. The copyright laws of the place where you are located also govern
what you can do with this work. Copyright laws in most countries are in
a constant state of change. If you are outside the United States, check
the laws of your country in addition to the terms of this agreement
before downloading, copying, displaying, performing, distributing or
creating derivative works based on this work or any other Project
Gutenberg-tm work. The Foundation makes no representations concerning
the copyright status of any work in any country outside the United
States.
1.E. Unless you have removed all references to Project Gutenberg:
1.E.1. The following sentence, with active links to, or other immediate
access to, the full Project Gutenberg-tm License must appear prominently
whenever any copy of a Project Gutenberg-tm work (any work on which the
phrase "Project Gutenberg" appears, or with which the phrase "Project
Gutenberg" is associated) is accessed, displayed, performed, viewed,
copied or distributed:
This eBook is for the use of anyone anywhere at no cost and with
almost no restrictions whatsoever. You may copy it, give it away or
re-use it under the terms of the Project Gutenberg License included
with this eBook or online at www.gutenberg.org
1.E.2. If an individual Project Gutenberg-tm electronic work is derived
from the public domain (does not contain a notice indicating that it is
posted with permission of the copyright holder), the work can be copied
and distributed to anyone in the United States without paying any fees
or charges. If you are redistributing or providing access to a work
with the phrase "Project Gutenberg" associated with or appearing on the
work, you must comply either with the requirements of paragraphs 1.E.1
through 1.E.7 or obtain permission for the use of the work and the
Project Gutenberg-tm trademark as set forth in paragraphs 1.E.8 or
1.E.9.
1.E.3. If an individual Project Gutenberg-tm electronic work is posted
with the permission of the copyright holder, your use and distribution
must comply with both paragraphs 1.E.1 through 1.E.7 and any additional
terms imposed by the copyright holder. Additional terms will be linked
to the Project Gutenberg-tm License for all works posted with the
permission of the copyright holder found at the beginning of this work.
1.E.4. Do not unlink or detach or remove the full Project Gutenberg-tm
License terms from this work, or any files containing a part of this
work or any other work associated with Project Gutenberg-tm.
1.E.5. Do not copy, display, perform, distribute or redistribute this
electronic work, or any part of this electronic work, without
prominently displaying the sentence set forth in paragraph 1.E.1 with
active links or immediate access to the full terms of the Project
Gutenberg-tm License.
1.E.6. You may convert to and distribute this work in any binary,
compressed, marked up, nonproprietary or proprietary form, including any
word processing or hypertext form. However, if you provide access to or
distribute copies of a Project Gutenberg-tm work in a format other than
"Plain Vanilla ASCII" or other format used in the official version
posted on the official Project Gutenberg-tm web site (www.gutenberg.org),
you must, at no additional cost, fee or expense to the user, provide a
copy, a means of exporting a copy, or a means of obtaining a copy upon
request, of the work in its original "Plain Vanilla ASCII" or other
form. Any alternate format must include the full Project Gutenberg-tm
License as specified in paragraph 1.E.1.
1.E.7. Do not charge a fee for access to, viewing, displaying,
performing, copying or distributing any Project Gutenberg-tm works
unless you comply with paragraph 1.E.8 or 1.E.9.
1.E.8. You may charge a reasonable fee for copies of or providing
access to or distributing Project Gutenberg-tm electronic works provided
that
- You pay a royalty fee of 20% of the gross profits you derive from
the use of Project Gutenberg-tm works calculated using the method
you already use to calculate your applicable taxes. The fee is
owed to the owner of the Project Gutenberg-tm trademark, but he
has agreed to donate royalties under this paragraph to the
Project Gutenberg Literary Archive Foundation. Royalty payments
must be paid within 60 days following each date on which you
prepare (or are legally required to prepare) your periodic tax
returns. Royalty payments should be clearly marked as such and
sent to the Project Gutenberg Literary Archive Foundation at the
address specified in Section 4, "Information about donations to
the Project Gutenberg Literary Archive Foundation."
- You provide a full refund of any money paid by a user who notifies
you in writing (or by e-mail) within 30 days of receipt that s/he
does not agree to the terms of the full Project Gutenberg-tm
License. You must require such a user to return or
destroy all copies of the works possessed in a physical medium
and discontinue all use of and all access to other copies of
Project Gutenberg-tm works.
- You provide, in accordance with paragraph 1.F.3, a full refund of any
money paid for a work or a replacement copy, if a defect in the
electronic work is discovered and reported to you within 90 days
of receipt of the work.
- You comply with all other terms of this agreement for free
distribution of Project Gutenberg-tm works.
1.E.9. If you wish to charge a fee or distribute a Project Gutenberg-tm
electronic work or group of works on different terms than are set
forth in this agreement, you must obtain permission in writing from
both the Project Gutenberg Literary Archive Foundation and Michael
Hart, the owner of the Project Gutenberg-tm trademark. Contact the
Foundation as set forth in Section 3 below.
1.F.
1.F.1. Project Gutenberg volunteers and employees expend considerable
effort to identify, do copyright research on, transcribe and proofread
public domain works in creating the Project Gutenberg-tm
collection. Despite these efforts, Project Gutenberg-tm electronic
works, and the medium on which they may be stored, may contain
"Defects," such as, but not limited to, incomplete, inaccurate or
corrupt data, transcription errors, a copyright or other intellectual
property infringement, a defective or damaged disk or other medium, a
computer virus, or computer codes that damage or cannot be read by
your equipment.
1.F.2. LIMITED WARRANTY, DISCLAIMER OF DAMAGES - Except for the "Right
of Replacement or Refund" described in paragraph 1.F.3, the Project
Gutenberg Literary Archive Foundation, the owner of the Project
Gutenberg-tm trademark, and any other party distributing a Project
Gutenberg-tm electronic work under this agreement, disclaim all
liability to you for damages, costs and expenses, including legal
fees. YOU AGREE THAT YOU HAVE NO REMEDIES FOR NEGLIGENCE, STRICT
LIABILITY, BREACH OF WARRANTY OR BREACH OF CONTRACT EXCEPT THOSE
PROVIDED IN PARAGRAPH F3. YOU AGREE THAT THE FOUNDATION, THE
TRADEMARK OWNER, AND ANY DISTRIBUTOR UNDER THIS AGREEMENT WILL NOT BE
LIABLE TO YOU FOR ACTUAL, DIRECT, INDIRECT, CONSEQUENTIAL, PUNITIVE OR
INCIDENTAL DAMAGES EVEN IF YOU GIVE NOTICE OF THE POSSIBILITY OF SUCH
DAMAGE.
1.F.3. LIMITED RIGHT OF REPLACEMENT OR REFUND - If you discover a
defect in this electronic work within 90 days of receiving it, you can
receive a refund of the money (if any) you paid for it by sending a
written explanation to the person you received the work from. If you
received the work on a physical medium, you must return the medium with
your written explanation. The person or entity that provided you with
the defective work may elect to provide a replacement copy in lieu of a
refund. If you received the work electronically, the person or entity
providing it to you may choose to give you a second opportunity to
receive the work electronically in lieu of a refund. If the second copy
is also defective, you may demand a refund in writing without further
opportunities to fix the problem.
1.F.4. Except for the limited right of replacement or refund set forth
in paragraph 1.F.3, this work is provided to you 'AS-IS' WITH NO OTHER
WARRANTIES OF ANY KIND, EXPRESS OR IMPLIED, INCLUDING BUT NOT LIMITED TO
WARRANTIES OF MERCHANTIBILITY OR FITNESS FOR ANY PURPOSE.
1.F.5. Some states do not allow disclaimers of certain implied
warranties or the exclusion or limitation of certain types of damages.
If any disclaimer or limitation set forth in this agreement violates the
law of the state applicable to this agreement, the agreement shall be
interpreted to make the maximum disclaimer or limitation permitted by
the applicable state law. The invalidity or unenforceability of any
provision of this agreement shall not void the remaining provisions.
1.F.6. INDEMNITY - You agree to indemnify and hold the Foundation, the
trademark owner, any agent or employee of the Foundation, anyone
providing copies of Project Gutenberg-tm electronic works in accordance
with this agreement, and any volunteers associated with the production,
promotion and distribution of Project Gutenberg-tm electronic works,
harmless from all liability, costs and expenses, including legal fees,
that arise directly or indirectly from any of the following which you do
or cause to occur: (a) distribution of this or any Project Gutenberg-tm
work, (b) alteration, modification, or additions or deletions to any
Project Gutenberg-tm work, and (c) any Defect you cause.
Section 2. Information about the Mission of Project Gutenberg-tm
Project Gutenberg-tm is synonymous with the free distribution of
electronic works in formats readable by the widest variety of computers
including obsolete, old, middle-aged and new computers. It exists
because of the efforts of hundreds of volunteers and donations from
people in all walks of life.
Volunteers and financial support to provide volunteers with the
assistance they need, is critical to reaching Project Gutenberg-tm's
goals and ensuring that the Project Gutenberg-tm collection will
remain freely available for generations to come. In 2001, the Project
Gutenberg Literary Archive Foundation was created to provide a secure
and permanent future for Project Gutenberg-tm and future generations.
To learn more about the Project Gutenberg Literary Archive Foundation
and how your efforts and donations can help, see Sections 3 and 4
and the Foundation web page at http://www.pglaf.org.
Section 3. Information about the Project Gutenberg Literary Archive
Foundation
The Project Gutenberg Literary Archive Foundation is a non profit
501(c)(3) educational corporation organized under the laws of the
state of Mississippi and granted tax exempt status by the Internal
Revenue Service. The Foundation's EIN or federal tax identification
number is 64-6221541. Its 501(c)(3) letter is posted at
http://pglaf.org/fundraising. Contributions to the Project Gutenberg
Literary Archive Foundation are tax deductible to the full extent
permitted by U.S. federal laws and your state's laws.
The Foundation's principal office is located at 4557 Melan Dr. S.
Fairbanks, AK, 99712., but its volunteers and employees are scattered
throughout numerous locations. Its business office is located at
809 North 1500 West, Salt Lake City, UT 84116, (801) 596-1887, email
[email protected]. Email contact links and up to date contact
information can be found at the Foundation's web site and official
page at http://pglaf.org
For additional contact information:
Dr. Gregory B. Newby
Chief Executive and Director
[email protected]
Section 4. Information about Donations to the Project Gutenberg
Literary Archive Foundation
Project Gutenberg-tm depends upon and cannot survive without wide
spread public support and donations to carry out its mission of
increasing the number of public domain and licensed works that can be
freely distributed in machine readable form accessible by the widest
array of equipment including outdated equipment. Many small donations
($1 to $5,000) are particularly important to maintaining tax exempt
status with the IRS.
The Foundation is committed to complying with the laws regulating
charities and charitable donations in all 50 states of the United
States. Compliance requirements are not uniform and it takes a
considerable effort, much paperwork and many fees to meet and keep up
with these requirements. We do not solicit donations in locations
where we have not received written confirmation of compliance. To
SEND DONATIONS or determine the status of compliance for any
particular state visit http://pglaf.org
While we cannot and do not solicit contributions from states where we
have not met the solicitation requirements, we know of no prohibition
against accepting unsolicited donations from donors in such states who
approach us with offers to donate.
International donations are gratefully accepted, but we cannot make
any statements concerning tax treatment of donations received from
outside the United States. U.S. laws alone swamp our small staff.
Please check the Project Gutenberg Web pages for current donation
methods and addresses. Donations are accepted in a number of other
ways including checks, online payments and credit card donations.
To donate, please visit: http://pglaf.org/donate
Section 5. General Information About Project Gutenberg-tm electronic
works.
Professor Michael S. Hart is the originator of the Project Gutenberg-tm
concept of a library of electronic works that could be freely shared
with anyone. For thirty years, he produced and distributed Project
Gutenberg-tm eBooks with only a loose network of volunteer support.
Project Gutenberg-tm eBooks are often created from several printed
editions, all of which are confirmed as Public Domain in the U.S.
unless a copyright notice is included. Thus, we do not necessarily
keep eBooks in compliance with any particular paper edition.
Most people start at our Web site which has the main PG search facility:
http://www.gutenberg.org
This Web site includes information about Project Gutenberg-tm,
including how to make donations to the Project Gutenberg Literary
Archive Foundation, how to help produce our new eBooks, and how to
subscribe to our email newsletter to hear about new eBooks.